Advanced Synthetic Route for Empagliflozin Intermediate Enhancing Commercial Scalability and Purity
The global demand for SGLT2 inhibitors, particularly empagliflozin, has surged due to their proven efficacy in managing type 2 diabetes and reducing cardiovascular risks. As the market expands, the need for robust, scalable, and cost-effective synthetic routes for key intermediates becomes paramount for pharmaceutical manufacturers. Patent CN113480497B discloses a groundbreaking synthetic method for producing the critical empagliflozin intermediate, (3S)-3-[4-[(5-bromo-2-chlorophenyl)methyl]phenoxy]tetrahydrofuran. This innovation addresses significant bottlenecks in existing manufacturing processes by utilizing a novel indium-catalyzed reduction strategy. By shifting away from hazardous reagents and expensive chiral starting materials, this technology offers a compelling value proposition for reliable pharmaceutical intermediate suppliers aiming to optimize their production lines. The following analysis details the technical superiority and commercial viability of this approach for high-purity API manufacturing.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
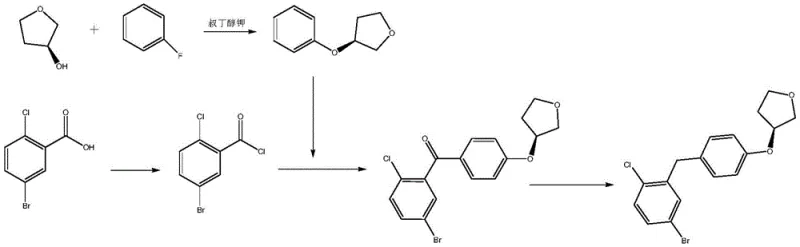
Traditional synthetic pathways for this specific tetrahydrofuran derivative have historically relied on chemically aggressive and economically inefficient strategies. One common prior art route involves the Friedel-Crafts acylation of (S)-3-phenoxytetrahydrofuran, which itself requires the use of expensive chiral starting materials like (S)-3-hydroxytetrahydrofuran. Furthermore, alternative methods often necessitate the use of boron tribromide for demethylation steps, a reagent known for its extreme moisture sensitivity, corrosive nature, and difficult handling requirements on an industrial scale. Other routes utilize potassium tert-butoxide for etherification, introducing severe safety hazards due to its pyrophoric properties and violent reaction with water. These conventional methods not only inflate the cost of goods sold (COGS) through expensive raw materials but also complicate waste management and operator safety protocols, making them less ideal for large-scale commercial production.
The Novel Approach
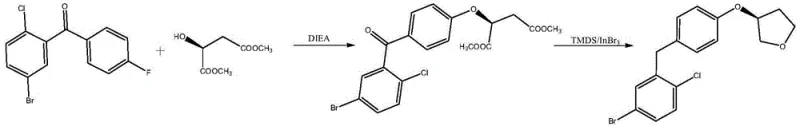
In stark contrast, the methodology outlined in CN113480497B introduces a streamlined two-step process that circumvents these historical challenges. The new route initiates with an etherification reaction between 3-(4-fluorophenylketone)-1-bromo-4-chlorobenzene and L-dimethyl malate, utilizing DIEA as a base under mild thermal conditions. This is followed by a sophisticated one-pot reduction and cyclization step catalyzed by indium bromide using TMDS (1,1,3,3-tetramethyldisiloxane) as the reducing agent. This approach effectively replaces the need for cryogenic conditions and hazardous strong bases, allowing the reaction to proceed at temperatures between 10°C and 30°C. The result is a significant simplification of the operational workflow, eliminating the need for specialized equipment to handle pyrophoric substances while simultaneously improving the overall atom economy of the synthesis.
Mechanistic Insights into Indium-Catalyzed Reduction and Cyclization
The core innovation of this patent lies in the dual functionality of the indium bromide and TMDS system, which orchestrates a complex cascade of reductions and cyclizations with high stereocontrol. Mechanistically, the indium catalyst activates the carbonyl groups of the succinate intermediate, facilitating the hydride transfer from TMDS. This unique catalytic environment allows for the simultaneous reduction of the ketone to a methylene group and the ester functionalities to hydroxyl groups. Crucially, the proximity of the newly formed hydroxyl groups to the ether linkage promotes an intramolecular dehydration, driving the formation of the tetrahydrofuran ring. This tandem process ensures that the chiral information from the L-dimethyl malate is preserved and transferred efficiently to the final product, resulting in high optical purity without the need for additional resolution steps.
From an impurity control perspective, this mechanism offers distinct advantages over traditional Lewis acid-catalyzed reactions. The mild acidity of the indium species minimizes side reactions such as polymerization or over-reduction, which are common pitfalls when using stronger reducing agents like lithium aluminum hydride. The specific interaction between the indium center and the oxygen atoms in the substrate creates a rigid transition state that favors the desired (S)-configuration. Furthermore, the workup procedure involving aqueous quenching and simple organic extraction effectively removes indium salts and siloxane byproducts, ensuring that the final crude material possesses a purity profile suitable for direct recrystallization. This inherent cleanliness of the reaction mixture translates directly to higher yields and reduced solvent consumption during purification.
