Scaling Enzymatic Asymmetric Aldol Reactions for High-Purity Pharmaceutical Intermediates
The pharmaceutical and fine chemical industries are currently witnessing a paradigm shift towards greener, more sustainable synthetic methodologies, driven by stringent regulatory pressures and the need for cost-effective manufacturing. A pivotal development in this arena is detailed in patent CN102517353A, which discloses the novel application of porcine pancreatic lipase (PPL) as a highly efficient biocatalyst for the asymmetric aldol reaction between heterocyclic ketones and aromatic aldehydes. This technology represents a significant breakthrough in asymmetric synthesis, offering a robust alternative to traditional metal-catalyzed or organocatalytic routes that often suffer from toxicity issues and harsh reaction conditions. By leveraging the promiscuous catalytic activity of this readily available enzyme, manufacturers can access chiral building blocks with high enantiomeric excess (ee) values under remarkably mild conditions. For R&D directors and process chemists, this patent outlines a pathway to synthesize complex heterocyclic intermediates—critical scaffolds in modern drug discovery—with improved stereocontrol and reduced environmental footprint, positioning it as a key technology for the next generation of reliable pharmaceutical intermediate supplier networks.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditionally, the construction of carbon-carbon bonds via the aldol reaction has relied heavily on stoichiometric amounts of strong bases or transition metal complexes, which present substantial challenges for industrial scale-up. Conventional metal-ligand catalysts, while effective for simple substrates, often exhibit poor performance when applied to sterically demanding heterocyclic ketones, leading to low yields and inadequate diastereoselectivity. Furthermore, the presence of residual heavy metals in the final product is a critical quality attribute (CQA) concern for high-purity pharmaceutical intermediates, necessitating expensive and time-consuming purification steps to meet regulatory limits. Organocatalysts have emerged as an alternative, yet they frequently require cryogenic temperatures, anhydrous conditions, and prolonged reaction times to achieve acceptable enantioselectivity, thereby increasing energy consumption and operational complexity. These limitations create a bottleneck in the commercial scale-up of complex polymer additives and active pharmaceutical ingredients (APIs), where efficiency and purity are paramount.
The Novel Approach
In stark contrast, the methodology described in CN102517353A utilizes porcine pancreatic lipase to catalyze the direct asymmetric aldol reaction in a water-containing organic solvent system, effectively bypassing the drawbacks of traditional chemistry. This enzymatic approach operates at ambient temperatures (preferably 30°C) and tolerates the presence of water, which not only simplifies the reaction setup but also aligns with green chemistry principles. The patent data demonstrates that PPL exhibits remarkable substrate tolerance, particularly with heterocyclic ketones such as 1-Boc-4-piperidone, tetrahydropyran-4-one, and tetrahydrothiopyran-4-one. By employing aromatic aldehydes bearing electron-withdrawing groups, the process achieves yields ranging from 31% to 56% with enantiomeric excess values reaching up to 87%. This novel biocatalytic route eliminates the need for toxic metals and harsh reagents, offering a streamlined path for cost reduction in API manufacturing while ensuring the production of high-value chiral intermediates with defined stereochemistry.
Mechanistic Insights into Porcine Pancreatic Lipase Catalyzed Asymmetric Aldol Reaction
The success of this transformation lies in the unique catalytic promiscuity of porcine pancreatic lipase, an enzyme naturally evolved for ester hydrolysis but capable of facilitating C-C bond formation under specific conditions. The reaction mechanism likely involves the formation of an enol or enamine-like intermediate within the enzyme's active site, which then attacks the electrophilic carbonyl carbon of the aromatic aldehyde. The patent data highlights the critical influence of the electronic nature of the aldehyde substrate; aromatic aldehydes substituted with strong electron-withdrawing groups (such as nitro, cyano, or halogen atoms) significantly enhance reactivity by increasing the positive polarity of the carbonyl carbon. This electronic activation facilitates the nucleophilic attack by the ketone enolate, explaining why substrates like 4-nitrobenzaldehyde perform exceptionally well compared to those with electron-donating groups. Furthermore, the steric environment of the enzyme's active pocket plays a pivotal role in stereocontrol, as evidenced by the high diastereoselectivity observed with ortho-substituted aldehydes.
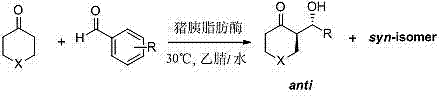
Stereoselectivity in this enzymatic system is governed by a delicate balance of steric hindrance and electronic effects within the transition state. The patent results indicate that increasing the steric bulk near the reaction center, such as using 2-nitrobenzaldehyde, can lead to improved enantioselectivity and cis-selectivity (syn-isomer formation), suggesting that the enzyme pocket restricts the approach of the nucleophile to a specific face of the aldehyde. This level of control is difficult to achieve with small-molecule catalysts without elaborate ligand design. Additionally, the stability of the enzyme's tertiary structure is crucial; experiments with urea-inactivated or PMSF-treated lipase showed a drastic loss of activity and selectivity, confirming that the catalytic function relies on the intact active site rather than non-specific protein interactions. Understanding these mechanistic nuances allows process chemists to fine-tune reaction parameters, such as solvent composition and water content, to maximize the efficiency of this commercial scale-up of complex pharmaceutical intermediates.
