Advanced One-Pot Synthesis of Alpha-Keto-Leucine-Calcium for Industrial Scale-Up
The pharmaceutical industry continuously seeks robust synthetic routes for critical metabolic intermediates, and the preparation of alpha-keto-leucine-calcium stands as a prime example of process innovation driven by patent CN101607888B. This specific intellectual property outlines a transformative methodology that shifts away from cumbersome multi-step isolations toward a streamlined, telescoped manufacturing protocol. For R&D directors and process chemists, the core value lies in the elimination of the intermediate isolation step, which traditionally acts as a bottleneck in throughput and yield efficiency. By integrating the condensation of hydantoin with isobutyraldehyde directly into the hydrolysis phase, the technology not only simplifies the operational workflow but also enhances the overall atom economy of the synthesis. This report analyzes the technical merits of this approach, offering supply chain leaders and procurement managers a clear view of how adopting such optimized routes can lead to substantial cost reductions and improved supply continuity for high-purity pharmaceutical intermediates.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of alpha-keto-leucine-calcium has relied on disjointed methodologies that suffer from significant operational inefficiencies and economic drawbacks. Traditional Route 1, for instance, necessitates the complete isolation and purification of the intermediate, isobutylidene hydantoin, before proceeding to hydrolysis, a practice that inherently incurs material losses at every transfer and filtration stage. Furthermore, alternative Routes 2 and 3 often employ harsh oxidation conditions or expensive reagents like diethyl cyanophosphonate, which introduce severe safety hazards and complicate waste stream management. These legacy processes typically require specialized equipment capable of handling aggressive chemicals and often result in lower overall yields due to the cumulative effect of purification losses. From a supply chain perspective, the reliance on difficult-to-source raw materials and the generation of complex organic waste streams create volatility in production scheduling and inflate the environmental compliance costs associated with manufacturing.
The Novel Approach
In stark contrast, the novel approach detailed in the patent data introduces a cohesive one-pot strategy that fundamentally restructures the production logic for this key uremia treatment precursor. By maintaining the reaction mixture in a single vessel through the condensation and hydrolysis phases, the process eliminates the need for intermediate separation, thereby maximizing equipment utilization and reducing the total cycle time per batch. The substitution of organic solvents with water for the primary reaction steps represents a significant leap toward green chemistry, drastically lowering the thermal load required for solvent recovery and minimizing the generation of hazardous volatile organic compounds. This methodological shift ensures that the reaction conditions remain mild and controllable, reducing the risk of thermal runaways and allowing for the use of standard stainless steel reactors rather than exotic alloy vessels. Consequently, this approach offers a scalable, economically superior pathway that aligns perfectly with modern demands for sustainable and cost-effective pharmaceutical intermediate manufacturing.
Mechanistic Insights into Base-Catalyzed Condensation and Hydrolysis
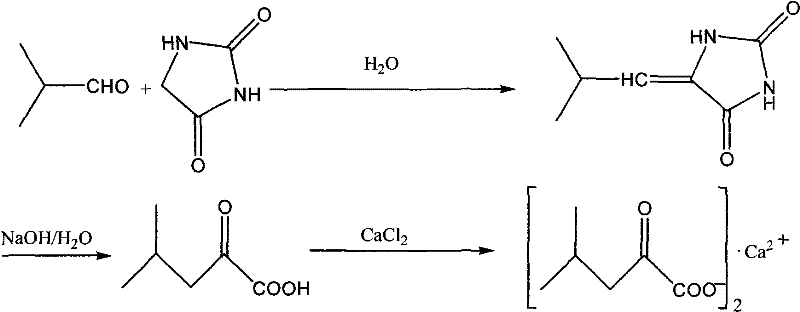
The chemical elegance of this synthesis lies in its sequential base-catalyzed transformations, beginning with the condensation of hydantoin and isobutyraldehyde to form the exocyclic double bond of isobutylidene hydantoin. In the presence of a mild base catalyst such as triethylamine or sodium acetate, the active methylene group of the hydantoin ring undergoes nucleophilic attack on the carbonyl carbon of the aldehyde, followed by dehydration to establish the stable olefinic linkage. Crucially, instead of isolating this solid intermediate, the process proceeds directly to alkaline hydrolysis by introducing a strong alkali solution, such as sodium hydroxide, into the same reaction matrix. This high-pH environment facilitates the ring-opening of the hydantoin moiety, cleaving the urea bond to release the alpha-keto acid structure while retaining the stereochemical integrity required for biological activity. The seamless transition from condensation to hydrolysis without workup prevents the degradation of the sensitive keto-acid functionality that might occur during prolonged exposure to air or additional purification steps.
Following the hydrolysis, the reaction mixture is acidified to precipitate the free alpha-keto-leucine, which is then extracted and concentrated to obtain a crude liquid ready for salt formation. The final step involves reacting this crude acid with calcium chloride in an alcoholic medium, where the carboxylate anion coordinates with the calcium cation to form the insoluble calcium salt. This precipitation mechanism serves as a powerful purification tool in itself, as the crystallization of the calcium salt effectively excludes many organic impurities that remain soluble in the alcohol-water matrix. The use of calcium chloride not only stabilizes the reactive keto-acid group but also converts the molecule into its pharmacologically active form used in compound alpha-ketoacid preparations for renal insufficiency. Understanding this mechanistic flow is vital for quality control teams to monitor critical parameters such as pH transitions and temperature profiles to ensure consistent product quality.
