Advanced Manufacturing of Deuterium-Labeled Ritonavir for Metabolic Research and Supply Chain Stability
Advanced Manufacturing of Deuterium-Labeled Ritonavir for Metabolic Research and Supply Chain Stability
The pharmaceutical industry's relentless pursuit of understanding drug metabolism has elevated the importance of stable isotope-labeled compounds, particularly for HIV protease inhibitors like Ritonavir. Patent CN103896870A discloses a robust and highly efficient preparation method for Deuterium-Labeled Ritonavir, addressing the critical need for high-purity standards in pharmacokinetic profiling. This technology leverages a strategic seven-step synthesis starting from isobutyric acid-D6, ensuring that the deuterium label is securely integrated into the molecular framework from the outset. By optimizing reaction conditions and solvent systems, the process achieves a final product purity exceeding 98% and an isotopic abundance greater than 99%, setting a new benchmark for reliability in the supply of complex pharmaceutical intermediates.
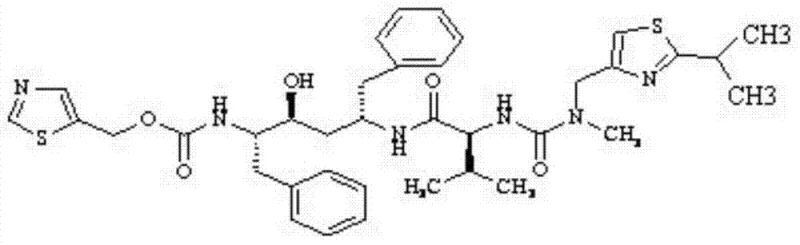
For R&D directors and procurement specialists, the significance of this patent lies in its ability to provide a consistent source of D6-Ritonavir for metabolic mechanism research. Unlike random labeling techniques that can lead to isotopic scrambling and unreliable data, this method targets specific methyl groups, preserving the integrity of the tracer throughout the biological assay. The structural complexity of Ritonavir, characterized by multiple chiral centers and heterocyclic rings, demands a synthesis route that is both chemoselective and high-yielding. The disclosed method meets these rigorous demands, offering a viable pathway for the commercial production of this high-value specialty chemical.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional approaches to synthesizing deuterated analogs of complex APIs often suffer from significant inefficiencies, particularly when relying on late-stage hydrogen-deuterium exchange reactions. These conventional methods frequently require harsh conditions that can compromise the stereochemical integrity of sensitive molecules like Ritonavir, leading to racemization and the formation of difficult-to-remove impurities. Furthermore, late-stage labeling typically results in poor isotopic incorporation rates, necessitating extensive and costly purification steps such as preparative HPLC to achieve the required >99% abundance. From a supply chain perspective, these inefficiencies translate into prolonged lead times and unpredictable batch-to-batch variability, which are unacceptable for GMP-grade intermediate manufacturing.
The Novel Approach
The novel approach detailed in the patent circumvents these pitfalls by employing a 'building block' strategy that introduces the deuterium label at the very beginning of the synthesis using isobutyric acid-D6. This foundational step ensures that the isotopic label is locked into the carbon skeleton before the construction of the complex thiazole and peptide backbone begins. By screening optimal reaction conditions for each of the seven steps, including precise control of temperature and stoichiometry, the process maximizes yield while minimizing side reactions. This rational design not only simplifies the purification workflow but also enhances the overall atom economy, making the production of deuterated Ritonavir more economically viable and environmentally sustainable compared to legacy methods.
Mechanistic Insights into Deuterated Thiazole Ring Construction
A critical juncture in this synthesis is the formation of the deuterated thiazole ring, which serves as the core scaffold carrying the isotopic label. The process begins with the conversion of the deuterated amide intermediate into a thioamide using thiophosphoric anhydride (Lawesson's reagent) in a high-boiling non-polar solvent like tetrahydrofuran. This thionation step is pivotal, as it activates the carbonyl carbon for subsequent nucleophilic attack. Following this, the thioamide undergoes a cyclization reaction with acetone halides, such as 1,3-dibromoacetone, in a ketone solvent. This ring-closing step effectively constructs the five-membered thiazole heterocycle while retaining the six deuterium atoms on the adjacent isopropyl group, demonstrating the robustness of the C-D bonds under the reaction conditions.
