Revolutionizing Heterocycle Synthesis: A Green, Metal-Free Alkenylation Strategy for Commercial Scale-Up
Revolutionizing Heterocycle Synthesis: A Green, Metal-Free Alkenylation Strategy for Commercial Scale-Up
The landscape of fine chemical synthesis is undergoing a paradigm shift towards sustainability and operational simplicity, driven by the urgent need to reduce environmental footprints and manufacturing costs in the pharmaceutical sector. Patent CN112300085A introduces a groundbreaking alkenylation method for methyl heterocyclic compounds that fundamentally alters the traditional approach to constructing C-C double bonds on heteroaromatic rings. This innovation replaces the conventional reliance on unstable aldehydes or expensive transition metal catalysts with a robust, base-mediated aerobic oxidative coupling strategy. By utilizing readily available alcohols as alkenylating reagents and molecular oxygen from the air as the oxidant, this technology offers a pathway to synthesize high-purity alkenyl heterocyclic compounds with exceptional atom economy. For R&D directors and process chemists, this represents a significant opportunity to streamline synthetic routes for complex API intermediates, eliminating the bottlenecks associated with metal removal and hazardous reagent handling.
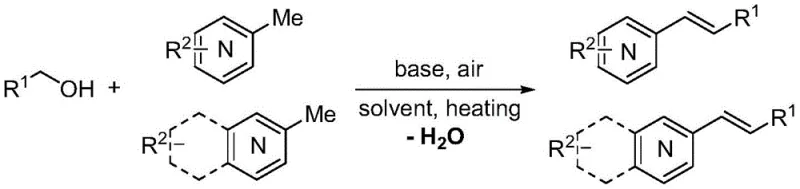
The core breakthrough lies in the direct C-olefination of the methyl position on heterocycles such as quinoxaline, quinoline, and benzothiazole without the participation of any transition metal catalyst. In traditional methodologies, introducing an alkenyl group often necessitates the use of precious metal complexes like palladium or ruthenium, which not only inflate raw material costs but also introduce stringent regulatory hurdles regarding residual metal limits in drug substances. Furthermore, the prior art frequently depends on aldehyde condensation reactions, which suffer from the inherent instability, toxicity, and strong odor of aldehyde reagents. The method disclosed in CN112300085A circumvents these issues entirely by employing stable, low-toxicity alcohols and common inorganic bases like potassium hydroxide or cesium hydroxide. This shift not only enhances the safety profile of the manufacturing process but also simplifies the downstream purification workflow, as the only byproduct generated is water, and the inorganic base can be easily removed via aqueous washes.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the alkenylation of methyl heterocycles has been dominated by two primary strategies, both of which present substantial drawbacks for large-scale industrial application. The first and most common approach involves the condensation of heterocyclic methyl compounds with aldehydes. While chemically feasible, this route is plagued by the use of aldehyde reagents that are often highly active, unstable upon storage, and possess heavy, unpleasant odors that complicate plant operations. Moreover, these condensation reactions typically require strong alkaline conditions or specific dehydration reagents, leading to the generation of significant amounts of chemical waste that must be treated before disposal. The second conventional method utilizes transition metal complexes to catalyze the coupling of alcohols with heterocycles. Although effective, these catalytic systems are expensive to procure, sensitive to air and moisture requiring inert gas protection, and complicated by the need for specialized ligands to stabilize the metal center. Perhaps most critically for the pharmaceutical industry, these methods inevitably lead to transition metal residues in the final product, necessitating costly and time-consuming purification steps to meet strict regulatory specifications for heavy metals.
The Novel Approach
In stark contrast to these legacy methods, the novel approach detailed in the patent leverages a transition-metal-free mechanism driven by simple inorganic bases and atmospheric oxygen. This method transforms the alkenylation process into a green, efficient, and operationally simple procedure. By using alcohols as the carbon source, the process accesses stable, cheap, and widely available starting materials that do not suffer from the volatility or toxicity issues of aldehydes. The reaction proceeds under air atmosphere at temperatures ranging from 100°C to 140°C, eliminating the need for expensive inert gas lines or specialized pressure equipment. The use of water-soluble inorganic bases such as KOH or CsOH ensures that the catalyst system is inexpensive and easy to handle. Crucially, the absence of transition metals means the resulting alkenyl heterocyclic compounds are inherently free from metal contamination, drastically reducing the burden on quality control and purification teams. This approach effectively decouples high-yield synthesis from high environmental impact, offering a sustainable solution for the production of valuable fine chemical intermediates.
