Advanced Aqueous Synthesis of Diethyl p-Toluenesulfonyloxymethylphosphonate for Antiviral APIs
The pharmaceutical industry's relentless pursuit of safer, greener, and more cost-effective manufacturing processes has led to significant innovations in the synthesis of critical antiviral intermediates. Patent CN109053799B discloses a groundbreaking synthesis method for diethyl p-toluenesulfonyloxymethylphosphonate, a pivotal building block for first-line therapeutic drugs such as tenofovir disoproxil fumarate (HBV/HIV), adefovir disoproxil (HBV), and cidofovir (CMV). This novel approach fundamentally shifts the paradigm from hazardous organic solvent-based systems to a benign aqueous phase transfer catalysis (PTC) regime. By leveraging water as the primary reaction medium and replacing expensive organic bases with cheap inorganic alternatives, this technology addresses the triple constraints of safety, environmental compliance, and economic efficiency that modern chemical supply chains face.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the production of diethyl p-toluenesulfonyloxymethylphosphonate has been plagued by significant operational and environmental drawbacks inherent to traditional organic synthesis. Conventional routes typically rely on volatile organic compounds (VOCs) such as toluene, dichloromethane, or N,N-dimethylformamide (DMF) as solvents, which pose severe flammability risks and require complex recovery systems to meet environmental regulations. Furthermore, these methods often employ triethylamine as both a catalyst and an acid scavenger; this reagent is not only costly and malodorous but also generates substantial amounts of amine salt waste that is difficult to treat. Prior art methods frequently suffer from mediocre yields, often ranging between 72% and 80%, and necessitate labor-intensive purification steps like silica gel column chromatography or multiple extraction cycles, which drastically increase production time and energy consumption.
The Novel Approach
In stark contrast, the methodology outlined in CN109053799B introduces a streamlined, two-step aqueous process that eliminates the need for toxic organic solvents entirely. The innovation lies in the strategic use of a phase transfer catalyst (such as quaternary ammonium salts or polyethylene glycols) to facilitate the reaction between diethyl phosphite and paraformaldehyde in water. This allows for a high-efficiency condensation followed directly by esterification with p-toluenesulfonyl chloride in the same medium. The result is a dramatic simplification of the workflow: the product separates via simple layering after the reaction, bypassing the need for distillation or chromatography. This shift not only enhances operator safety by removing explosive solvents but also drastically reduces the generation of hazardous waste, aligning perfectly with green chemistry principles.

Mechanistic Insights into Phase Transfer Catalyzed Aqueous Synthesis
The core of this technological breakthrough is the efficient mass transfer enabled by the phase transfer catalyst in a heterogeneous aqueous system. In the initial condensation step, the inorganic base (e.g., sodium carbonate or potassium carbonate) deprotonates the diethyl phosphite at the interface. The phase transfer catalyst, typically a lipophilic quaternary ammonium cation, shuttles the reactive phosphite anion into the organic-like microenvironment where it reacts with the depolymerized formaldehyde. This mechanism accelerates the formation of the diethyl hydroxymethylphosphonate intermediate while suppressing side reactions such as the hydrolysis of the phosphonate ester, which is a common issue in purely aqueous acidic or basic conditions without PTC.
Following the condensation, the subsequent esterification with p-toluenesulfonyl chloride (TsCl) proceeds with remarkable selectivity. The controlled addition of TsCl in batches at low temperatures prevents the exothermic runaway often seen in organic solvents, thanks to the high heat capacity of water. The phase transfer catalyst continues to play a vital role by activating the hydroxymethyl intermediate for nucleophilic attack on the sulfonyl chloride. This precise control over reaction kinetics minimizes the formation of impurities, such as bis-tosylated byproducts or hydrolyzed phosphonic acids, ensuring that the final crude product achieves a purity of over 98% directly after phase separation.
