Optimizing Peramivir Production: A Cost-Effective and Green Synthetic Route for Global Supply Chains
The pharmaceutical industry's relentless pursuit of efficient antiviral therapies has placed significant focus on the scalable production of neuraminidase inhibitors like Peramivir. Patent CN102372657B discloses a groundbreaking synthesis method that addresses critical bottlenecks in the manufacturing of this potent anti-influenza and avian influenza virus medicine. By fundamentally re-engineering the reduction and guanidinylation steps, this technology offers a robust alternative to legacy processes, specifically targeting the elimination of precious metal catalysts and toxic heavy metal reagents. For R&D directors and procurement strategists, this patent represents a pivotal shift towards more sustainable and economically viable pharmaceutical intermediates production. The methodology leverages a sophisticated sodium borohydride and nickel chloride reduction system, effectively bypassing the reliance on prohibitively expensive platinum dioxide while maintaining high stereochemical integrity throughout the synthetic sequence.
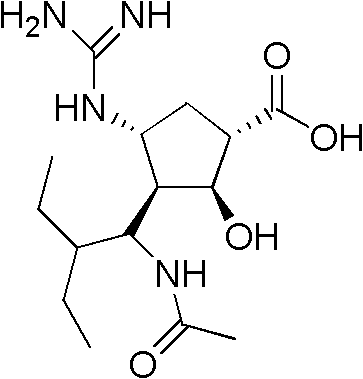
This technical breakthrough is not merely an academic exercise but a commercially driven solution designed to enhance cost reduction in API manufacturing. The process initiates with the catalytic ring opening of (1R,4S)-2-azabicyclo[2.2.1]hept-5-en-3-one, proceeding through a streamlined series of transformations that include amino protection, 1,3-dipolar cycloaddition, and a novel hydro-reduction protocol. The final stages involve a unique deprotection and guanidinylation sequence utilizing self-prepared chloroformamidine hydrochloride, which circumvents the regulatory and environmental hurdles associated with traditional mercury-based reagents. This holistic approach ensures that the commercial scale-up of complex antiviral intermediates is achieved with greater operational simplicity and reduced environmental footprint, aligning perfectly with modern green chemistry mandates.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
The historical landscape of Peramivir synthesis, particularly the routes pioneered by entities like BioCryst Pharmaceuticals, has been fraught with significant operational and economic challenges that hinder widespread adoption. As illustrated in the prior art pathways, the conventional methodology typically involves a protracted nine-step reaction sequence that inherently accumulates yield losses and increases material throughput costs. A critical pain point in these legacy processes is the dependence on platinum dioxide (PtO2) for hydrogenation steps, a catalyst that not only commands a premium price in the global market but also necessitates rigorous recovery protocols to prevent metal contamination in the final active pharmaceutical ingredient. Furthermore, the traditional guanidinylation steps often employ mercury(II) chloride, a highly toxic heavy metal reagent that generates hazardous waste streams requiring complex and costly disposal procedures, thereby violating the principles of sustainable manufacturing.
Beyond the economic and environmental burdens, the conventional routes frequently utilize hazardous solvents such as benzene and phenyl isocyanate, which pose severe health risks to operators and require specialized containment infrastructure. The hydrogenation processes in these older methods often demand high-pressure conditions, introducing significant safety risks and requiring capital-intensive equipment capable of withstanding extreme operational parameters. These factors collectively contribute to extended lead times and volatile supply chains, making the reliable sourcing of high-purity Peramivir intermediates a persistent challenge for downstream drug manufacturers. The cumulative effect of these inefficiencies is a manufacturing profile that is ill-suited for the rapid response capabilities required during influenza outbreaks, highlighting the urgent need for process intensification and hazard elimination.
The Novel Approach
In stark contrast to the cumbersome legacy pathways, the synthesis method disclosed in CN102372657B introduces a streamlined seven-step protocol that dramatically simplifies the production workflow while enhancing overall process safety. The cornerstone of this innovation is the substitution of the expensive platinum dioxide catalyst with an in-situ generated reduction system comprising sodium borohydride and nickel chloride hexahydrate. This chemical reduction strategy operates under mild atmospheric conditions, eliminating the need for high-pressure hydrogenation equipment and significantly lowering the barrier to entry for contract manufacturing organizations. By replacing the toxic mercury chloride reagent with self-prepared chloroformamidine hydrochloride, the process effectively removes heavy metal contamination risks, simplifying the purification train and ensuring the final product meets stringent pharmacopeial standards for residual metals without extensive chelation treatments.
