Revolutionizing Amide Sulfone and N-Substituted Imine Production via Aqueous Protonic Acid Catalysis
Revolutionizing Amide Sulfone and N-Substituted Imine Production via Aqueous Protonic Acid Catalysis
The landscape of fine chemical manufacturing is undergoing a significant transformation driven by the urgent need for greener, more sustainable synthetic pathways, a shift perfectly exemplified by the methodologies disclosed in Chinese Patent CN100422145C. This pivotal intellectual property introduces a robust, two-stage protocol for the preparation of amide sulfones and their subsequent conversion into N-substituted imines, addressing critical pain points regarding waste generation and operational safety that have long plagued the industry. By leveraging a unique protonic acid-catalyzed condensation in aqueous media followed by a biphasic base-mediated elimination, this technology offers a compelling alternative to traditional anhydrous and acid-heavy processes. For R&D directors and process chemists, the ability to conduct these reactions in water-organic mixtures at near-room temperature represents a paradigm shift towards safer, more scalable chemistry. Furthermore, the inherent design of the process facilitates the recovery of valuable sulfinate salts, aligning perfectly with the principles of circular economy and atom economy that modern supply chains demand. This report provides a deep technical and commercial analysis of this breakthrough, highlighting its potential to redefine cost structures and supply reliability for high-value pharmaceutical intermediates.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of amide sulfones, which serve as crucial precursors for N-substituted imines, has been fraught with significant environmental and operational challenges that hinder large-scale adoption. Conventional literature and industrial practices predominantly rely on the use of formic acid or acetic acid not merely as catalysts but as bulk solvents, necessitating the use of massive molar excesses ranging from 30 to 100 times the stoichiometric requirement. This reliance creates a substantial burden on downstream processing, as the neutralization and disposal of such vast quantities of spent acid generate enormous volumes of hazardous wastewater, driving up waste treatment costs exponentially. Moreover, these traditional routes often require harsh reaction conditions, including high temperatures and strictly anhydrous environments, which increase energy consumption and pose safety risks related to solvent volatility and thermal runaway. The difficulty in isolating pure products from such acidic sludge often leads to lower overall yields and compromised purity profiles, which are unacceptable for high-grade pharmaceutical applications. Consequently, the industry has been in dire need of a methodology that decouples the reaction efficiency from the generation of toxic waste streams.
The Novel Approach
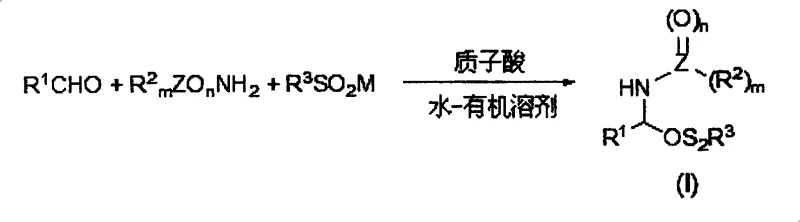
In stark contrast to the wasteful legacy methods, the technology described in CN100422145C utilizes a sophisticated yet operationally simple condensation reaction mediated by protonic acids within a water or water-organic solvent mixture. This innovative approach allows for the direct three-component coupling of an aldehyde, an amide source, and a sulfinate salt to generate the target amide sulfone structure with high efficiency and minimal environmental footprint. The use of water as a primary medium not only suppresses the volatility of organic solvents but also leverages the hydrophobic effect to drive the reaction forward, often resulting in the precipitation of the product as a solid that can be easily filtered. This eliminates the need for complex extraction procedures typically associated with homogeneous acid catalysis and significantly reduces the volume of organic waste generated. The mild reaction conditions, operable between -30°C and 100°C, further enhance the safety profile, making the process suitable for sensitive substrates that might decompose under vigorous acidic conditions. By shifting the paradigm from bulk acid usage to catalytic protonic activation in aqueous media, this method delivers a cleaner, safer, and more economically viable pathway for intermediate synthesis.
Mechanistic Insights into Aqueous Protonic Acid Catalyzed Condensation
The core of this technological advancement lies in the precise mechanistic orchestration of the condensation step, where the protonic acid acts as a potent activator for the carbonyl group of the aldehyde within the aqueous environment. In this system, the protonic acid, such as thionamic acid with a pKa between 0.5 and 4, protonates the aldehyde oxygen, increasing its electrophilicity and facilitating nucleophilic attack by the nitrogen of the amide component. Simultaneously, the sulfinate anion, stabilized in the aqueous phase, participates in the reaction sequence to form the stable C-N and C-S bonds characteristic of the amide sulfone scaffold. The presence of water is critical not just as a solvent but as a thermodynamic driver; as the reaction proceeds, the decreasing solubility of the increasingly hydrophobic amide sulfone product forces it out of the solution as a precipitate. This precipitation effectively pulls the equilibrium towards product formation, adhering to Le Chatelier's principle without the need for exotic dehydrating agents. This mechanism ensures that even with catalytic amounts of acid, high conversion rates are achieved, minimizing the formation of side products that typically arise from over-acidification or thermal degradation.
