Advanced Manufacturing of High-Purity 2-Indole Carboxylic Acid for Pharmaceutical Applications
The pharmaceutical industry continuously seeks robust and scalable synthetic routes for critical heterocyclic intermediates, and the methodology disclosed in patent CN102432524A represents a significant advancement in the production of 2-indole carboxylic acid. This compound, identified by CAS number 1477-50-5, serves as a pivotal building block for various bioactive molecules, yet its industrial synthesis has historically faced challenges regarding complexity and cost. The patented approach introduces a streamlined two-step sequence that begins with the condensation of o-nitrotoluene and diethyl oxalate, followed by a unique reductive cyclization utilizing ferrous sulfate. By leveraging common chemical feedstocks and avoiding exotic catalysts, this process addresses the critical need for reliable pharma intermediates supplier capabilities while maintaining stringent quality standards. The technical elegance of this route lies not only in its operational simplicity but also in its ability to consistently deliver product purity exceeding 99% via HPLC analysis. For R&D directors and procurement specialists alike, understanding the mechanistic nuances and commercial implications of this patent is essential for optimizing supply chains and reducing manufacturing overheads in the competitive landscape of fine chemical production.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of indole derivatives has often relied on methodologies that involve harsh reaction conditions, expensive transition metal catalysts, or multi-step sequences that suffer from cumulative yield losses. Traditional routes to 2-substituted indoles might require precious metals like palladium or rhodium for cyclization, which introduces significant cost burdens related to catalyst recovery and residual metal limits in the final API. Furthermore, many existing protocols necessitate rigorous anhydrous conditions or cryogenic temperatures, demanding specialized equipment and increasing energy consumption substantially. The removal of impurities in these conventional pathways can be notoriously difficult, often requiring complex chromatographic separations that are impractical for large-scale commercial manufacturing. Additionally, the use of hazardous reagents in older methods poses environmental and safety risks that complicate regulatory compliance and waste disposal logistics. These factors collectively contribute to higher production costs and longer lead times, creating bottlenecks for companies seeking cost reduction in pharmaceutical intermediates manufacturing.
The Novel Approach
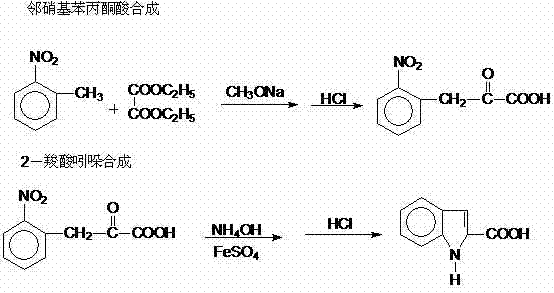
In stark contrast, the method described in the patent utilizes a highly efficient strategy that circumvents the need for precious metals by employing ferrous sulfate as the reducing agent for the cyclization step. This innovative approach starts with the formation of o-nitrophenyl pyruvic acid through a base-catalyzed condensation, a reaction that proceeds smoothly under reflux conditions with sodium methoxide. The subsequent transformation involves reacting this intermediate with ammonia water and ferrous sulfate at moderate temperatures between 70°C and 90°C, effectively reducing the nitro group and inducing ring closure simultaneously. This tandem reduction-cyclization eliminates the need for separate reduction and cyclization stages, thereby shortening the overall process time and minimizing solvent usage. The workup procedure is equally straightforward, involving simple filtration to remove iron sludge followed by acidification to precipitate the product, which avoids the need for complex extraction protocols. This operational simplicity translates directly into enhanced process robustness, making it an ideal candidate for the commercial scale-up of complex pharmaceutical intermediates.
Mechanistic Insights into Ferrous Sulfate Mediated Reductive Cyclization
The core chemical transformation in this synthesis is a fascinating example of reductive cyclization driven by the redox potential of ferrous ions in an alkaline ammoniacal medium. Initially, o-nitrotoluene undergoes a Claisen-type condensation with diethyl oxalate in the presence of sodium methoxide to form an enolate intermediate, which upon hydrolysis and acidification yields o-nitrophenyl pyruvic acid. This alpha-keto acid derivative possesses the necessary electronic configuration to participate in the subsequent ring-forming reaction. When treated with ammonia and ferrous sulfate, the nitro group is reduced, likely proceeding through nitroso and hydroxylamine intermediates, while the adjacent carbonyl functionality facilitates nucleophilic attack by the nitrogen species. The ferrous ions act as electron donors, getting oxidized to ferric states which precipitate as iron hydroxides or oxides, easily removable by filtration. This mechanism ensures that the cyclization occurs regioselectively to form the indole nucleus without affecting other sensitive functional groups that might be present in more complex analogues. Understanding this redox cycle is crucial for process chemists aiming to optimize reaction kinetics and minimize the formation of side products such as azo-compounds or over-reduced species.
Impurity control in this process is inherently managed by the physical properties of the byproducts and the specificity of the reaction conditions. The primary inorganic byproduct, iron sludge, is insoluble and is quantitatively removed via filtration prior to the final isolation step, preventing metal contamination in the organic phase. Organic impurities, such as unreacted o-nitrotoluene, are effectively removed during the steam distillation step in the first stage, ensuring that the intermediate o-nitrophenyl pyruvic acid enters the cyclization reactor with high purity. The final recrystallization from ethanol further purifies the crude 2-indole carboxylic acid, leveraging the differential solubility of the product versus potential isomeric impurities or oligomers. This multi-layered purification strategy, embedded within the reaction design itself, guarantees that the final material meets the rigorous specifications required for high-purity pharmaceutical intermediates. For quality assurance teams, this inherent cleanliness of the process reduces the analytical burden and ensures batch-to-batch consistency, which is vital for regulatory filings.
