Revolutionizing Asymmetric Synthesis: Lipozyme TLIM Catalysis for Commercial Scale-up
Revolutionizing Asymmetric Synthesis: Lipozyme TLIM Catalysis for Commercial Scale-up
The landscape of fine chemical synthesis is undergoing a paradigm shift towards greener, more sustainable methodologies, driven by the urgent need to reduce environmental impact while maintaining high efficiency. A pivotal development in this arena is detailed in patent CN101892273B, which discloses the novel application of immobilized lipase Lipozyme TLIM as a highly effective catalyst for asymmetric Michael addition reactions. This technology represents a significant departure from conventional chemical catalysis, offering a robust platform for the production of complex chiral intermediates essential for the pharmaceutical and agrochemical industries. By leveraging the unique catalytic properties of this thermophilic fungal lipase, manufacturers can achieve superior stereocontrol and yield under remarkably mild conditions, addressing critical pain points related to waste generation and energy consumption in modern process chemistry.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditionally, the Michael addition reaction, a fundamental carbon-carbon bond-forming transformation, has been predominantly catalyzed by strong bases, amines, carboxylic acids, or Lewis acids. While these homogeneous catalytic systems are well-established, they suffer from inherent drawbacks that pose significant challenges for large-scale industrial application. The use of stoichiometric amounts of bases or toxic Lewis acids often leads to severe environmental pollution and necessitates complex, costly waste treatment protocols. Furthermore, these harsh chemical environments frequently result in poor stereoselectivity, requiring additional resolution steps that drastically reduce overall process efficiency. The sensitivity of many functional groups to strong acidic or basic conditions also limits the substrate scope, forcing chemists to employ extensive protecting group strategies that add unnecessary steps and cost to the synthetic route.
The Novel Approach
In stark contrast, the novel approach utilizing immobilized lipase Lipozyme TLIM offers a transformative solution by operating within a benign, water-containing organic solvent system. As illustrated in the reaction scheme below, this biocatalytic method facilitates the nucleophilic addition of active methylene compounds to alpha,beta-unsaturated carbonyl compounds or nitroolefins with exceptional precision. The process eliminates the need for hazardous reagents, functioning effectively at moderate temperatures between 25°C and 85°C. This not only enhances safety but also significantly reduces energy demands. The immobilized nature of the enzyme further allows for straightforward separation from the reaction mixture via simple filtration, enabling catalyst recovery and reuse, which is a critical factor for economic viability in continuous manufacturing processes.
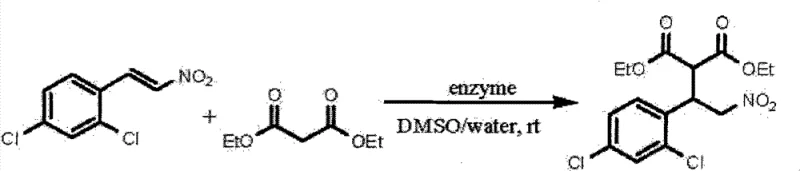
Mechanistic Insights into Lipozyme TLIM-Catalyzed Asymmetric Michael Addition
The exceptional performance of Lipozyme TLIM in this context is not merely due to its proteinaceous nature but is rooted in its specific three-dimensional structure and active site architecture. Detailed mechanistic studies within the patent reveal that the catalytic activity is intimately linked to specific amino acid residues, particularly serine and sulfhydryl groups, as well as tryptophan and tyrosine residues. Experiments utilizing specific inhibitors such as phenylmethylsulfonyl fluoride (PMSF) and N-bromosuccinimide (NBS) demonstrated that blocking these active sites completely abolishes catalytic activity, confirming that the reaction proceeds through a specific enzymatic mechanism rather than non-specific protein interactions. This specificity is crucial for achieving the high enantioselectivity and diastereoselectivity observed, distinguishing it from control experiments using non-catalytic proteins like ovalbumin which showed negligible activity.
Furthermore, the reaction environment plays a pivotal role in modulating the enzyme's conformation and activity. The patent data highlights the importance of a biphasic or water-miscible organic solvent system, specifically dimethyl sulfoxide (DMSO) mixed with water, in optimizing the reaction kinetics. The presence of water is essential for maintaining the hydration shell of the enzyme, preserving its active conformation, while the organic solvent ensures the solubility of hydrophobic organic substrates. This delicate balance allows for a broad substrate scope, accommodating various aromatic and heterocyclic substituted beta-nitroolefins as well as cyclic ketones. The ability of the enzyme to discriminate between enantiomers in the transition state leads to products with high optical purity, a feature that is often difficult to replicate with small molecule organocatalysts without expensive chiral ligands.
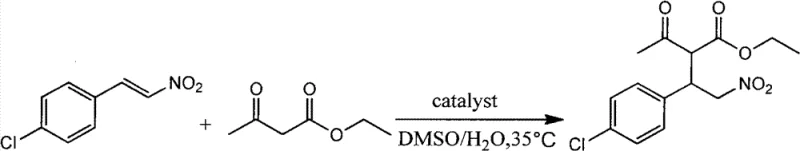
How to Synthesize Michael Addition Adducts Efficiently
Implementing this biocatalytic protocol requires careful attention to reaction parameters to maximize yield and selectivity. The general procedure involves dissolving the Michael acceptor and donor in a mixture of DMSO and deionized water, followed by the addition of the immobilized enzyme. The reaction is typically stirred at temperatures around 35°C to 65°C, depending on the specific substrate reactivity. Monitoring via thin-layer chromatography (TLC) is recommended to determine the endpoint, after which the enzyme is recovered by filtration. The subsequent workup involves standard extraction and purification techniques, such as flash column chromatography, to isolate the pure product. For a comprehensive, step-by-step guide tailored to your specific substrate, please refer to the standardized synthesis protocol provided below.