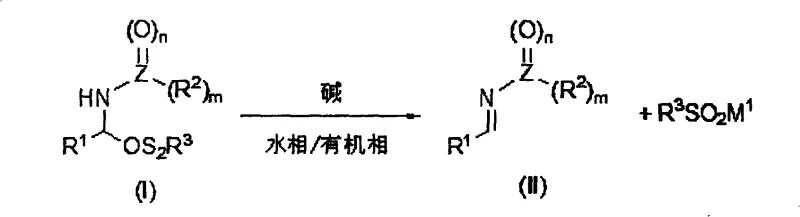
Following the formation of the amide sulfone, the subsequent transformation into the N-substituted imine relies on a clean base-mediated elimination mechanism that elegantly separates the product from the leaving group. When the isolated amide sulfone is subjected to a biphasic system comprising an organic solvent and an alkaline aqueous solution, the base abstracts the acidic proton adjacent to the sulfur-nitrogen bond. This triggers an elimination cascade where the sulfinic acid moiety is expelled as a water-soluble sulfinate salt, leaving behind the desired N-substituted imine in the organic phase. This phase separation is the linchpin of the process's purity profile; the organic layer contains the neutral imine product free from inorganic salts, while the aqueous layer retains the sulfinate byproduct. This spatial segregation allows for the straightforward recovery of the sulfinate salt, which can potentially be recycled back into the front end of the process, thereby closing the material loop. Such a mechanism not only simplifies purification but also drastically reduces the impurity load, ensuring that the final imine meets the stringent specifications required for downstream pharmaceutical synthesis.
How to Synthesize Amide Sulfone Efficiently
The practical implementation of this synthesis route involves a streamlined workflow that capitalizes on the physical properties of the reactants and products to minimize unit operations. The process begins with the preparation of a reaction mixture containing the aldehyde, the specific amide derivative, and the metal sulfinate salt dispersed in a water-alcohol co-solvent system. Upon the addition of the protonic acid catalyst and gentle heating or stirring at ambient temperature, the condensation occurs rapidly, often leading to the visible crystallization of the amide sulfone product directly from the reaction broth. This direct isolation via filtration bypasses the need for energy-intensive distillation or chromatographic purification at the intermediate stage. The second stage involves suspending this crude solid in a chlorinated or non-chlorinated organic solvent and treating it with a mild aqueous base, such as sodium carbonate or bicarbonate, to effect the elimination. The resulting biphasic mixture is simply separated, with the organic layer dried and concentrated to yield the high-purity N-substituted imine. Detailed standardized synthesis steps for specific derivatives are provided in the guide below.
- Condense aldehyde, amide, and sulfinate salt in a water-organic solvent mixture using a protonic acid catalyst at mild temperatures (-30 to 100°C) to form the amide sulfone intermediate.
- Isolate the solid amide sulfone product via filtration and washing, utilizing the low solubility of the product in the aqueous medium.
- React the isolated amide sulfone in a biphasic system with an aqueous base to eliminate sulfinic acid, yielding the N-substituted imine in the organic phase while recovering sulfinate salts in the water phase.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain leaders, the adoption of this aqueous-based synthesis technology translates into tangible strategic advantages that extend far beyond simple laboratory curiosity. The fundamental shift away from bulk organic acids and volatile solvents fundamentally alters the cost structure of manufacturing these critical intermediates, offering a pathway to significant margin improvement. By utilizing water as the primary reaction medium, the process inherently reduces the fire hazard classification of the manufacturing facility, potentially lowering insurance premiums and relaxing storage requirements for flammable inventory. Furthermore, the ability to recover and recycle the sulfinate component from the aqueous waste stream introduces a circular economy element that insulates the supply chain from fluctuations in raw material pricing. The operational simplicity of the process, which avoids cryogenic conditions and high-pressure reactors, means that production can be scaled up using existing general-purpose equipment without the need for specialized capital investment. These factors combine to create a supply proposition that is not only cost-effective but also resilient and sustainable.