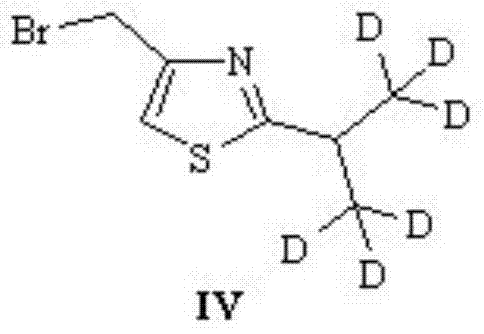
Following the formation of the thiazole core, the synthesis proceeds through a series of peptide couplings that require meticulous control to prevent epimerization. The intermediate thiazole derivative is subjected to electrophilic substitution with methylamine to generate the necessary amine functionality for chain elongation. Subsequent coupling with a valine derivative involves the activation of the carboxylic acid group, typically using chloroformates to form mixed anhydrides in situ. This activation strategy is chosen for its mildness and compatibility with the sensitive deuterated motifs. The final assembly involves coupling the deuterated fragment with the remaining Ritonavir core, a step that demands high chemoselectivity to ensure the final API intermediate meets the stringent purity specifications required for clinical research applications.
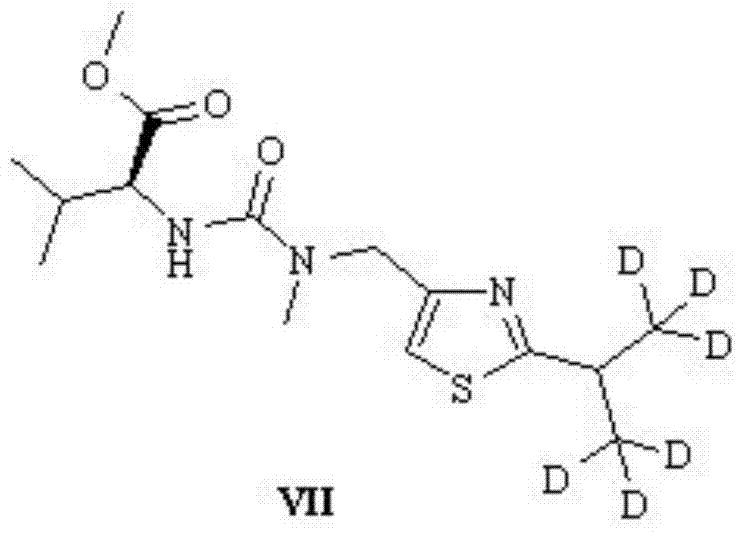
How to Synthesize Deuterated Ritonavir Efficiently
The synthesis of Deuterated Ritonavir described in this patent represents a streamlined pathway that balances chemical complexity with operational simplicity. By breaking down the synthesis into seven distinct, optimized stages, the process allows for quality control checkpoints at each intermediate stage, ensuring that any deviations are caught early. The use of common industrial solvents and reagents facilitates easy technology transfer from the laboratory to pilot and commercial scales. For process chemists looking to implement this route, the key lies in maintaining strict anhydrous conditions during the activation steps and controlling the temperature profiles during the exothermic cyclization reactions to maximize the yield of the desired stereoisomer.
- Acylation and aminolysis of isobutyric acid-D6 to form the deuterated amide intermediate.
- Thionation using thiophosphoric anhydride followed by cyclization with acetone halides to construct the deuterated thiazole ring.