How to Synthesize Chiral Aldol Products Efficiently
To implement this technology effectively, precise control over reaction parameters is essential to replicate the high selectivity reported in the patent. The optimal protocol involves dissolving the heterocyclic ketone and aromatic aldehyde in a mixture of acetonitrile and water, with a specific water-to-solvent volume ratio of 0.1 being critical for balancing enzyme activity and substrate solubility. The reaction is typically conducted at 30°C with stirring for approximately 120 hours, although monitoring via TLC is recommended to determine the exact endpoint for specific substrate pairs. The following guide outlines the standardized procedure derived from the patent embodiments to ensure reproducible results and high purity.
- Prepare the reaction system by mixing acetonitrile and water in a volume ratio of approximately 9: 1 (water content 0.1) within a reaction flask.
- Add the heterocyclic ketone (e.g., 1-Boc-4-piperidone) and the aromatic aldehyde (e.g., 4-nitrobenzaldehyde) to the solvent system, maintaining a molar ratio between 1: 1 and 1:3.
- Introduce porcine pancreatic lipase (approx. 50mg per 0.25mmol ketone) and stir the mixture at a controlled temperature of 30°C for 120 hours.
- Upon completion, filter the mixture, wash the cake with dichloromethane, extract the filtrate, dry over anhydrous sodium sulfate, and purify via flash column chromatography.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain leaders, the adoption of this enzymatic technology offers tangible strategic advantages beyond mere technical feasibility. The shift from metal-based catalysis to biocatalysis fundamentally alters the cost structure and risk profile of the manufacturing process. By eliminating the need for expensive transition metal ligands and the associated rigorous purification steps required to remove trace metals, the overall cost of goods sold (COGS) can be significantly optimized. Furthermore, the use of a commercially available, non-toxic enzyme like porcine pancreatic lipase mitigates supply chain risks associated with the sourcing of rare earth metals or specialized organocatalysts, ensuring greater continuity of supply for critical intermediates.
- Cost Reduction in Manufacturing: The enzymatic process operates under mild conditions (30°C) and utilizes inexpensive, commodity solvents like acetonitrile, which drastically reduces energy consumption compared to cryogenic organocatalytic methods. The elimination of heavy metal catalysts removes the necessity for specialized scavenging resins or complex chromatographic purification solely for metal removal, leading to substantial cost savings in downstream processing. Additionally, the high stereoselectivity reduces the formation of unwanted diastereomers, minimizing material waste and improving the overall atom economy of the synthesis.
- Enhanced Supply Chain Reliability: Porcine pancreatic lipase is a robust, commercially available biocatalyst with a stable supply chain, unlike many bespoke chiral ligands that may have long lead times or single-source dependencies. The reaction's tolerance to water and ambient temperatures simplifies equipment requirements, allowing production to be flexible across different manufacturing sites without the need for specialized low-temperature reactors. This flexibility enhances the resilience of the supply chain against disruptions and allows for faster response times to market demand fluctuations.
- Scalability and Environmental Compliance: The green nature of this process aligns perfectly with increasingly strict environmental regulations regarding solvent use and heavy metal discharge. The aqueous-organic solvent system is easier to handle and recycle compared to strictly anhydrous systems, and the biodegradable nature of the enzyme catalyst simplifies waste treatment protocols. This environmental compliance facilitates smoother regulatory approvals and reduces the long-term liability associated with hazardous waste disposal, making it an ideal candidate for sustainable large-scale production.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this porcine pancreatic lipase catalyzed aldol reaction. These insights are derived directly from the experimental data and beneficial effects described in the patent documentation, providing clarity on substrate scope, reaction optimization, and scalability potential for prospective partners.
Q: Why is Porcine Pancreatic Lipase preferred over traditional metal catalysts for this aldol reaction?
A: Porcine Pancreatic Lipase offers superior environmental safety by eliminating toxic heavy metals, operates under mild conditions (30°C), and provides excellent enantioselectivity (up to 87% ee) for heterocyclic substrates where traditional organocatalysts often struggle.
Q: What specific substrate characteristics are required for high yield and selectivity?
A: The reaction performs optimally with aromatic aldehydes containing strong electron-withdrawing substituents (such as nitro or cyano groups) on the phenyl ring, which activate the carbonyl carbon for nucleophilic attack by the enolizable heterocyclic ketone.
Q: Is this enzymatic process suitable for large-scale commercial manufacturing?
A: Yes, the process utilizes commercially available enzymes and standard organic solvents like acetonitrile. The mild temperature requirements and absence of sensitive metal catalysts significantly simplify scale-up and reduce downstream purification costs.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Porcine Pancreatic Lipase Catalyst Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of biocatalysis in modern pharmaceutical synthesis and are committed to bridging the gap between academic innovation and industrial application. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the promising results seen in patent CN102517353A can be successfully translated into robust manufacturing processes. We maintain stringent purity specifications and operate rigorous QC labs to guarantee that every batch of chiral intermediate meets the exacting standards required by global regulatory bodies, providing our clients with peace of mind regarding product quality and consistency.
We invite you to collaborate with us to explore the full potential of this enzymatic aldol reaction for your specific drug discovery programs. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your project needs, demonstrating how this green chemistry approach can optimize your budget without compromising on quality. Please contact us today to request specific COA data and route feasibility assessments, and let us help you accelerate your path to market with reliable, high-performance chemical solutions.