- Cost Reduction in Manufacturing: The elimination of massive excesses of formic or acetic acid removes a major cost center associated with both raw material procurement and hazardous waste disposal fees. By replacing stoichiometric acid usage with catalytic protonic acid in water, the process drastically reduces the volume of chemical inputs required per kilogram of product, leading to substantial savings in material costs. Additionally, the simplified workup procedure, which relies on filtration and phase separation rather than complex distillations, lowers energy consumption and labor hours per batch. The recovery of sulfinate salts from the aqueous phase further enhances economic efficiency by reducing the net consumption of sulfur-containing reagents. Overall, the process architecture is designed to minimize variable costs, making the final intermediate more price-competitive in the global market.
- Enhanced Supply Chain Reliability: The reliance on commodity chemicals such as water, common alcohols, and widely available aldehydes ensures that the supply chain is not vulnerable to the bottlenecks often associated with specialty solvents or exotic catalysts. The mild reaction conditions reduce the risk of batch failures due to thermal excursions or equipment malfunction, thereby improving on-time delivery performance and consistency. Because the process generates less hazardous waste, regulatory compliance is streamlined, reducing the risk of production shutdowns due to environmental permitting issues. The robustness of the chemistry across a wide range of substrates means that the same production line can be flexibly adapted to manufacture different intermediates, enhancing the agility of the supply response to market demand changes.
- Scalability and Environmental Compliance: The use of water as a solvent inherently aligns with green chemistry principles, significantly reducing the emission of volatile organic compounds (VOCs) into the atmosphere. This environmental benefit simplifies the permitting process for capacity expansion and ensures long-term operational continuity in regions with strict environmental regulations. The exothermic nature of the condensation is easily managed in aqueous media due to the high heat capacity of water, allowing for safe scale-up from pilot plant to multi-ton commercial production without complex engineering controls. The solid-state isolation of the intermediate simplifies logistics, as solids are generally easier and safer to transport and store than liquid intermediates. This combination of safety, scalability, and sustainability makes the technology ideal for long-term strategic partnerships.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this amide sulfone synthesis technology, derived directly from the patent specifications and process data. Understanding these details is crucial for evaluating the feasibility of integrating this route into existing manufacturing portfolios. The answers provided reflect the specific advantages of the aqueous protonic acid system over traditional methods, focusing on yield, purity, and waste management. We encourage technical teams to review these insights to fully appreciate the operational benefits offered by this innovative approach.
Q: How does this new method improve upon traditional amide sulfone synthesis?
A: Traditional methods rely on excessive amounts (30-100 equivalents) of formic or acetic acid as both solvent and reagent, creating massive waste disposal issues. This patented method utilizes a water-organic solvent system with catalytic protonic acid, drastically reducing waste generation and eliminating the need for hazardous anhydrous conditions.
Q: Can the sulfinic acid byproduct be recovered in this process?
A: Yes, a key advantage of the elimination step is the biphasic nature of the reaction. The sulfinic acid forms a water-soluble salt in the aqueous phase, allowing for easy separation from the organic product and enabling the recovery and potential recycling of the sulfinate species.
Q: What types of substrates are compatible with this synthesis route?
A: The method demonstrates broad substrate scope, accommodating various aldehydes including alkyl, aryl, heteroaryl, and trifluoromethyl-substituted variants. It is also compatible with different amide sources (sulfonamides, phosphoramidates, carbamates) and sulfinate salts, making it highly versatile for diverse intermediate production.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Amide Sulfone Supplier
At NINGBO INNO PHARMCHEM, we recognize that the transition to greener, more efficient synthetic routes is essential for maintaining competitiveness in the modern pharmaceutical landscape. As a premier CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the theoretical benefits of patent CN100422145C are fully realized in practical, large-scale operations. Our state-of-the-art facilities are equipped to handle the specific requirements of aqueous-organic biphasic reactions, and our rigorous QC labs enforce stringent purity specifications to guarantee that every batch of amide sulfone or N-substituted imine meets the highest industry standards. We are committed to delivering not just a chemical product, but a reliable supply solution that enhances your own downstream processing efficiency.
We invite you to engage with our technical procurement team to discuss how this advanced synthesis method can be tailored to your specific project needs. By leveraging our expertise, you can obtain a Customized Cost-Saving Analysis that quantifies the potential economic benefits of switching to this aqueous-based protocol for your specific target molecules. We encourage you to request specific COA data and route feasibility assessments to validate the performance of our materials in your processes. Let us collaborate to build a more sustainable and cost-effective supply chain for your critical pharmaceutical intermediates.