- Sequential peptide coupling and hydrolysis steps to assemble the final Ritonavir backbone while preserving isotopic integrity.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this patented synthesis route offers substantial strategic advantages over sourcing deuterated materials from limited specialty vendors. The primary benefit stems from the use of readily available starting materials, specifically isobutyric acid-D6, which eliminates the dependency on scarce or prohibitively expensive custom-labeled precursors. This accessibility translates directly into a more resilient supply chain, reducing the risk of production delays caused by raw material shortages. Furthermore, the high overall yield reported in the patent implies a significant reduction in the cost of goods sold, as less starting material is wasted in purification losses, allowing for more competitive pricing in the global market for stable isotope-labeled standards.
- Cost Reduction in Manufacturing: The elimination of complex late-stage labeling steps removes the need for expensive catalytic systems and extensive chromatographic purification, which are major cost drivers in isotope chemistry. By integrating the label early, the process minimizes the number of unit operations required to achieve high isotopic purity, thereby lowering energy consumption and solvent usage. This streamlined workflow results in a drastic simplification of the manufacturing process, leading to substantial cost savings that can be passed down to the end-user without compromising on the quality or specification of the final deuterated API intermediate.
- Enhanced Supply Chain Reliability: The reliance on standard chemical reagents such as phosphorus pentachloride, thiophosphoric anhydride, and common organic solvents ensures that the supply chain is not vulnerable to the bottlenecks often associated with specialized catalysts. The robustness of the reaction conditions, which tolerate a range of temperatures and solvent choices, provides manufacturing flexibility that is crucial for maintaining continuous production schedules. This operational resilience ensures that lead times for high-purity deuterated compounds can be significantly reduced, providing R&D teams with the timely materials they need to accelerate drug development timelines and meet regulatory submission deadlines.
- Scalability and Environmental Compliance: The process is designed with scalability in mind, utilizing reaction vessels and workup procedures that are easily adaptable from kilogram to multi-ton scales. The avoidance of heavy metal catalysts simplifies the waste treatment process, aligning with increasingly stringent environmental regulations regarding heavy metal discharge in pharmaceutical manufacturing. Additionally, the high purity of the crude product reduces the solvent load associated with recrystallization and column chromatography, contributing to a greener manufacturing footprint. This combination of scalability and environmental compliance makes the technology an attractive option for long-term commercial partnerships focused on sustainable chemical production.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production and application of Deuterated Ritonavir. These insights are derived directly from the experimental data and process descriptions found in the patent literature, providing a transparent view of the technology's capabilities. Understanding these details is essential for stakeholders evaluating the feasibility of incorporating this labeled standard into their metabolic research pipelines or supply chains.
Q: What is the isotopic abundance of the Deuterated Ritonavir produced via this method?
A: The patented process ensures an isotopic abundance greater than 99%, which is critical for accurate metabolic tracing and pharmacokinetic studies without signal interference from natural abundance isotopes.
Q: How does this synthesis route improve upon traditional labeling methods?
A: By utilizing isobutyric acid-D6 as a starting material, the method incorporates the deuterium label early in the synthesis. This 'bottom-up' approach avoids the low yields and complex purification often associated with late-stage hydrogen-deuterium exchange reactions.
Q: Is this process scalable for commercial production of API intermediates?
A: Yes, the process utilizes standard industrial solvents like tetrahydrofuran and methylene dichloride, and avoids exotic catalysts. The overall yield is reported to be greater than 80%, indicating strong potential for commercial scale-up.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Deuterated Ritonavir Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-quality isotopically labeled compounds play in advancing pharmaceutical research and development. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that we can meet the demanding volume requirements of global clinical trials and commercial launches. We are committed to delivering products with stringent purity specifications and rigorous QC labs testing every batch to guarantee isotopic abundance and chemical integrity. Our state-of-the-art facilities are equipped to handle the specific safety and containment requirements associated with potent API intermediates, providing a secure and compliant manufacturing environment.
We invite you to collaborate with us to optimize your supply chain for deuterated materials. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your specific project needs. We are ready to provide specific COA data and route feasibility assessments to demonstrate how our advanced manufacturing capabilities can support your drug development goals efficiently and reliably.
