Advanced Synthesis of Sodium Sugammadex Intermediate for Commercial Scale-up
Advanced Synthesis of Sodium Sugammadex Intermediate for Commercial Scale-up
The pharmaceutical landscape for neuromuscular blocking reversal agents has been significantly transformed by the introduction of sugammadex sodium, a modified gamma-cyclodextrin capable of encapsulating steroidal muscle relaxants. Patent CN114181331B, published in mid-2023, introduces a pivotal advancement in the manufacturing of the key intermediate required for this blockbuster drug. This intellectual property details a novel synthetic route that converts gamma-cyclodextrin directly into octa-6-perdeoxy-6-perhalo-gamma-cyclodextrin (Compound I) using a thioether-halogen activation system. Unlike traditional methods that rely on cumbersome phosphine reagents, this approach leverages the unique reactivity of thioethers to facilitate halogenation under remarkably mild conditions. The structural complexity of the final API, as illustrated below, demands an intermediate of exceptional purity to ensure effective encapsulation of rocuronium or vecuronium without introducing toxic impurities.
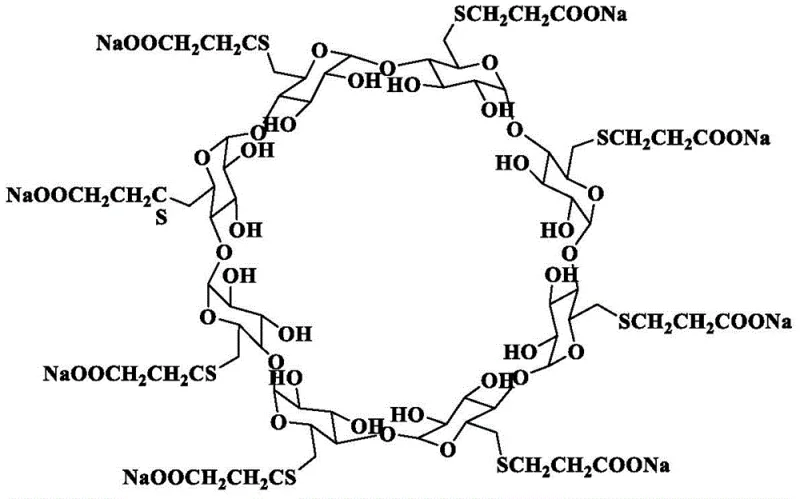
This technical breakthrough addresses the longstanding challenges associated with the functionalization of all eight primary hydroxyl groups on the cyclodextrin rim. By optimizing the activation step, the patent provides a pathway that is not only chemically elegant but also industrially viable. For R&D directors and process chemists, this represents a shift towards greener chemistry with higher atom economy. For procurement and supply chain leaders, it signals a potential stabilization of the supply chain for this critical anesthesia adjunct through more robust and cost-efficient manufacturing protocols. The following analysis dissects the mechanistic advantages and commercial implications of adopting this thioether-mediated halogenation strategy for the production of high-purity pharmaceutical intermediates.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the preparation of octa-6-perdeoxy-6-perhalo-gamma-cyclodextrin has relied heavily on halogenating agents such as phosphorus tribromide (PBr3), phosphorus pentachloride (PCl5), or combinations of elemental halogens with triphenylphosphine (TPP). While these methods are chemically feasible, they suffer from severe drawbacks when applied to large-scale industrial synthesis. The primary issue lies in the stoichiometry and the nature of the by-products. Since gamma-cyclodextrin possesses eight reactive sites, the reaction requires a substantial molar excess of halogenating reagents to drive the conversion to completion. When TPP is employed, it is oxidized to triphenylphosphine oxide (TPPO), a by-product with a high molecular weight and poor solubility profile that often co-precipitates with the product or forms emulsions during workup.
Furthermore, the removal of these phosphine oxide impurities is notoriously difficult, often necessitating multiple recrystallization steps or complex chromatographic separations that drastically reduce the overall yield. The use of aggressive reagents like PBr3 also poses significant safety hazards due to their corrosive nature and the evolution of acidic gases, requiring specialized equipment and stringent safety protocols. These factors collectively contribute to a process with low atom utilization, high waste generation, and elevated production costs, making the conventional routes less attractive for modern, sustainability-focused pharmaceutical manufacturing. The difficulty in achieving consistent purity levels above 98% using these legacy methods has been a persistent bottleneck in the supply chain for sugammadex intermediates.
The Novel Approach
The methodology disclosed in patent CN114181331B circumvents these issues by employing a thioether compound, such as dimethyl sulfide, in conjunction with elemental halogens (bromine or iodine). This system generates a reactive sulfonium species in situ that acts as a highly efficient halogen carrier, facilitating the substitution of the C6 hydroxyl groups on the gamma-cyclodextrin ring. The reaction proceeds under significantly milder conditions, typically between 20°C and 55°C for bromination, which minimizes thermal degradation of the sensitive carbohydrate backbone. Crucially, the by-products generated from the thioether oxidation are generally more soluble and easier to separate from the cyclodextrin derivative compared to solid phosphine oxides.