The novel approach also optimizes the construction of the cyclopentane core through a highly stereoselective 1,3-dipolar cycloaddition, ensuring that the critical chiral centers are established early in the synthesis with high fidelity. This precision reduces the burden on downstream chiral separations, which are often the most yield-limiting steps in complex molecule synthesis. The use of readily available starting materials and common laboratory reagents further enhances the supply chain resilience, allowing for rapid scaling from kilogram to multi-ton quantities without the bottleneck of sourcing exotic catalysts. Consequently, this methodology provides a robust framework for reducing lead time for high-purity antiviral intermediates, enabling manufacturers to respond agilely to market demands while maintaining a competitive cost structure that is essential for generic drug development.
Mechanistic Insights into NaBH4-NiCl2 Reduction and Guanidinylation
The mechanistic elegance of this synthesis lies in its ability to achieve complex transformations using simple, earth-abundant reagents. The reduction of the isoxazole intermediate to the corresponding amino-alcohol is facilitated by the synergistic interaction between sodium borohydride and nickel chloride. In this system, the nickel salt is reduced by the borohydride to generate active nickel species, potentially resembling Raney nickel in situ, which acts as a powerful hydrogenation catalyst in the presence of the hydride source. This allows for the cleavage of the N-O bond in the isoxazole ring and the simultaneous reduction of the double bond without affecting other sensitive functional groups such as the ester or the carbamate protecting groups. The reaction proceeds with excellent diastereoselectivity, preserving the stereochemical information established during the earlier cycloaddition step, which is crucial for the biological activity of the final Peramivir molecule.
Following the reduction, the installation of the guanidine moiety is achieved through a nucleophilic substitution reaction using chloroformamidine hydrochloride. This reagent, prepared directly from cyanamide and hydrochloric acid as shown in the reaction scheme, serves as a potent electrophile that reacts efficiently with the free amine generated after Boc-deprotection. The mechanism avoids the formation of stable mercury-amidine complexes that plague traditional methods, thereby driving the reaction to completion with minimal side products. The subsequent hydrolysis of the methyl ester under alkaline conditions is straightforward and high-yielding, finalizing the construction of the carboxylic acid functionality essential for neuraminidase binding. This sequence demonstrates how fundamental organic transformations can be optimized to create a seamless, telescoped process that maximizes atom economy and minimizes waste generation.

How to Synthesize Peramivir Efficiently
The execution of this synthesis requires precise control over reaction parameters, particularly temperature and stoichiometry, to ensure optimal yields and purity profiles. The process begins with the acid-catalyzed methanolysis of the bicyclic ketone, followed by protection of the resulting amine with di-tert-butyl dicarbonate to prevent side reactions during the subsequent cycloaddition. The 1,3-dipolar cycloaddition with 2-ethylbutyraldehyde oxime must be conducted at low temperatures to control the formation of the nitrile oxide intermediate and ensure the correct regio- and stereochemistry of the isoxazole adduct. Detailed standard operating procedures for each step, including workup and purification protocols, are critical for reproducibility.
- Perform catalytic ring opening and amino protection using HCl gas and Boc2O to form the cyclopentene intermediate.
- Execute 1,3-dipolar cycloaddition with 2-ethylbutyraldehyde oxime to construct the isoxazole ring system.
- Conduct hydro-reduction using a Sodium Borohydride and Nickel Chloride system, followed by N-acetylation.
- Complete deprotection and guanidinylation using self-prepared chloroformamidine hydrochloride, followed by ester hydrolysis.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this synthesis route translates directly into tangible strategic advantages that bolster the bottom line and mitigate operational risk. The primary value driver is the substantial reduction in raw material costs achieved by eliminating the dependency on platinum group metals, the prices of which are subject to extreme volatility and geopolitical supply constraints. By switching to a nickel-based reduction system, manufacturers can lock in lower and more predictable input costs, facilitating more accurate long-term budgeting and pricing strategies for the final API. Additionally, the removal of mercury reagents drastically reduces the cost of environmental compliance and waste disposal, as the effluent streams no longer require specialized heavy metal treatment facilities, thereby lowering the overall cost of goods sold (COGS).
