Advanced Synthesis of 2-Amino-5-isoindoline-1,3-dione Methyl 1,3,4-thiadiazole for Pharma Intermediates
Advanced Synthesis of 2-Amino-5-isoindoline-1,3-dione Methyl 1,3,4-thiadiazole for Pharma Intermediates
The pharmaceutical and agrochemical industries are constantly seeking robust, scalable pathways for constructing complex heterocyclic scaffolds that serve as critical building blocks for next-generation therapeutics. Patent CN102757427A introduces a highly efficient methodology for the preparation of 2-amino-5-isoindoline-1,3-dione methyl 1,3,4-thiadiazole, a compound exhibiting significant potential as a bioactive intermediate. This specific molecular architecture integrates the pharmacophore of the 1,3,4-thiadiazole ring with an isoindoline-dione moiety, creating a unique structural motif that has been associated with diverse biological activities ranging from antimicrobial to anticonvulsant properties. The disclosed technology represents a significant leap forward in process chemistry, offering a streamlined route that bypasses the cumbersome multi-step sequences often associated with traditional heterocycle functionalization. By leveraging a direct cyclodehydration strategy mediated by phosphorus oxychloride, this innovation addresses the critical industry demand for reliable pharmaceutical intermediate supplier capabilities that can deliver high-purity materials without compromising on throughput or environmental safety standards.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of substituted 1,3,4-thiadiazoles has been plagued by significant operational challenges that hinder commercial viability and cost-effectiveness in large-scale manufacturing environments. Traditional routes often rely on the use of unstable hydrazine derivatives or require harsh acidic conditions that lead to extensive decomposition of sensitive functional groups, resulting in poor mass balance and difficult purification protocols. Furthermore, conventional methodologies frequently necessitate the use of expensive transition metal catalysts or protecting group strategies that add unnecessary synthetic steps, thereby inflating the overall cost of goods sold (COGS) and extending the production lead time. These legacy processes often generate substantial quantities of hazardous waste streams, complicating regulatory compliance and increasing the burden on wastewater treatment facilities. For procurement managers and supply chain heads, these inefficiencies translate into volatile pricing structures and unreliable delivery schedules, making it difficult to secure a consistent supply of high-purity OLED material or pharmaceutical precursors needed for continuous production lines.
The Novel Approach
In stark contrast to these archaic techniques, the novel approach detailed in the patent utilizes a convergent synthesis strategy that maximizes atom economy and minimizes waste generation through a cleverly designed one-pot cyclization sequence. The core innovation lies in the direct condensation of phthaloylglycine with thiosemicarbazide in the presence of phosphorus oxychloride, which acts simultaneously as a solvent and a powerful dehydrating agent to drive the formation of the thiadiazole ring. This method eliminates the need for isolated intermediates prior to the cyclization step, significantly reducing the handling time and exposure to potentially hazardous reagents. As illustrated in the reaction scheme below, the process flows seamlessly from the initial acylation to the final ring closure, demonstrating a level of operational simplicity that is rare in complex heterocyclic chemistry.
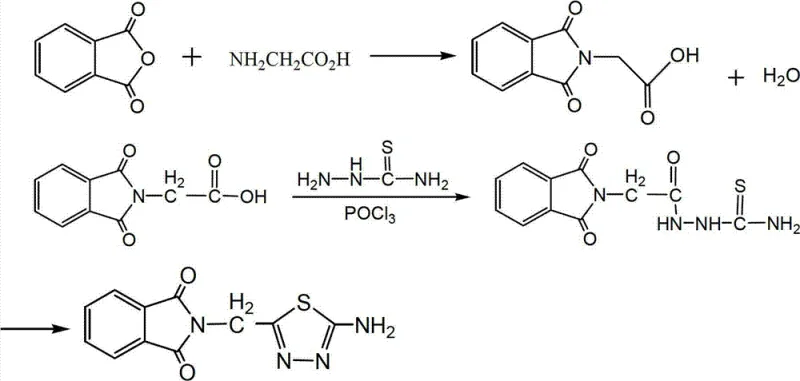
The implementation of this novel route allows for precise control over reaction parameters, such as temperature and stoichiometry, ensuring that the formation of unwanted regioisomers or polymeric by-products is kept to an absolute minimum. This technological advancement not only enhances the overall yield, which has been reported to exceed 80.6% in optimized embodiments, but also simplifies the downstream processing requirements, making it an ideal candidate for cost reduction in electronic chemical manufacturing and fine chemical production sectors.