Mechanistic Insights into Base-Mediated Aerobic Oxidative Coupling
The mechanistic underpinning of this transformation involves a sophisticated interplay between base-mediated deprotonation and aerobic oxidation, facilitating the direct formation of a carbon-carbon double bond. Initially, the strong inorganic base abstracts a proton from the methyl group of the heterocyclic substrate, generating a reactive carbanion or radical species stabilized by the adjacent electron-deficient nitrogen atoms in the heterocycle. Simultaneously, the alcohol reagent undergoes oxidation, likely facilitated by the basic environment and molecular oxygen, to form an aldehyde intermediate in situ. This transient aldehyde then undergoes a condensation reaction with the activated methyl heterocycle, followed by dehydration to yield the final alkenyl product. The use of air as the terminal oxidant is particularly elegant, as it regenerates the active species and drives the equilibrium forward by producing water as the sole stoichiometric byproduct. This mechanism avoids the formation of complex organometallic intermediates, thereby sidestepping the potential for side reactions associated with metal coordination and ensuring a cleaner reaction profile.
From an impurity control perspective, this mechanism offers distinct advantages for maintaining high product purity. Since no transition metals are involved, there is no risk of metal-catalyzed decomposition pathways or the formation of metal-complexed impurities that are notoriously difficult to separate. The reaction conditions are mild enough to preserve sensitive functional groups on the aromatic rings, such as halogens or methoxy groups, which might otherwise be compromised under harsher metal-catalyzed conditions. Furthermore, the byproduct profile is exceptionally clean, consisting primarily of water and unreacted starting materials that can be easily separated. The inorganic base used in the reaction is water-soluble, allowing for its complete removal through a simple aqueous workup, leaving the organic phase containing the product largely free of inorganic salts. This streamlined purification process not only improves the overall yield by minimizing product loss during chromatography or recrystallization but also ensures that the final material meets the rigorous purity standards required for pharmaceutical intermediates.
How to Synthesize Alkenyl Heterocyclic Compounds Efficiently
The practical implementation of this synthesis route is designed for ease of execution in both laboratory and pilot plant settings. The protocol typically involves charging a reaction vessel with the methyl heterocycle, the chosen alcohol reagent, and a suitable organic solvent such as toluene or xylene. A specific amount of inorganic base, generally between 40 to 100 mol%, is added to initiate the reaction. The mixture is then heated to temperatures between 120°C and 140°C under an open air or oxygen atmosphere for a duration of 18 to 36 hours. This straightforward procedure eliminates the need for gloveboxes or Schlenk lines, making it accessible to facilities with standard chemical processing capabilities. The detailed standardized synthesis steps for specific derivatives are outlined below to guide process development teams in replicating these results.
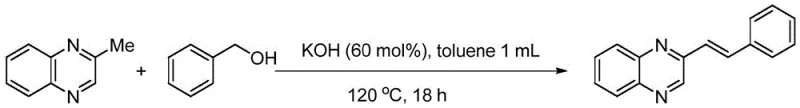
- Combine the methyl heterocyclic compound (e.g., 2-methylquinoxaline) and the alcohol reagent (e.g., benzyl alcohol) in an organic solvent such as toluene or xylene.
- Add a water-soluble inorganic base, preferably KOH or CsOH, in an amount ranging from 40 to 100 mol% relative to the substrate.
- Seal the reaction vessel and heat the mixture to a temperature between 100°C and 140°C under an air atmosphere for 18 to 36 hours to facilitate oxidative condensation.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this technology translates into tangible strategic benefits that extend far beyond the laboratory bench. The elimination of transition metal catalysts removes a major cost driver from the bill of materials, as precious metals like palladium and their specialized ligands are subject to significant price volatility and supply constraints. Additionally, the switch to alcohols as reagents leverages a commodity chemical market that is stable and abundant, reducing the risk of raw material shortages that often plague specialty aldehyde suppliers. The simplified operational requirements, such as the ability to run reactions under air rather than inert gas, lower the capital expenditure needed for specialized reactor infrastructure and reduce utility costs associated with nitrogen or argon consumption. These factors collectively contribute to a more resilient and cost-effective supply chain for critical heterocyclic intermediates.
