Scalable Manufacturing of Tenapanor Intermediates via Novel Grignard-Sulfur Dioxide Insertion Technology
Introduction to Advanced Tenapanor Manufacturing Technologies
The pharmaceutical landscape for treating constipation-predominant irritable bowel syndrome (IBS-C) has been significantly advanced by the development of Tenapanor, a potent sodium/hydrogen exchanger 3 (NHE3) inhibitor. As global demand for this therapeutic agent grows, the efficiency and safety of its supply chain become paramount for stakeholders. A pivotal breakthrough in this domain is detailed in Chinese Patent CN112047881B, which discloses a revolutionary preparation method that fundamentally alters the synthetic strategy for constructing the critical sulfonamide linkage. Unlike traditional multi-step approaches relying on precious metal catalysis, this innovation leverages a direct Grignard reagent formation followed by sulfur dioxide insertion and oxidative amidation. This technical evolution represents a paradigm shift towards greener, more cost-effective pharmaceutical intermediates manufacturing, addressing long-standing challenges regarding toxicity and operational complexity.
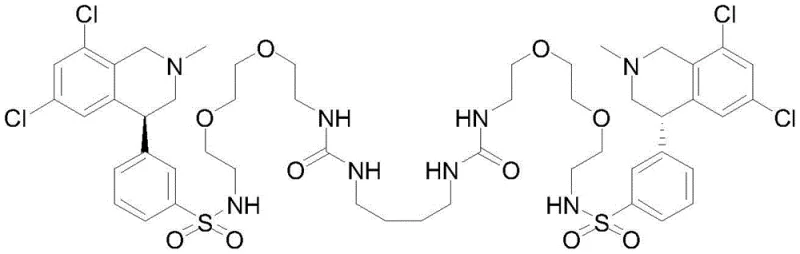
The strategic importance of this patent lies in its ability to streamline the production of high-purity Tenapanor while mitigating severe environmental and safety hazards associated with legacy processes. For R&D directors and procurement managers alike, understanding the mechanistic underpinnings of this Grignard-based route is essential for evaluating potential technology transfers or licensing opportunities. The method utilizes readily available starting materials such as (S)-4-(3-bromophenyl)-6,8-dichloro-2-methyl-1,2,3,4-tetrahydroisoquinoline and transforms them through a sophisticated yet operationally simple sequence. By integrating sulfur dioxide directly into the reaction matrix without isolating unstable intermediates, the process achieves remarkable convergence, setting a new benchmark for commercial scale-up of complex API intermediates.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Prior art methodologies, such as those disclosed in WO2010078449, have historically relied on palladium-catalyzed cross-coupling reactions to install the sulfur functionality necessary for the sulfonamide bond. These conventional routes typically involve the use of benzyl mercaptan, a reagent notorious for its extremely foul odor and significant air pollution potential, posing serious challenges for facility containment and worker safety. Furthermore, the subsequent chlorination step often necessitates the handling of chlorine gas, a highly toxic and corrosive substance that requires specialized equipment and rigorous safety protocols. Beyond the safety concerns, the reliance on palladium catalysts introduces substantial cost burdens due to the high price of the metal and the stringent requirements for removing trace heavy metals from the final active pharmaceutical ingredient to meet regulatory standards.