Mechanistic Insights into POCl3-Mediated Cyclodehydration
The mechanistic pathway underpinning this synthesis is a fascinating example of electrophilic activation followed by nucleophilic attack and subsequent dehydration, driven by the strong affinity of phosphorus oxychloride for oxygen atoms. In the initial phase, the carbonyl oxygen of the phthaloylglycine intermediate coordinates with the phosphorus center of POCl3, generating a highly reactive imidoyl chloride species that is primed for nucleophilic assault. The amino group of the thiosemicarbazide then attacks this activated carbon center, forming a transient tetrahedral intermediate that rapidly collapses to expel a phosphate species and establish the new carbon-nitrogen bond essential for the heterocyclic framework. This step is critical for R&D directors focusing on purity and impurity profiles, as the kinetics of this transformation dictate the ratio of the desired thiadiazole product versus potential open-chain urea by-products. The subsequent heating to 110°C facilitates the intramolecular cyclization and the elimination of water, effectively aromatizing the 1,3,4-thiadiazole ring and locking the molecule into its thermodynamically stable conformation.
From an impurity control perspective, the protocol incorporates a sophisticated workup procedure that leverages pH-dependent solubility differences to isolate the target compound with exceptional fidelity. After the reaction reaches completion, the mixture is carefully quenched and the pH is adjusted to 8 using a sodium hydroxide solution, a step that neutralizes residual acidic species and precipitates the product while leaving soluble impurities in the aqueous phase. The final recrystallization from absolute ethanol serves as a polishing step, removing trace organic contaminants and ensuring that the final solid meets the stringent purity specifications required for clinical trial materials. This rigorous attention to detail in the purification stage underscores the process's suitability for the commercial scale-up of complex polymer additives and high-value active pharmaceutical ingredients where trace metal or organic impurities can be detrimental to downstream biological assays.
How to Synthesize 2-Amino-5-isoindoline-1,3-dione Methyl 1,3,4-thiadiazole Efficiently
Executing this synthesis requires careful attention to thermal management and reagent addition rates to maintain the delicate balance between reaction velocity and selectivity. The process begins with the preparation of the key precursor, phthaloylglycine, which is synthesized by heating glycine and phthalic anhydride to 140°C, a step that must be monitored closely to prevent charring or degradation of the amino acid backbone. Once the precursor is secured, the cyclization reaction is initiated by suspending the phthaloylglycine and thiosemicarbazide in phosphorus oxychloride, followed by a controlled reflux period that allows the system to reach equilibrium before the hydrolysis step is introduced. The detailed standardized synthesis steps, including exact molar ratios and specific temperature ramping profiles, are outlined in the guide below to ensure reproducibility across different manufacturing sites.
- Preparation of Phthaloylglycine: React glycine with phthalic anhydride at 140°C for 30 minutes, followed by hot water precipitation and ethanol recrystallization.
- Cyclization Reaction: Reflux phthaloylglycine with thiosemicarbazide and phosphorus oxychloride (POCl3) at 80°C, then hydrolyze and heat to 110°C.
- Purification: Adjust pH to 8 using sodium hydroxide, filter the precipitate, and recrystallize from absolute ethanol to obtain the white solid product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain executives, the adoption of this patented methodology offers a compelling value proposition that extends far beyond simple chemical transformation, addressing fundamental pain points related to cost stability and logistical reliability. By utilizing commodity-grade starting materials such as phthalic anhydride and glycine, which are produced on a massive global scale, the process insulates the supply chain from the volatility often associated with exotic or scarce reagents. This reliance on abundant feedstocks ensures that production schedules can be maintained without interruption, even during periods of global raw material shortages, thereby enhancing supply chain reliability and securing long-term contracts with key stakeholders. Furthermore, the elimination of expensive transition metal catalysts removes the need for costly and time-consuming heavy metal scavenging steps, which traditionally add significant overhead to the manufacturing budget and extend the batch cycle time.