How to Synthesize Alpha-Keto-Leucine-Calcium Efficiently
Implementing this synthesis requires precise control over stoichiometry and thermal conditions to maximize the benefits of the telescoped design. The process begins by mixing hydantoin, a base catalyst, and water, heating the solution to initiate the condensation with isobutyraldehyde under reflux. Once the condensation is complete, strong alkali is added directly to the hot mixture to drive the hydrolysis, followed by cooling and extraction to isolate the crude acid.
- Condense hydantoin with isobutyraldehyde in aqueous alkali to form isobutylidene hydantoin.
- Perform direct alkaline hydrolysis on the reaction mixture without isolating the intermediate.
- React the crude alpha-keto-leucine with calcium chloride in alcohol to precipitate the final calcium salt.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this patented methodology translates into tangible strategic advantages that extend beyond simple unit cost calculations. The primary driver of value is the drastic simplification of the manufacturing workflow, which reduces the dependency on complex, multi-vessel processing lines and minimizes the labor hours required for intermediate handling and cleaning. By utilizing water as the primary solvent for the initial stages, the process eliminates the need for purchasing, storing, and recovering large volumes of expensive organic solvents, thereby insulating the production cost from fluctuations in petrochemical markets. Furthermore, the accessibility of raw materials like hydantoin and isobutyraldehyde ensures a stable supply base, reducing the risk of production stoppages due to raw material shortages that often plague more exotic synthetic routes. These factors combine to create a resilient supply chain capable of meeting high-volume demands with greater predictability and lower operational overhead.
- Cost Reduction in Manufacturing: The elimination of the intermediate isolation step fundamentally alters the cost structure by removing entire unit operations such as filtration, drying, and re-dissolution of the isobutylidene hydantoin. This reduction in processing steps directly correlates to lower energy consumption, as there is less need for heating and cooling cycles associated with multiple workups, and significantly reduces the wear and tear on equipment. Additionally, the avoidance of expensive reagents found in alternative routes, such as phosphonates or specialized oxidants, ensures that the raw material bill remains competitive and stable. The overall result is a leaner manufacturing process where the cost per kilogram of the final API intermediate is optimized through engineering efficiency rather than mere bargaining power.
- Enhanced Supply Chain Reliability: Reliability in the supply of critical renal care intermediates is paramount, and this process enhances security by relying on commodity chemicals that are widely produced and globally available. The robustness of the aqueous-based reaction system means that production is less susceptible to disruptions caused by the scarcity of specialized anhydrous solvents or sensitive catalysts. Moreover, the mild reaction conditions reduce the likelihood of batch failures due to thermal excursions or safety incidents, ensuring a consistent output of material that can be reliably scheduled into downstream formulation processes. This stability allows supply chain planners to maintain lower safety stock levels while still guaranteeing continuity of supply for finished drug products.
- Scalability and Environmental Compliance: Scaling chemical processes often introduces unforeseen challenges, but the simplicity of this one-pot aqueous method makes it inherently scalable from pilot plants to multi-ton commercial reactors without significant re-engineering. The environmental profile is markedly improved due to the reduction in organic waste streams and the absence of heavy metal catalysts or toxic oxidants, simplifying wastewater treatment and regulatory compliance. This alignment with green chemistry principles not only reduces disposal costs but also future-proofs the manufacturing site against tightening environmental regulations, making it a sustainable choice for long-term production partnerships.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production of alpha-keto-leucine-calcium, derived directly from the operational data and comparative analysis of the patent literature. These insights are intended to clarify the feasibility of the technology for potential partners and highlight the specific advantages over legacy manufacturing methods.
Q: How does the one-pot method improve yield compared to traditional routes?
A: By eliminating the isolation and purification steps of the intermediate isobutylidene hydantoin, mechanical losses are significantly reduced, leading to a reported yield increase of approximately 10% over conventional multi-step methods.
Q: What are the environmental benefits of using water as the primary solvent?
A: Replacing organic solvents with water for the condensation and hydrolysis steps drastically reduces VOC emissions and lowers the cost of solvent recovery and waste disposal, aligning with green chemistry principles.
Q: Is this process suitable for large-scale commercial production?
A: Yes, the process utilizes mild reaction conditions and readily available raw materials like hydantoin and isobutyraldehyde, requiring no specialized high-pressure equipment, which facilitates easy scale-up to metric ton quantities.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Alpha-Keto-Leucine-Calcium Supplier
At NINGBO INNO PHARMCHEM, we recognize that the transition from laboratory innovation to commercial reality requires a partner with deep technical expertise and robust infrastructure. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the theoretical benefits of advanced synthetic routes like the one described in CN101607888B are fully realized in practice. We operate stringent purity specifications and maintain rigorous QC labs equipped to analyze complex impurity profiles, guaranteeing that every batch of alpha-keto-leucine-calcium meets the exacting standards required for pharmaceutical applications. Our commitment to quality is matched by our dedication to process safety and environmental stewardship, making us an ideal partner for global health initiatives.
We invite procurement leaders and R&D directors to engage with us for a Customized Cost-Saving Analysis tailored to your specific volume requirements and quality targets. By collaborating with our technical procurement team, you can access specific COA data and route feasibility assessments that demonstrate exactly how our optimized manufacturing capabilities can enhance your supply chain efficiency. Contact us today to discuss how we can support your long-term goals with reliable, high-quality pharmaceutical intermediates.