- Cost Reduction in Manufacturing: The economic benefits of this process are multifaceted, stemming from both direct material savings and indirect operational efficiencies. The replacement of platinum dioxide with nickel chloride and sodium borohydride removes one of the most expensive line items in the bill of materials, while the avoidance of toxic mercury salts eliminates the need for costly hazardous waste incineration services. Furthermore, the shortened reaction sequence from nine steps to seven steps inherently improves the overall process yield, meaning less starting material is required to produce the same amount of final product, amplifying the cost savings across the entire production volume.
- Enhanced Supply Chain Reliability: Supply chain resilience is significantly improved by the use of commodity chemicals that are widely available from multiple global suppliers, reducing the risk of single-source bottlenecks. Unlike specialized catalysts that may have long lead times or limited production capacity, reagents like sodium borohydride and nickel chloride are produced at massive scales for various industries, ensuring consistent availability even during periods of high demand. This reliability allows for smoother production planning and inventory management, ensuring that critical antiviral medications can be manufactured without interruption, which is vital for public health security.
- Scalability and Environmental Compliance: The process is inherently designed for scalability, utilizing mild reaction conditions that do not require exotic high-pressure reactors or cryogenic cooling beyond standard industrial capabilities. This ease of scale-up allows manufacturers to rapidly increase production capacity to meet surges in demand, such as during flu seasons or pandemics, without significant capital expenditure. Moreover, the green chemistry profile of the route, characterized by the absence of benzene and mercury, ensures full compliance with increasingly stringent international environmental regulations, safeguarding the manufacturer's license to operate and enhancing their corporate sustainability credentials.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis technology. These insights are derived directly from the comparative data and experimental results presented in the patent documentation, providing clarity on the practical implications of adopting this novel route. Understanding these nuances is essential for technical teams evaluating the feasibility of technology transfer and for commercial teams assessing the value proposition of this manufacturing method.
Q: How does this synthesis method reduce costs compared to the BioCryst route?
A: This method significantly lowers production costs by replacing expensive platinum dioxide (PtO2) catalysts with a cost-effective sodium borohydride and nickel chloride reduction system. Furthermore, it eliminates the need for toxic mercury chloride reagents, reducing waste treatment expenses.
Q: What are the safety advantages of this new Peramivir synthesis route?
A: The process avoids high-pressure hydrogenation conditions required by traditional methods and eliminates the use of highly toxic benzene and mercury compounds, creating a safer operational environment suitable for industrial scale-up.
Q: Is this synthetic route suitable for large-scale commercial manufacturing?
A: Yes, the route is designed for industrial suitability with mild reaction conditions, fewer steps (7 steps vs 9 steps), and simplified purification processes, ensuring high scalability and supply chain reliability.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Peramivir Supplier
At NINGBO INNO PHARMCHEM, we recognize that the transition from laboratory innovation to commercial reality requires a partner with deep technical expertise and robust manufacturing capabilities. As a leading CDMO, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the theoretical benefits of this patent are fully realized in practice. Our state-of-the-art facilities are equipped to handle the specific requirements of this synthesis, including the safe handling of reactive reagents and the rigorous purification steps necessary to meet stringent purity specifications. Our dedicated QC labs employ advanced analytical techniques to monitor every stage of the process, guaranteeing that the final Peramivir intermediate consistently exceeds quality standards.
We invite forward-thinking pharmaceutical companies to collaborate with us to leverage this cost-effective and environmentally friendly synthesis route. By partnering with our technical procurement team, you can access a Customized Cost-Saving Analysis tailored to your specific volume requirements and supply chain constraints. We encourage you to contact us today to request specific COA data and route feasibility assessments, allowing you to make informed decisions that will drive efficiency and profitability in your antiviral drug portfolio. Let us help you secure a reliable supply of high-quality Peramivir intermediates that support your mission to deliver life-saving medicines to patients worldwide.