This novel approach allows for a streamlined workup procedure involving simple filtration and pH-adjusted crystallization, eliminating the need for extensive purification sequences. The patent data indicates that this method can achieve yields exceeding 90% with purity levels approaching 99.9%, a significant improvement over the 80% yields and lower purities often seen with phosphine-based routes. By replacing expensive and hazardous phosphine reagents with commodity chemicals like dimethyl sulfide and bromine, the process inherently reduces raw material costs and simplifies inventory management. This shift not only enhances the economic viability of the synthesis but also aligns with green chemistry principles by reducing the E-factor (mass of waste per mass of product) of the manufacturing process.
Mechanistic Insights into Thioether-Mediated Halogenation
The core of this innovation lies in the activation mechanism where the thioether acts as a nucleophilic catalyst for the halogenation of the primary alcohols. In the presence of a halogen source like bromine, the sulfur atom of the thioether attacks the halogen molecule to form a reactive sulfonium halide intermediate. This activated complex then interacts with the primary hydroxyl groups at the C6 position of the gamma-cyclodextrin. The reaction likely proceeds through an SN2-type mechanism where the hydroxyl group is converted into a good leaving group via the sulfonium species, followed by nucleophilic attack by the halide ion. This pathway avoids the formation of stable phosphine-oxygen bonds that characterize the traditional TPP method, thereby preventing the accumulation of hard-to-remove oxide impurities.
The selectivity for the primary C6 hydroxyls over the secondary C2 and C3 hydroxyls is driven by steric factors and the inherent higher nucleophilicity of the primary alcohols on the cyclodextrin rim. The reaction conditions are carefully tuned to ensure complete conversion of all eight sites without degrading the glycosidic linkages that hold the cyclic structure together. The use of polar aprotic solvents like N,N-dimethylformamide (DMF) or dimethyl sulfoxide (DMSO) is critical, as they stabilize the ionic intermediates and ensure the solubility of the bulky cyclodextrin substrate throughout the reaction. The subsequent addition of an alkali base, such as sodium methoxide, serves to neutralize any acidic by-products and induce the crystallization of the target halogenated intermediate by adjusting the pH to a range of 9 to 12.
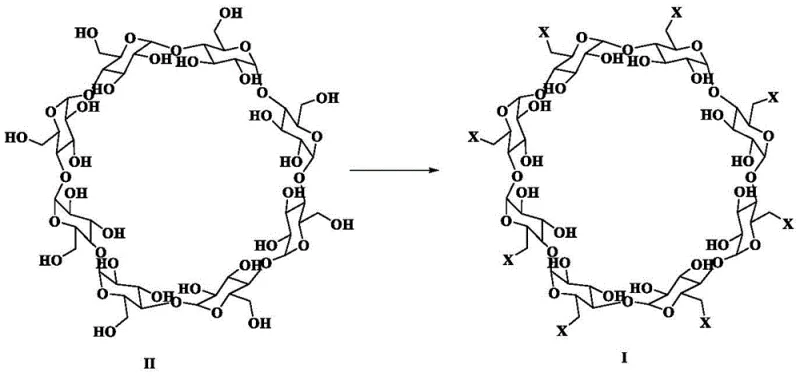
Impurity control is inherently built into this mechanism. Since the thioether reagents are volatile or form water-soluble oxidation products, they can be effectively removed during the aqueous workup or concentration steps. This contrasts sharply with the TPP method where the oxide by-product is a solid that mimics the physical properties of the product. The high purity achieved (often >99.8% as per the patent examples) suggests that side reactions such as over-halogenation or ring opening are minimal under the controlled temperature regimes specified. This mechanistic clarity provides R&D teams with a robust framework for troubleshooting and further optimization, ensuring that the process remains stable even upon scale-up to multi-ton batches.
How to Synthesize Octa-6-perdeoxy-6-perbromo-gamma-cyclodextrin Efficiently
The synthesis of this critical intermediate is now more accessible due to the optimized protocol described in the patent. The process begins with the preparation of the active halogenating solution, followed by the controlled addition of the cyclodextrin substrate. Maintaining strict temperature control during the exothermic mixing of the thioether and halogen is essential to prevent the formation of side products. Once the reaction is complete, the isolation of the product is facilitated by a pH-swing crystallization technique that leverages the solubility differences between the target molecule and the reaction matrix. For detailed operational parameters and safety guidelines, please refer to the standardized synthesis steps outlined below.
- Under inert gas protection, dissolve a thioether compound (e.g., dimethyl sulfide) in a polar aprotic solvent like DMF and add halogen (Br2 or I2) at controlled low temperatures.
- Add gamma-cyclodextrin (Compound II) to the mixture and maintain the reaction temperature between 20°C and 55°C for bromination or higher for iodination until completion.