- Prepare the reaction mixture by combining the Michael acceptor (e.g., beta-nitrostyrene derivatives), Michael donor (e.g., 1,3-dicarbonyl compounds), deionized water, and an organic solvent such as DMSO in a round-bottomed flask.
- Add the immobilized lipase Lipozyme TLIM catalyst to the mixture and stir at a controlled temperature ranging from 25°C to 85°C, preferably around 35°C to 65°C, until the reaction is complete as monitored by TLC.
- Filter the reaction mixture to recover the enzyme catalyst, wash the filter cake with dichloromethane, combine the filtrates, extract with dichloromethane, dry over anhydrous sodium sulfate, and purify the crude product via flash column chromatography.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of Lipozyme TLIM catalysis presents a compelling value proposition centered on cost efficiency and operational reliability. The shift from traditional chemical catalysis to this enzymatic process fundamentally alters the cost structure of manufacturing complex intermediates. By eliminating the need for expensive chiral ligands, toxic heavy metals, and harsh reagents, the raw material costs are significantly optimized. Moreover, the simplified downstream processing—where the catalyst is removed by simple filtration rather than complex quenching and extraction procedures—reduces solvent consumption and labor hours, leading to substantial overall cost reductions in pharmaceutical intermediate manufacturing.
- Cost Reduction in Manufacturing: The economic benefits extend beyond mere reagent costs. The high selectivity of the enzyme minimizes the formation of by-products and impurities, which directly translates to higher yields and reduced waste disposal costs. In traditional processes, the removal of trace metal contaminants often requires specialized scavenging resins or additional crystallization steps, adding significant expense. With Lipozyme TLIM, the product stream is inherently cleaner, reducing the burden on quality control laboratories and shortening the production cycle time. This efficiency gain allows for a more competitive pricing structure for the final API intermediates.
- Enhanced Supply Chain Reliability: Supply chain resilience is bolstered by the robustness and reusability of the immobilized enzyme. The patent data confirms that Lipozyme TLIM can be recycled for multiple batches without a catastrophic loss of activity, ensuring a consistent supply of catalyst and reducing dependency on single-use reagents. This stability mitigates the risk of production delays caused by reagent shortages or quality fluctuations. Additionally, the mild reaction conditions reduce the stress on reactor equipment, lowering maintenance requirements and extending the lifespan of capital assets, thereby ensuring uninterrupted production schedules.
- Scalability and Environmental Compliance: From a regulatory and sustainability perspective, this technology aligns perfectly with modern green chemistry principles. The use of water as a co-solvent and the absence of toxic heavy metals simplify the environmental compliance landscape, reducing the regulatory burden associated with waste discharge permits. The process is inherently scalable, as demonstrated by the successful optimization of solvent ratios and substrate concentrations in the patent examples. This scalability ensures that the transition from laboratory benchtop to multi-ton commercial production is seamless, supporting the growing demand for high-purity pharmaceutical intermediates without compromising environmental standards.
Frequently Asked Questions (FAQ)
To assist technical teams in evaluating the feasibility of this technology for their specific applications, we have compiled answers to common inquiries based on the patent specifications. These questions address critical aspects such as catalyst longevity, solvent compatibility, and substrate versatility. Understanding these technical nuances is essential for integrating this biocatalytic route into existing manufacturing workflows and maximizing its potential benefits.
Q: Can Lipozyme TLIM be reused in Michael addition reactions?
A: Yes, the patent data indicates that immobilized lipase Lipozyme TLIM can be recycled. Experimental results show the enzyme maintains catalytic activity and selectivity for up to three cycles, although activity gradually decreases by the fourth cycle.
Q: What is the optimal solvent system for this biocatalytic process?
A: The process utilizes a water-containing organic solvent system. Preferred organic solvents include DMSO, THF, DMF, or ethanol, with a preferred water-to-organic solvent volume ratio ranging from 0:100 to 30:100, optimally around 10:100.
Q: How does Lipozyme TLIM compare to traditional chemical catalysts?
A: Unlike traditional base or Lewis acid catalysts which often cause environmental issues and require harsh conditions, Lipozyme TLIM operates under mild temperatures (25-85°C) and offers high stereoselectivity. It avoids the use of toxic heavy metals and simplifies downstream processing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Michael Addition Adducts Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of biocatalysis in modern drug discovery and development. Our team of expert chemists possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that innovative technologies like Lipozyme TLIM catalysis are seamlessly translated into robust industrial processes. We are committed to delivering high-purity pharmaceutical intermediates that meet stringent purity specifications, supported by our rigorous QC labs equipped with state-of-the-art analytical instrumentation. Our dedication to quality and consistency makes us a trusted partner for global pharmaceutical companies seeking reliable sources for complex chiral building blocks.
We invite you to explore how this advanced enzymatic technology can optimize your supply chain and reduce your overall manufacturing costs. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your specific project needs. We are ready to provide specific COA data and comprehensive route feasibility assessments to demonstrate how our capabilities can support your next breakthrough therapy. Let us collaborate to bring your most challenging synthetic targets to life with efficiency and precision.