How to Synthesize 2-Indole Carboxylic Acid Efficiently
The execution of this synthesis requires careful attention to stoichiometry and temperature control to maximize yield and purity. The process begins by dissolving o-nitrotoluene, diethyl oxalate, and sodium methoxide in a reactor, where they are heated to reflux for 5 to 10 hours to ensure complete condensation. Following this, water is added, and the mixture is refluxed again for 4 to 6 hours to hydrolyze the ester groups, after which unreacted o-nitrotoluene is distilled off to drive the equilibrium forward. Once the intermediate o-nitrophenyl pyruvic acid is isolated via acidification and filtration, it is subjected to the cyclization conditions with ammonia water and ferrous sulfate at 70-90°C for 4 to 8 hours. The detailed standardized operating procedures, including specific molar ratios and workup parameters, are outlined below to guide process implementation.
- Reflux o-nitrotoluene, diethyl oxalate, and sodium methoxide for 5-10 hours, then hydrolyze and acidify to isolate o-nitrophenyl pyruvic acid.
- React the isolated o-nitrophenyl pyruvic acid with ammonia water and ferrous sulfate at 70-90°C for 4-8 hours to induce cyclization.
- Filter off iron sludge, acidify the filtrate to pH 2-3 to precipitate the crude product, and recrystallize with ethanol for final purification.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, this patented methodology offers substantial benefits that directly impact the bottom line and supply chain resilience for chemical manufacturers. The reliance on commodity chemicals such as o-nitrotoluene and ferrous sulfate means that raw material sourcing is not constrained by geopolitical factors or limited supplier bases, ensuring a stable and continuous supply flow. The elimination of expensive noble metal catalysts removes a significant cost center and simplifies the procurement process, as there is no need to manage the logistics of catalyst recovery or purchase specialized ligands. Furthermore, the operational simplicity of the process, characterized by standard reflux and filtration unit operations, allows for production in multipurpose reactors without the need for specialized high-pressure or cryogenic equipment. This flexibility enhances asset utilization rates and reduces capital expenditure requirements for new production lines. Consequently, manufacturers can achieve significant cost savings in fine chemical intermediates manufacturing while maintaining high margins.
- Cost Reduction in Manufacturing: The economic advantage of this route is primarily driven by the substitution of costly reagents with inexpensive, bulk-available alternatives like ferrous sulfate and ammonia. By avoiding the use of precious metals, the process eliminates the associated costs of catalyst purchasing, regeneration, and the rigorous testing required to certify low residual metal levels in the final product. Additionally, the high atom economy of the condensation step and the efficient conversion in the cyclization step minimize raw material waste, further driving down the cost of goods sold. The simplified workup procedure reduces solvent consumption and energy usage associated with complex separations, contributing to a leaner and more cost-effective production model that enhances overall profitability.
- Enhanced Supply Chain Reliability: The use of widely available starting materials ensures that production schedules are not vulnerable to disruptions in the supply of niche or specialized reagents. O-nitrotoluene and diethyl oxalate are produced on a massive global scale, providing multiple sourcing options that mitigate the risk of single-supplier dependency. The robustness of the reaction conditions also means that the process is less sensitive to minor variations in raw material quality, reducing the likelihood of batch failures that could disrupt delivery timelines. This reliability is critical for maintaining just-in-time inventory levels and meeting the demanding delivery windows of downstream pharmaceutical clients, thereby strengthening the manufacturer's reputation as a dependable partner.
- Scalability and Environmental Compliance: The process is inherently designed for scalability, utilizing unit operations such as distillation, filtration, and crystallization that are easily transferred from pilot plant to full commercial scale. The generation of iron sludge as the primary waste stream is manageable through standard wastewater treatment protocols, avoiding the generation of toxic heavy metal waste that requires specialized disposal methods. This environmental profile aligns with increasingly stringent global regulations on chemical manufacturing, reducing the compliance burden and potential liability associated with hazardous waste handling. The ability to scale up without significant process redesign ensures that supply can be rapidly ramped up to meet market demand, supporting the commercial growth of downstream drug products.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production of 2-indole carboxylic acid using this specific patented methodology. These answers are derived directly from the experimental data and claims presented in the patent documentation to provide accurate guidance for technical teams. Understanding these details helps in assessing the feasibility of adopting this route for internal production or evaluating external suppliers.
Q: What is the purity level achievable with this synthesis method?
A: According to patent CN102432524A, the refined 2-indole carboxylic acid achieves a content of greater than 99% as determined by HPLC analysis, ensuring suitability for high-grade pharmaceutical applications.
Q: What are the key raw materials required for this process?
A: The process utilizes readily available and cost-effective starting materials including o-nitrotoluene, diethyl oxalate, sodium methoxide, ammonia water, and ferrous sulfate, which simplifies supply chain management.
Q: How is the waste managed in this specific reductive cyclization?
A: The process generates iron sludge as a byproduct during the reduction step, which is removed via simple filtration before the final acidification and crystallization steps, facilitating easier waste treatment compared to heavy metal catalysis.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 2-Indole Carboxylic Acid Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-quality intermediates play in the success of pharmaceutical development pipelines. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that we can meet your volume requirements with consistency and precision. We adhere to stringent purity specifications and operate rigorous QC labs to guarantee that every batch of 2-indole carboxylic acid meets or exceeds the >99% purity benchmark established by the patent. Our commitment to technical excellence allows us to navigate the complexities of chemical synthesis, delivering materials that facilitate smooth downstream processing for our clients.
We invite you to collaborate with us to leverage this advanced synthesis technology for your projects. Please contact our technical procurement team to request a Customized Cost-Saving Analysis tailored to your specific volume needs. We are ready to provide specific COA data and route feasibility assessments to demonstrate how our manufacturing capabilities can support your supply chain goals and drive value for your organization.