- Cost Reduction in Manufacturing: The economic impact of this process is profound due to the drastic simplification of the input materials and downstream processing. By removing the need for expensive transition metal catalysts and ligands, the direct material costs are significantly lowered. Furthermore, the absence of metal residues eliminates the need for costly scavenging resins or additional purification cycles dedicated to heavy metal removal, which are standard expenses in traditional metal-catalyzed processes. The use of cheap, commodity-grade alcohols instead of specialized aldehydes further drives down the cost of goods sold. The simplified workup, relying on aqueous extraction to remove the inorganic base, reduces solvent consumption and waste disposal fees, leading to substantial overall cost savings in the manufacturing of complex alkenyl heterocycles.
- Enhanced Supply Chain Reliability: Supply chain stability is greatly enhanced by the reliance on widely available and stable reagents. Alcohols are produced on a massive industrial scale for various sectors, ensuring a consistent and reliable supply compared to niche aldehyde reagents which may have limited suppliers. The robustness of the reaction conditions, which do not require strict exclusion of moisture or oxygen, reduces the risk of batch failures due to environmental fluctuations or equipment leaks. This operational resilience ensures higher on-time delivery rates and consistent product availability for downstream customers. Moreover, the reduced dependency on imported precious metals mitigates geopolitical supply risks, securing the continuity of production for critical pharmaceutical intermediates.
- Scalability and Environmental Compliance: Scaling this process to commercial volumes is straightforward due to the benign nature of the reagents and the simplicity of the reaction engineering. The use of air as an oxidant avoids the safety hazards associated with handling high-pressure pure oxygen or hazardous chemical oxidants, facilitating safer scale-up in large reactors. From an environmental compliance standpoint, the process aligns perfectly with green chemistry principles by generating water as the only byproduct and avoiding toxic heavy metal waste streams. This significantly reduces the environmental burden and simplifies the permitting process for manufacturing facilities. The ease of waste treatment and the high atom economy of the reaction make it an ideal candidate for sustainable manufacturing practices, helping companies meet increasingly stringent environmental regulations and corporate sustainability goals.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this alkenylation technology. These answers are derived directly from the experimental data and technical specifications provided in the patent documentation, ensuring accuracy and relevance for process evaluation. Understanding these details is crucial for assessing the feasibility of integrating this method into existing production workflows.
Q: Does this alkenylation method leave transition metal residues in the final product?
A: No, the method described in patent CN112300085A specifically eliminates the need for transition metal catalysts and ligands. It relies on common water-soluble inorganic bases like KOH or CsOH, ensuring the final alkenyl heterocyclic compounds are free from heavy metal contamination, which is critical for pharmaceutical applications.
Q: What oxidant is required for this C-olefination reaction?
A: The process utilizes molecular oxygen from the air as the terminal oxidant. This eliminates the need for expensive or hazardous stoichiometric oxidants and inert gas protection, significantly simplifying the operational requirements and reducing safety risks associated with high-pressure oxygen systems.
Q: Can this method be applied to diverse heterocyclic scaffolds beyond quinoxaline?
A: Yes, the patent demonstrates broad substrate scope. The methodology is effective for various heterocyclic systems including quinoline, benzothiazole, pyrimidine, and pyrazine. It also tolerates a wide range of functional groups on the alcohol component, such as halogens, alkyl groups, and alkoxy groups.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Alkenyl Heterocyclic Compound Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of the transition-metal-free alkenylation technology described in CN112300085A for the production of high-value pharmaceutical intermediates. As a leading CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that innovative laboratory methods are successfully translated into robust industrial processes. Our state-of-the-art facilities are equipped to handle the specific requirements of this aerobic oxidative coupling, including high-temperature reactors capable of operating safely under air atmosphere. We maintain stringent purity specifications and operate rigorous QC labs to guarantee that every batch of alkenyl heterocyclic compound meets the highest quality standards, free from metal residues and ready for immediate use in API synthesis.
We invite global partners to leverage our technical expertise to optimize their supply chains and reduce manufacturing costs. Our team is prepared to conduct a Customized Cost-Saving Analysis tailored to your specific project needs, demonstrating how this green chemistry approach can improve your margins. We encourage you to contact our technical procurement team to request specific COA data for our catalog of heterocyclic intermediates and to discuss route feasibility assessments for your custom synthesis projects. Together, we can drive efficiency and sustainability in the next generation of chemical manufacturing.