- Cost Reduction in Manufacturing: The streamlined nature of this synthesis directly translates to substantial cost savings by reducing the total number of unit operations required to produce the final intermediate. By combining the acylation and cyclization steps into a cohesive workflow, the process minimizes solvent consumption, energy usage for heating and cooling, and labor hours associated with intermediate isolation and drying. The high yield reported in the patent data means that less raw material is wasted per kilogram of finished product, optimizing the mass efficiency of the plant and lowering the effective cost per mole of the active ingredient. Additionally, the use of ethanol for recrystallization is economically favorable compared to more exotic solvent systems, further driving down the operational expenditure associated with solvent recovery and disposal.
- Enhanced Supply Chain Reliability: The robustness of this chemical route provides a significant buffer against supply chain disruptions, as the reagents involved are not subject to the same geopolitical or regulatory constraints as specialized catalysts. The simplicity of the equipment requirements—standard glass-lined reactors capable of handling corrosive acids—means that the process can be easily transferred between different manufacturing facilities without the need for specialized hardware investments. This flexibility allows for diversified sourcing strategies, enabling companies to qualify multiple production sites and mitigate the risk of single-point failures. Consequently, lead times for high-purity pharmaceutical intermediates can be significantly reduced, ensuring that downstream drug development programs remain on schedule and that commercial launch dates are met without delay.
- Scalability and Environmental Compliance: From an environmental, health, and safety (EHS) perspective, this process demonstrates a marked improvement over traditional methods by generating fewer by-products and utilizing a closed-loop system for the handling of phosphorus oxychloride. The ability to neutralize acidic waste streams effectively and recover solvents like ethanol aligns with modern green chemistry principles, reducing the environmental footprint of the manufacturing operation. This compliance with stringent environmental regulations facilitates smoother permitting processes and reduces the liability associated with hazardous waste disposal. Moreover, the process has been demonstrated to scale effectively from laboratory benchtop quantities to multi-kilogram batches without loss of yield or purity, proving its viability for commercial scale-up of complex fine chemicals and ensuring a steady flow of material for industrial applications.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis route, providing clarity for technical teams evaluating the feasibility of integrating this intermediate into their existing portfolios. These answers are derived directly from the experimental data and process descriptions found within the patent literature, ensuring that the information provided is accurate and actionable for decision-makers. Understanding the nuances of this technology is essential for optimizing procurement strategies and aligning R&D efforts with the most efficient manufacturing pathways available in the current market landscape.
Q: What is the expected yield for this thiadiazole synthesis?
A: According to patent CN102757427A, the optimized process achieves yields exceeding 80.6%, with specific embodiments demonstrating yields up to 85%.
Q: Are the raw materials for this process readily available?
A: Yes, the synthesis utilizes commodity chemicals such as phthalic anhydride, glycine, and thiosemicarbazide, ensuring robust supply chain continuity.
Q: How is product purity ensured in this method?
A: The process includes a specific pH adjustment step followed by recrystallization from absolute ethanol, which effectively removes by-products and ensures high purity specifications.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 2-Amino-5-isoindoline-1,3-dione Methyl 1,3,4-thiadiazole Supplier
At NINGBO INNO PHARMCHEM, we recognize that the successful commercialization of advanced therapeutic agents depends heavily on the availability of high-quality, consistently manufactured intermediates. Our team of expert process chemists possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your project can transition smoothly from clinical development to full-scale market supply. We are committed to maintaining stringent purity specifications and operating rigorous QC labs that utilize state-of-the-art analytical instrumentation to verify the identity and quality of every batch we produce. Our dedication to technical excellence means that we can adapt the patented synthesis of 2-amino-5-isoindoline-1,3-dione methyl 1,3,4-thiadiazole to meet your specific volume requirements while adhering to the highest standards of safety and regulatory compliance.
We invite you to engage with our technical procurement team to discuss how our manufacturing capabilities can support your specific project needs and drive value for your organization. By requesting a Customized Cost-Saving Analysis, you can gain deeper insights into how our optimized processes can reduce your overall COGS and improve your margin structure. We encourage you to contact us today to索取 specific COA data and route feasibility assessments, allowing us to demonstrate our commitment to being your trusted partner in the complex world of fine chemical synthesis and pharmaceutical intermediate supply.
