Advanced Synthesis of 2-Pyrone Compounds via Lignin Model [3+3] Cycloaddition for Commercial Scale-Up
Introduction to Sustainable Heterocycle Synthesis
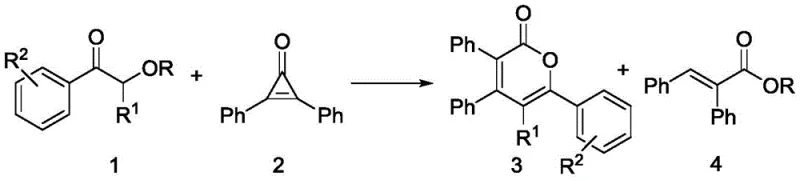
The chemical industry is currently witnessing a paradigm shift towards utilizing renewable biomass feedstocks for the production of high-value pharmaceutical intermediates. Patent CN113214204A introduces a groundbreaking methodology for the synthesis of 2-pyrone compounds and alpha,beta-unsaturated chain ester compounds, leveraging the abundant structural motifs found in lignin. This technology represents a significant advancement in organic synthesis, specifically targeting the valorization of lignin model compounds through a highly efficient [3+3] cycloaddition reaction. By employing potassium tert-butoxide as a cost-effective catalyst, this process transforms simple lignin derivatives into complex heterocyclic scaffolds that are critical for drug discovery and fine chemical manufacturing. The ability to selectively cleave the recalcitrant beta-O-4 bonds under mild conditions opens new avenues for sustainable chemistry, offering a reliable pharma intermediate supplier pathway that aligns with green chemistry principles while maintaining rigorous purity standards required for active pharmaceutical ingredient (API) production.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the depolymerization of lignin to access aromatic building blocks has been plagued by severe thermodynamic and kinetic barriers. Conventional industrial methods typically rely on base-catalyzed hydrolysis using strong bases like sodium hydroxide or potassium hydroxide at extremely elevated temperatures, often reaching approximately 200°C. These harsh conditions are not only energy-intensive but also lack selectivity, leading to the degradation of valuable structural features. The reported yields for such traditional processes are notoriously low, generally capping at merely 20-23%, and the product distribution is limited to simple monomers such as catechol, guaiacol, and eugenol. Furthermore, the extreme thermal stress often results in the formation of complex, intractable tars and char, complicating downstream purification and significantly increasing the environmental footprint of the manufacturing process. This inefficiency creates a bottleneck for supply chain heads who require consistent, high-volume access to functionalized aromatic precursors without the burden of excessive waste treatment.
The Novel Approach
In stark contrast, the methodology disclosed in the patent utilizes a sophisticated [3+3] cycloaddition strategy that operates under remarkably mild conditions, typically between 30°C and 80°C. This approach fundamentally changes the economic equation by replacing energy-intensive thermal cracking with a catalytic bond-forming event. The use of potassium tert-butoxide allows for the selective activation of the lignin model compound, facilitating a clean transformation into 2-pyrone derivatives with isolated yields frequently exceeding 90%, and in optimized cases reaching up to 99%. This dramatic improvement in efficiency means that for every kilogram of starting material, the output of valuable product is exponentially higher than traditional methods. Additionally, the reaction demonstrates excellent functional group tolerance, accommodating various substituents such as halogens, alkoxy groups, and alkyl chains without the need for protecting groups, thereby streamlining the synthetic route and reducing the overall step count for complex molecule assembly.
Mechanistic Insights into KOtBu-Catalyzed Cycloaddition
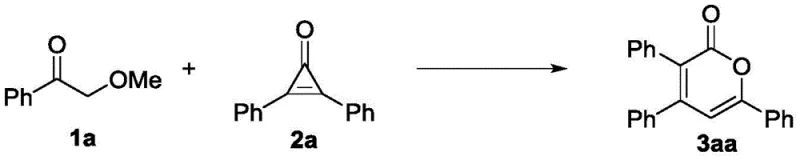
The core of this technological breakthrough lies in the precise mechanistic pathway facilitated by the potassium tert-butoxide catalyst. The reaction initiates with the deprotonation of the alpha-carbon of the lignin model ketone, generating a reactive enolate species. This nucleophile then engages in a selective cleavage of the beta-O-4 ether linkage, a bond that is notoriously difficult to break under mild conditions. The resulting phenoxide and enolate fragments are perfectly poised to undergo a cascade reaction with the highly strained cyclopropenone electrophile. The ring strain of the cyclopropenone drives the [3+3] cycloaddition, leading to the formation of the six-membered 2-pyrone ring system. This mechanism is distinct from radical-based depolymerization methods, offering superior control over the regioselectivity and stereochemistry of the final product. The absence of transition metals in the catalytic cycle is particularly noteworthy for R&D directors, as it eliminates the risk of heavy metal contamination in the final API, simplifying the regulatory approval process and reducing the need for expensive metal scavenging resins during purification.
Furthermore, the reaction exhibits a unique divergence based on the nature of the leaving group in the lignin model. When the R group is an alkyl moiety, the reaction proceeds exclusively to form the 2-pyrone compound. However, when R is an aryl group, the residual aryloxide fragment acts as a nucleophile that intercepts the reaction intermediate, leading to the concurrent formation of alpha,beta-unsaturated chain ester compounds. This dual-product capability effectively doubles the atom economy of the process, converting what would traditionally be waste phenolic by-products into commercially valuable ester intermediates. The solvent choice plays a critical role in stabilizing the ionic intermediates; screening data indicates that 1,2-dimethoxyethane (DME) provides the optimal dielectric environment, outperforming other polar aprotic solvents like DMF or dioxane, likely due to its ability to effectively solvate the potassium cation while maintaining the reactivity of the anionic species.