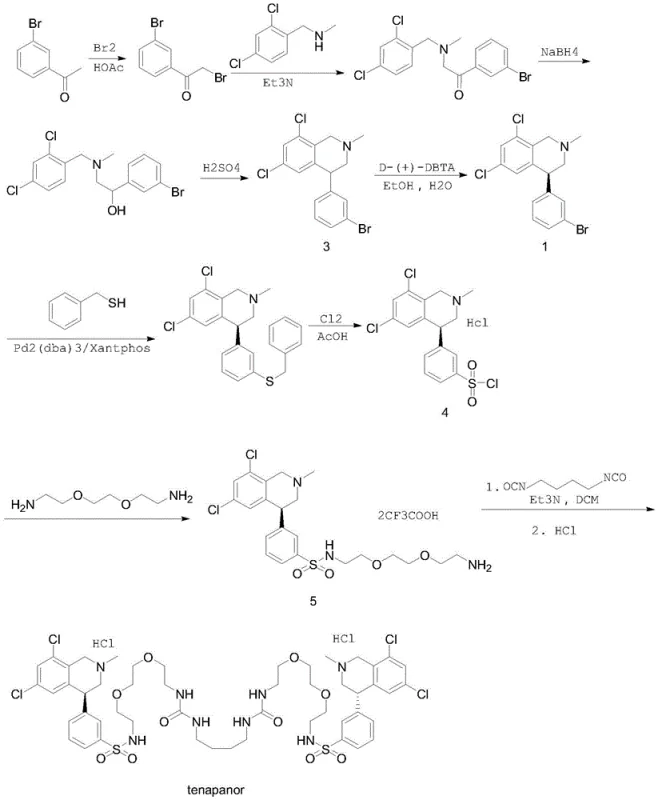
In addition to safety and cost issues, the traditional liquid-phase preparation of key intermediates often involves cumbersome isolation and purification steps that reduce overall throughput. The use of trifluoroacetic acid salts and multiple protection-deprotection sequences further complicates the workflow, leading to increased solvent consumption and waste generation. For a reliable API intermediate supplier, these inefficiencies translate into longer lead times and higher variability in batch quality. The cumulative effect of these drawbacks makes the conventional palladium-mediated pathways less attractive for modern, high-volume manufacturing environments where sustainability and operational excellence are critical performance indicators.
The Novel Approach
The methodology presented in CN112047881B offers a transformative solution by bypassing the need for transition metal catalysis entirely. Instead, it employs a "Turbo" Grignard reagent strategy, utilizing an isopropyl magnesium chloride-lithium chloride complex to activate the aryl bromide precursor under mild conditions. This activation allows for the direct insertion of sulfur dioxide to generate a sulfinic acid magnesium chloride salt in situ, which is then immediately subjected to oxidative amidation. This one-pot design eliminates the isolation of hazardous intermediates and removes the dependency on odorous thiols or toxic chlorine gas. The result is a streamlined process that significantly enhances the safety profile of the manufacturing operation while simultaneously improving the atom economy of the transformation.
Moreover, the novel approach demonstrates superior scalability due to its simplified work-up procedures and the use of inexpensive oxidants like sodium hypochlorite. By conducting the reaction in solvents such as tetrahydrofuran or 2-methyltetrahydrofuran, the process maintains excellent solubility profiles for the reactants and intermediates, ensuring consistent reaction kinetics. This robustness is vital for cost reduction in pharmaceutical intermediates manufacturing, as it minimizes the risk of batch failures and reduces the consumption of auxiliary materials. The elimination of palladium not only cuts direct material costs but also simplifies the downstream purification train, allowing for faster turnaround times and more reliable supply continuity for global markets.
Mechanistic Insights into Grignard-Mediated Sulfur Dioxide Insertion
The core of this innovative synthesis lies in the precise control of organometallic chemistry to achieve selective functionalization. The process initiates with the formation of the Grignard reagent from the chiral tetrahydroisoquinoline bromide using iPrMgCl·LiCl. The presence of lithium chloride is crucial as it enhances the solubility and reactivity of the magnesium species, facilitating rapid halogen-metal exchange even at low temperatures ranging from -20°C to 0°C. This "Turbo" Grignard species is highly nucleophilic yet sufficiently stable to allow for controlled addition of sulfur dioxide. Upon introduction of SO2 gas, the Grignard reagent undergoes insertion to form the corresponding sulfinic acid magnesium chloride salt. This step is exothermic and requires careful thermal management, typically maintained between -20°C and 35°C, to prevent side reactions such as homocoupling or decomposition of the sensitive organometallic intermediate.