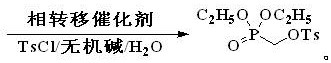
How to Synthesize Diethyl p-Toluenesulfonyloxymethylphosphonate Efficiently
The synthesis protocol described in the patent offers a robust framework for manufacturing this key intermediate with high reproducibility. The process begins with the preparation of an aqueous solution containing the phase transfer catalyst and inorganic base, into which paraformaldehyde is dissolved. Diethyl phosphite is then added dropwise under strict temperature control to manage the exotherm, followed by a heating phase to drive the condensation to completion. Once the intermediate is formed, the reaction mixture is cooled, and p-toluenesulfonyl chloride is introduced in portions to effect the esterification. The detailed standardized synthesis steps for implementing this green chemistry route are provided below.
- Dissolve paraformaldehyde, inorganic base (e.g., sodium carbonate), and a phase transfer catalyst in water, then add diethyl phosphite dropwise at low temperature.
- Heat the mixture to 60-110°C to perform the condensation reaction, forming the diethyl hydroxymethylphosphonate intermediate.
- Cool the reaction, add p-toluenesulfonyl chloride in batches, heat to 20-60°C for esterification, and separate the product via layering.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this aqueous synthesis route translates into tangible strategic advantages beyond mere regulatory compliance. The elimination of expensive organic solvents and the replacement of triethylamine with commodity inorganic salts result in a substantially reduced bill of materials. Furthermore, the simplified workup procedure, which relies on gravity separation rather than energy-intensive distillation or chromatography, significantly shortens the batch cycle time. This efficiency gain allows for higher throughput in existing reactor trains, effectively increasing capacity without capital expenditure on new equipment.
- Cost Reduction in Manufacturing: The substitution of high-cost organic solvents and bases with water and inorganic carbonates leads to a drastic reduction in raw material expenses. Additionally, the removal of complex purification steps like column chromatography lowers labor and utility costs, while the high yield minimizes the cost of goods sold by maximizing output per batch.
- Enhanced Supply Chain Reliability: By utilizing widely available and non-hazardous raw materials, the risk of supply disruption due to solvent shortages or transportation restrictions is mitigated. The inherent safety of the aqueous process also reduces the likelihood of unplanned shutdowns caused by safety incidents, ensuring a more consistent and reliable delivery schedule for downstream API manufacturers.
- Scalability and Environmental Compliance: The process is inherently scalable due to the excellent heat dissipation properties of water, allowing for safe operation in large-scale reactors. The significant reduction in VOC emissions and hazardous waste generation simplifies environmental permitting and waste disposal logistics, future-proofing the supply chain against increasingly stringent global environmental regulations.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this novel synthesis technology. These insights are derived directly from the experimental data and beneficial effects reported in the patent literature, providing a clear understanding of the process capabilities and limitations for potential partners.
Q: What are the primary advantages of the aqueous synthesis method over traditional organic solvent routes?
A: The aqueous method eliminates toxic organic solvents like toluene and dichloromethane, significantly reducing VOC emissions and fire hazards while lowering raw material costs through the use of inexpensive inorganic bases.
Q: What yields and purity levels can be expected from this phase transfer catalysis process?
A: The patented process consistently achieves two-step reaction yields exceeding 93% and product purity levels greater than 98%, minimizing the need for complex purification steps like column chromatography.
Q: Is this synthesis route scalable for industrial production of antiviral intermediates?
A: Yes, the use of water as a solvent and simple layering for separation makes the process highly scalable, safe, and environmentally compliant for large-scale manufacturing of HIV and HBV drug intermediates.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Diethyl p-Toluenesulfonyloxymethylphosphonate Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-quality intermediates play in the global supply of antiviral medications. As a specialized CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that our clients receive materials that meet stringent purity specifications. Our rigorous QC labs are equipped to verify the identity and purity of every batch, guaranteeing that the diethyl p-toluenesulfonyloxymethylphosphonate we supply is fully compliant with the highest industry standards for pharmaceutical manufacturing.
We invite you to collaborate with us to leverage this advanced aqueous synthesis technology for your supply chain. Our technical team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements. Please contact our technical procurement team today to request specific COA data and route feasibility assessments, and let us demonstrate how our innovative manufacturing capabilities can drive value for your organization.