- Filter the reaction mixture, concentrate the filtrate, adjust pH to alkaline conditions (pH 9-12) to induce crystallization, and recrystallize the crude product to obtain high-purity Compound I.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this thioether-based synthesis route offers tangible strategic benefits beyond mere technical elegance. The primary advantage lies in the drastic simplification of the downstream processing. By eliminating the generation of solid phosphine oxide sludge, the manufacturing facility can significantly reduce the time and solvent volume required for purification. This reduction in processing time directly translates to increased reactor turnover rates, allowing for higher production throughput without the need for additional capital investment in hardware. Furthermore, the reliance on commodity chemicals like dimethyl sulfide and bromine, rather than specialized and expensive phosphine ligands, insulates the supply chain from volatility in the pricing of niche reagents.
- Cost Reduction in Manufacturing: The elimination of triphenylphosphine and its derivatives removes a major cost driver from the bill of materials. Triphenylphosphine is not only expensive but also contributes significantly to waste disposal costs due to the difficulty in treating phosphine-containing effluents. By switching to a thioether system, the process achieves a leaner material profile where the reagents are cheaper and the waste stream is easier to manage. The higher yields reported in the patent examples mean that less raw material is wasted per kilogram of finished intermediate, further driving down the unit cost of goods sold. This efficiency creates a buffer against raw material price fluctuations and improves the overall margin profile for the manufactured intermediate.
- Enhanced Supply Chain Reliability: The robustness of the new method enhances supply continuity by reducing the risk of batch failures due to purification issues. In the old process, a single batch of difficult-to-remove phosphine oxide could delay a shipment by weeks while additional recrystallizations were attempted. The new method's ability to consistently produce high-purity material in fewer steps ensures a more predictable lead time for customers. Additionally, the reagents used are widely available from multiple global suppliers, reducing the risk of single-source bottlenecks. This diversification of the supply base is critical for maintaining the uninterrupted flow of materials required for the production of life-saving anesthesia reversal agents.
- Scalability and Environmental Compliance: Scaling chemical processes often amplifies heat transfer and mixing challenges, but the mild temperature range of this reaction (20-55°C) makes it highly amenable to large-scale reactor operations. The absence of highly corrosive reagents like PBr3 reduces the maintenance burden on reactor vessels and piping, extending equipment lifespan. From an environmental perspective, the improved atom economy and the use of less hazardous reagents align with increasingly stringent global regulations on pharmaceutical manufacturing emissions. This compliance reduces the regulatory burden on the manufacturer and minimizes the risk of production shutdowns due to environmental non-compliance, ensuring a stable long-term supply for partners.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis technology. These answers are derived directly from the experimental data and claims within the patent documentation, providing a factual basis for decision-making. Understanding these nuances is essential for evaluating the feasibility of integrating this route into existing manufacturing portfolios.
Q: What are the primary advantages of the thioether-halogen method over traditional phosphine-based halogenation?
A: The thioether-halogen method eliminates the need for high molecular weight phosphine reagents like triphenylphosphine (TPP), which generate difficult-to-remove phosphine oxide by-products. This results in significantly simplified purification processes, higher atom utilization, and improved overall yields suitable for industrial production.
Q: What represents the critical quality attribute for the sugammadex intermediate produced via this route?
A: The critical quality attribute is the high purity of the octa-6-perdeoxy-6-perhalo-gamma-cyclodextrin (Compound I), which serves as the activated precursor. The novel method achieves purity levels exceeding 99% through efficient crystallization, minimizing impurities that could affect the final API's safety profile.
Q: Is this synthesis method scalable for commercial manufacturing of pharmaceutical intermediates?
A: Yes, the process utilizes mild reaction conditions (20-55°C) and commercially available reagents like dimethyl sulfide and bromine. The absence of complex post-reaction removal steps for heavy by-products makes the process highly robust and easily scalable from pilot to multi-ton commercial production.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Sodium Sugammadex Intermediate Supplier
At NINGBO INNO PHARMCHEM, we recognize that the transition from laboratory innovation to commercial reality requires a partner with deep technical expertise and unwavering commitment to quality. Our team has extensively analyzed the pathways described in patent CN114181331B and is fully prepared to execute this advanced thioether-halogenation protocol at scale. We possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your supply needs are met with precision and consistency. Our facilities are equipped with rigorous QC labs and stringent purity specifications that guarantee every batch of sodium sugammadex intermediate meets the highest international standards for pharmaceutical use.
We invite you to collaborate with us to leverage this cutting-edge synthesis technology for your supply chain. By partnering with our technical procurement team, you can access a Customized Cost-Saving Analysis tailored to your specific volume requirements. We encourage you to reach out today to request specific COA data and route feasibility assessments that demonstrate how our optimized manufacturing capabilities can enhance your project's efficiency and profitability. Let us be your trusted partner in delivering high-quality pharmaceutical intermediates that power the next generation of therapeutic solutions.