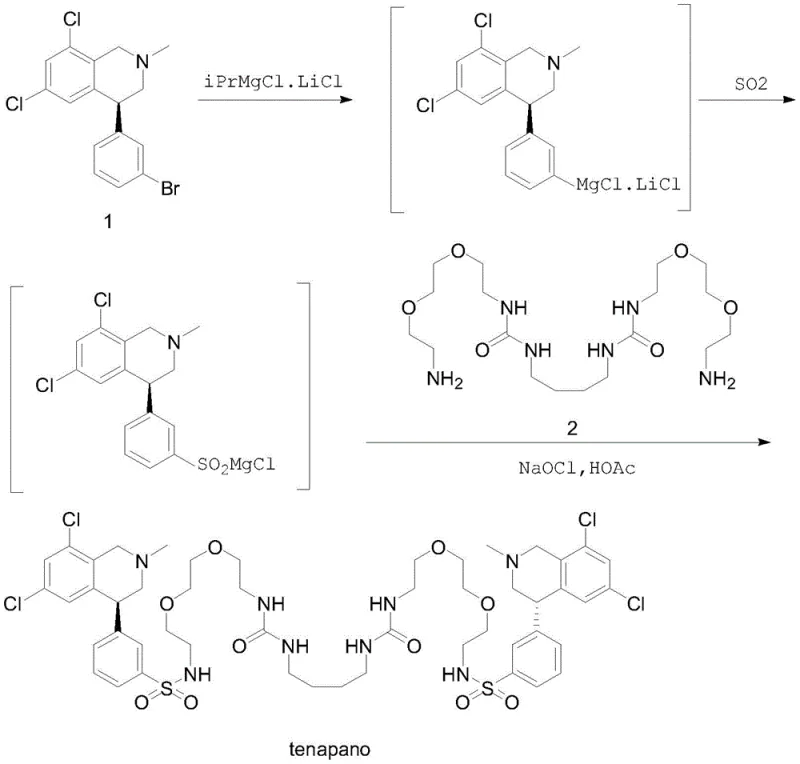
Following the formation of the sulfinate, the reaction mixture is treated with the diamine linker (Compound 2) in the presence of sodium hypochlorite and acetic acid. This oxidative amidation step converts the sulfinate into a reactive sulfonyl species, likely a sulfonyl chloride or a hypervalent sulfur intermediate, which is immediately trapped by the amine nucleophile to form the stable sulfonamide bond. The use of aqueous sodium hypochlorite as the oxidant is particularly advantageous from a green chemistry perspective, as it generates benign salt byproducts compared to harsher chlorinating agents. The entire sequence is designed to proceed without isolating the sulfinate, thereby minimizing exposure to air and moisture which could degrade the intermediate. This mechanistic elegance ensures high purity of the final product by suppressing the formation of impurities associated with intermediate handling and storage.
How to Synthesize Tenapanor Efficiently
The execution of this synthesis requires strict adherence to anhydrous conditions during the Grignard formation and SO2 insertion phases, followed by a controlled transition to the oxidative amidation conditions. Operators must ensure that the stoichiometry of the Turbo Grignard reagent is carefully balanced, typically using a slight excess (1.0 to 1.2 equivalents) relative to the bromo-substrate to drive the conversion to completion. The introduction of sulfur dioxide must be monitored to ensure saturation of the reaction medium without excessive pressure buildup. Once the sulfinate is formed, the addition of the amine linker and the oxidant system must be timed precisely to maximize the coupling efficiency. Detailed standard operating procedures for temperature ramps, addition rates, and quenching protocols are essential for reproducing the high yields reported in the patent examples.
- Preparation of Turbo Grignard Reagent: React (S)-4-(3-bromophenyl)-6,8-dichloro-2-methyl-1,2,3,4-tetrahydroisoquinoline with isopropyl magnesium chloride-lithium chloride complex at -20 to 0°C.
- Sulfur Dioxide Insertion: Introduce sulfur dioxide gas into the Grignard solution at -20 to 35°C to form the sulfinic acid magnesium chloride salt intermediate in situ.