How to Synthesize 2-Pyrone Derivatives Efficiently
Implementing this synthesis route in a laboratory or pilot plant setting requires strict adherence to the optimized parameters defined in the patent to ensure maximum yield and reproducibility. The process is designed to be operationally simple, avoiding the need for specialized high-pressure equipment or cryogenic conditions. The following guide outlines the standardized protocol derived from the exemplary embodiments, ensuring that technical teams can replicate the high success rates reported in the intellectual property documentation. For detailed procedural nuances regarding specific substrate variations, refer to the comprehensive experimental section of the source patent.
- Combine lignin model compound 1, cyclopropenone compound 2, and 10 mol% potassium tert-butoxide (KOtBu) in an aprotic polar solvent such as DME under an inert argon atmosphere.
- Seal the reaction vessel and heat the mixture in an oil bath at a temperature ranging from 30°C to 80°C, stirring until the reaction is complete (typically 12 to 36 hours).
- Quench the reaction with water, extract the organic layer with ethyl ether, remove the solvent under reduced pressure, and purify the crude residue via silica gel column chromatography.
Commercial Advantages for Procurement and Supply Chain Teams
From a procurement and supply chain perspective, this technology offers transformative benefits that directly impact the bottom line and operational resilience. The shift from high-temperature hydrolysis to mild catalytic cycloaddition represents a fundamental reduction in energy consumption, which translates to lower utility costs per kilogram of product. Moreover, the use of commodity chemicals like potassium tert-butoxide and dimethoxyethane ensures that the raw material supply chain is robust and less susceptible to the volatility associated with exotic catalysts or rare earth metals. The high selectivity of the reaction minimizes the formation of side products, drastically simplifying the purification workflow and reducing the volume of organic solvents required for chromatography, which is a major cost driver in fine chemical manufacturing.
- Cost Reduction in Manufacturing: The elimination of extreme thermal conditions (dropping from 200°C to as low as 30°C) significantly lowers the energy burden on the production facility, allowing for the use of standard glass-lined reactors rather than specialized high-pressure vessels. The catalyst loading is minimal at 10 mol%, and the catalyst itself is inexpensive and widely available globally, removing a potential bottleneck in raw material sourcing. Additionally, the high yields (up to 99%) mean that less starting material is wasted, maximizing the return on investment for every batch produced and reducing the cost of goods sold (COGS) for the final pharmaceutical intermediate.
- Enhanced Supply Chain Reliability: By utilizing lignin model compounds derived from biomass, this process taps into a renewable and virtually inexhaustible feedstock, insulating the supply chain from the fluctuations of petrochemical markets. The simplicity of the reaction setup, which requires only an inert atmosphere and moderate heating, makes it highly adaptable to existing manufacturing infrastructure without the need for capital-intensive retrofitting. This flexibility ensures that production can be scaled up rapidly to meet market demand, providing a reliable pharma intermediate supplier capability that can respond swiftly to the needs of downstream API manufacturers.
- Scalability and Environmental Compliance: The mild reaction conditions inherently improve process safety, reducing the risks associated with high-temperature operations and making the scale-up from gram to ton scale more predictable and manageable. The reduction in waste generation, coupled with the potential to valorize both the pyrone and ester co-products, aligns with increasingly stringent environmental regulations and corporate sustainability goals. This green chemistry profile not only mitigates regulatory risk but also enhances the brand value of the end-product, appealing to eco-conscious stakeholders in the global pharmaceutical and agrochemical sectors.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis method. These answers are derived directly from the experimental data and technical specifications provided in the patent literature, ensuring accuracy and relevance for decision-makers evaluating this technology for adoption. Understanding these details is crucial for assessing the feasibility of integrating this route into your current production portfolio.
Q: How does this method compare to traditional lignin depolymerization?
A: Traditional base-catalyzed hydrolysis typically requires harsh conditions around 200°C and yields only 20-23% of simple phenols. This novel [3+3] cycloaddition method operates at mild temperatures (30-80°C) and achieves yields up to 99% for complex 2-pyrone scaffolds.
Q: Does this process generate co-products?
A: Yes, when the starting lignin model compound contains an aryl group at the R position, the reaction selectively cleaves the beta-O-4 bond to simultaneously generate valuable alpha,beta-unsaturated chain ester compounds alongside the 2-pyrone product.
Q: What is the optimal solvent system for this transformation?
A: While various aprotic polar solvents like dioxane, THF, and DMA were screened, 1,2-dimethoxyethane (DME) was identified as the superior solvent, providing the highest isolated yields (up to 98%) for the target 2-pyrone compounds.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 2-Pyrone Compound Supplier
At NINGBO INNO PHARMCHEM, we recognize the immense potential of this lignin-valorization technology to redefine the production of heterocyclic building blocks. As a premier CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory discovery to industrial reality is seamless. Our state-of-the-art facilities are equipped to handle the specific requirements of base-catalyzed reactions, and our rigorous QC labs enforce stringent purity specifications to guarantee that every batch of 2-pyrone intermediate meets the exacting standards of the global pharmaceutical industry. We are committed to delivering high-purity OLED material and pharma intermediates that drive innovation in your drug development pipelines.
We invite you to collaborate with us to leverage this cutting-edge synthesis method for your next project. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements, demonstrating how this efficient route can optimize your budget. Please contact us to request specific COA data and route feasibility assessments, and let us help you secure a sustainable, cost-effective supply of these critical chemical intermediates.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