How to Synthesize (3S)-3-[4-[(5-bromo-2-chlorophenyl)methyl]phenoxy]tetrahydrofuran Efficiently
Implementing this synthesis requires precise control over reaction parameters to maximize yield and stereoselectivity. The process begins with the preparation of the ether intermediate in a solvent system such as tetrahydrofuran or dioxane, maintaining temperatures between 15°C and 45°C to prevent degradation. Following isolation, the reduction step is conducted under an inert nitrogen atmosphere in toluene, where the stoichiometry of TMDS and the loading of the indium catalyst are critical variables. Detailed standard operating procedures regarding addition rates, quenching protocols, and crystallization conditions are essential for reproducible results. For a comprehensive guide on the specific molar ratios and post-treatment workflows, please refer to the standardized synthesis steps provided below.
- Etherify 3-(4-fluorophenylketone)-1-bromo-4-chlorobenzene with L-dimethyl malate using DIEA at 15-45°C.
- Reduce the intermediate using TMDS and Indium Bromide catalyst in toluene at 10-30°C.
- Quench, wash, and purify via recrystallization or pulping to obtain the final tetrahydrofuran derivative.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this patented methodology represents a strategic opportunity to de-risk the supply of empagliflozin intermediates. By replacing volatile and hazardous reagents with stable, commercially available alternatives, manufacturers can significantly reduce the regulatory burden and insurance costs associated with chemical storage and transport. The elimination of cryogenic requirements (-78°C) typically needed for organolithium reactions allows for the use of standard jacketed reactors, thereby lowering capital expenditure barriers for contract manufacturing organizations. Additionally, the use of L-dimethyl malate as a chiral pool source is far more cost-effective than purchasing protected chiral tetrahydrofurans, leading to substantial raw material cost savings.
- Cost Reduction in Manufacturing: The economic benefits of this route are driven primarily by the substitution of expensive chiral building blocks with commodity chemicals like L-dimethyl malate. Traditional routes often rely on multi-step syntheses to install the chiral tetrahydrofuran ring, whereas this method constructs the ring in situ during the reduction phase. This consolidation of steps reduces solvent usage, energy consumption, and labor hours per kilogram of output. Furthermore, the avoidance of boron tribromide eliminates the need for specialized corrosion-resistant equipment and complex neutralization procedures, further driving down operational expenditures.
- Enhanced Supply Chain Reliability: Supply continuity is bolstered by the use of robust reagents that are less susceptible to supply chain disruptions caused by hazardous material transport regulations. Potassium tert-butoxide and boron tribromide often face shipping restrictions and require cold chain logistics, whereas TMDS and indium bromide are stable solids or liquids that can be sourced from multiple global vendors. This diversification of the supply base ensures that production schedules remain uninterrupted even during periods of logistical volatility, providing a more reliable pharmaceutical intermediate supplier experience for downstream API manufacturers.
- Scalability and Environmental Compliance: The environmental footprint of this process is markedly lower due to the generation of less hazardous waste streams. The aqueous workup generates benign salt solutions rather than heavy metal sludge or corrosive acidic waste, simplifying wastewater treatment compliance. The high purity of the crude product (>98%) minimizes the need for extensive chromatographic purification, which is a major source of solvent waste in fine chemical synthesis. These factors collectively enhance the scalability of the process, allowing for seamless transition from pilot plant batches to multi-ton commercial production without encountering significant engineering bottlenecks.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this indium-catalyzed synthesis. Understanding these nuances is critical for R&D teams evaluating technology transfer and for quality assurance personnel establishing specification limits. The answers provided are derived directly from the experimental data and beneficial effects reported in the patent literature, ensuring accuracy and relevance for industrial application.
Q: What are the advantages of using Indium Bromide over traditional reducing agents?
A: Indium Bromide allows for a mild reduction temperature (10-30°C) and facilitates simultaneous reduction and cyclization, avoiding harsh conditions associated with boron tribromide or lithium aluminum hydride.
Q: How does this route improve safety compared to prior art?
A: This method eliminates the use of highly flammable potassium tert-butoxide and moisture-sensitive boron tribromide, significantly reducing operational hazards and storage risks.
Q: What is the expected purity of the final intermediate?
A: The patented process consistently yields the target empagliflozin intermediate with a purity greater than 99.0%, meeting stringent pharmaceutical specifications.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Empagliflozin Intermediate Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of securing a stable supply of high-quality intermediates for the global diabetes care market. Our technical team has extensively analyzed the route disclosed in CN113480497B and validated its potential for large-scale manufacturing. We possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that our clients receive consistent quality regardless of order volume. Our facility is equipped with rigorous QC labs capable of verifying stringent purity specifications, including chiral HPLC analysis to guarantee the optical integrity of the tetrahydrofuran moiety.
We invite pharmaceutical partners to engage with us for a Customized Cost-Saving Analysis tailored to your specific volume requirements. By leveraging this advanced synthetic technology, we can offer competitive pricing structures that reflect the efficiencies of the new process. Please contact our technical procurement team to request specific COA data, route feasibility assessments, and sample availability. Let us collaborate to bring safer and more affordable SGLT2 inhibitor therapies to patients worldwide through superior chemical manufacturing.