- Oxidative Amidation: React the sulfinate intermediate with the diamine linker (Compound 2) using sodium hypochlorite and acetic acid at -5 to 35°C to yield Tenapanor.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain leaders, the adoption of this Grignard-based route offers compelling economic and logistical benefits that extend beyond simple yield improvements. The most significant advantage is the complete elimination of palladium catalysts, which are subject to volatile market pricing and supply constraints. By substituting this precious metal with abundant magnesium reagents, manufacturers can achieve substantial cost savings on raw materials while insulating their supply chains from geopolitical fluctuations affecting rare metal availability. Furthermore, the removal of benzyl mercaptan and chlorine gas from the process inventory drastically reduces the regulatory burden and insurance costs associated with storing and handling hazardous chemicals, contributing to a lower total cost of ownership for the production facility.
- Cost Reduction in Manufacturing: The avoidance of expensive palladium catalysts and the simplification of the purification process lead to significant reductions in production costs. Without the need for specialized scavengers to remove trace heavy metals, the downstream processing becomes faster and less resource-intensive. Additionally, the one-pot nature of the reaction reduces solvent usage and energy consumption associated with multiple isolation and drying steps, further enhancing the overall economic efficiency of the manufacturing campaign.
- Enhanced Supply Chain Reliability: By utilizing commodity chemicals like sulfur dioxide and sodium hypochlorite, the process relies on a robust and widely available supply base, minimizing the risk of raw material shortages. The simplified operational workflow also reduces the likelihood of batch delays caused by complex multi-step syntheses, ensuring more predictable delivery schedules for customers. This reliability is critical for maintaining continuous production of the final drug product and meeting the demands of the global pharmaceutical market.
- Scalability and Environmental Compliance: The process is inherently safer and more environmentally friendly, aligning with increasingly stringent global regulations on industrial emissions and waste disposal. The absence of noxious odors and toxic gases facilitates easier permitting and community relations for manufacturing sites. Moreover, the scalability of the Grignard-SO2 insertion chemistry has been demonstrated to be robust, allowing for seamless transition from pilot plant to multi-ton commercial production without significant re-engineering of the process equipment.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this novel synthesis route. These insights are derived directly from the experimental data and comparative analysis provided in the patent documentation, offering clarity on the practical implications of adopting this technology for large-scale production.
Q: How does the new Grignard-SO2 route improve safety compared to traditional methods?
A: The novel process eliminates the use of highly toxic benzyl mercaptan and hazardous chlorine gas required in prior palladium-catalyzed routes, significantly reducing occupational health risks and environmental pollution.
Q: What are the cost advantages of avoiding palladium catalysts in Tenapanor synthesis?
A: By replacing expensive palladium catalysts with cost-effective magnesium-based reagents, the process drastically reduces raw material costs and removes the need for complex heavy metal removal steps, streamlining purification.
Q: Is this synthesis method suitable for large-scale industrial production?
A: Yes, the one-pot procedure simplifies operations by avoiding intermediate isolation, shortens reaction cycles, and utilizes common reagents like sodium hypochlorite, making it highly scalable for commercial manufacturing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Tenapanor Intermediate Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of the Grignard-mediated sulfonation technology for the efficient production of Tenapanor intermediates. As a dedicated CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that our clients receive a consistent and high-quality supply of critical materials. Our state-of-the-art facilities are equipped to handle sensitive organometallic reactions and gaseous reagents like sulfur dioxide with the highest standards of safety and precision. We maintain stringent purity specifications and operate rigorous QC labs to guarantee that every batch meets the exacting requirements of the pharmaceutical industry.
We invite you to collaborate with us to leverage this advanced manufacturing capability for your supply chain needs. Our technical team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements, demonstrating how this route can optimize your COGS. Please contact our technical procurement team today to request specific COA data and route feasibility assessments, and let us help you secure a sustainable and competitive supply of high-purity Tenapanor intermediates.
