Optimizing Edoxaban Tosylate Intermediate Production for Global Pharmaceutical Supply Chains
The pharmaceutical industry continuously seeks robust synthetic pathways for critical anticoagulant agents like Edoxaban Tosylate, a direct Factor Xa inhibitor widely prescribed for thromboembolism prevention. Patent CN114456194B introduces a transformative methodology for synthesizing the key intermediate, 4,5,6,7-tetrahydro-5-methyl-thiazolo[5,4-c]pyridine-2-carboxylic acid, addressing long-standing inefficiencies in prior art. This innovation leverages a strategic sequence starting from readily available N-methyl-4-piperidone, bypassing the need for hazardous organolithium reagents that have historically plagued production scalability. By integrating mild reaction conditions and stable intermediates, this protocol offers a compelling value proposition for reliable pharmaceutical intermediates supplier networks aiming to secure consistent quality. The technical breakthrough lies not merely in the chemical transformation but in the holistic optimization of safety, cost, and environmental compliance, making it an ideal candidate for cost reduction in API manufacturing where margin pressure is intense.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of this pivotal thiazolo-pyridine scaffold has relied heavily on routes involving 4-aminopyridine or N-methyl-4-piperidone coupled with dangerous reagents like n-butyl lithium and elemental sulfur. These conventional pathways, while chemically valid, present severe operational hazards including pyrophoric risks and the handling of volatile, toxic iodomethane. The dependency on cryogenic conditions for lithiation steps drastically increases energy consumption and equipment complexity, creating bottlenecks for commercial scale-up of complex pharmaceutical intermediates. Furthermore, the use of elemental sulfur often leads to difficult-to-remove impurities and unpleasant odors, complicating waste treatment and increasing the environmental footprint. Such factors collectively undermine supply chain continuity, as any disruption in the supply of specialized hazardous reagents can halt production lines entirely, posing a significant risk to procurement managers overseeing global API sourcing strategies.
The Novel Approach
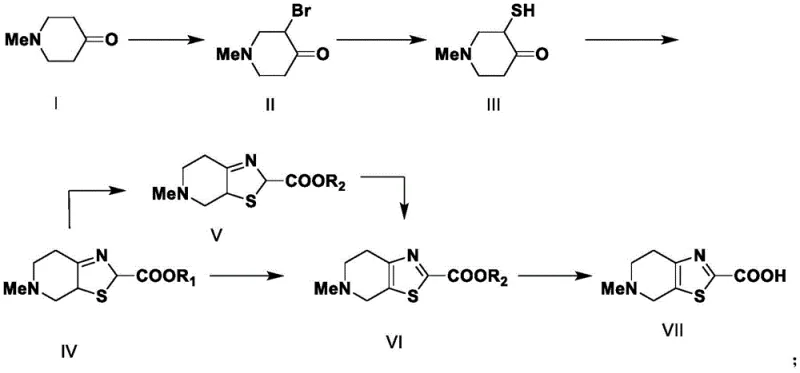
In stark contrast, the novel methodology detailed in the patent utilizes a bromination-thiolation-cyclization sequence that operates under significantly milder and safer conditions. By substituting hazardous lithiation with a controlled bromination using hydrobromic acid and bromine, the process eliminates pyrophoric risks entirely. The subsequent thiolation employs sodium sulfide in an aqueous medium, avoiding the handling of solid elemental sulfur and its associated safety liabilities. This shift allows for reactions to proceed at near-ambient temperatures, reducing energy costs and simplifying reactor requirements. The streamlined workflow enhances the stability of intermediates during transport and storage, directly supporting reducing lead time for high-purity pharmaceutical intermediates by minimizing quarantine and testing delays associated with unstable compounds. This approach represents a paradigm shift towards greener, more economically viable manufacturing practices.
Mechanistic Insights into Bromination and Thiolation Cascade
The core of this synthetic advancement lies in the precise control of the alpha-bromination of N-methyl-4-piperidone, which sets the stage for nucleophilic substitution. The reaction is conducted in glacial acetic acid with hydrobromic acid, maintaining temperatures between 20-25°C to prevent poly-bromination or degradation. This careful thermal management ensures high selectivity for the mono-brominated species, which is crucial for downstream purity. Following isolation, the bromo-ketone undergoes nucleophilic attack by sulfide ions generated from sodium sulfide. This thiolation step is exothermic and requires cooling to -5-0°C to manage reaction kinetics and prevent over-reaction or decomposition of the sensitive mercapto group. The resulting mercapto-ketone is a versatile synthon that retains the integrity of the piperidine ring while introducing the necessary sulfur atom for thiazole formation.
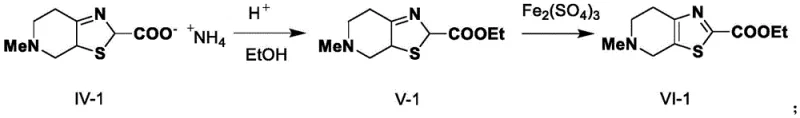
Subsequent cyclization with glyoxylic acid and ammonia constructs the thiazole ring fused to the piperidine system. This condensation reaction proceeds efficiently in methanol, leveraging the nucleophilicity of the thiol and the electrophilicity of the aldehyde group in glyoxylic acid. The formation of the ammonium carboxylate salt intermediate stabilizes the molecule, preventing premature hydrolysis or decarboxylation. This mechanistic pathway avoids the harsh basic conditions often required in traditional Hantzsch-type thiazole syntheses, thereby preserving the stereochemical integrity and minimizing racemization risks. The final aromatization to the thiazolo-pyridine system is achieved through oxidative dehydrogenation using ferric sulfate, a mild oxidant that cleanly removes hydrogen without over-oxidizing the sulfur or nitrogen atoms. This sequence ensures a clean impurity profile, critical for meeting stringent regulatory standards for high-purity pharmaceutical intermediates.
How to Synthesize 4,5,6,7-tetrahydro-5-methyl-thiazolo[5,4-c]pyridine-2-carboxylic acid Efficiently
Implementing this synthesis requires strict adherence to the specified stoichiometric ratios and thermal profiles to maximize yield and safety. The process begins with the preparation of the bromo-ketone, followed immediately by the thiolation to minimize storage time of the reactive halide. Operators must ensure efficient mixing during the addition of sodium sulfide to prevent local hot spots. The cyclization step benefits from extended stirring times at ambient temperature to drive the equilibrium towards the ring-closed product. Finally, the oxidative step should be monitored via HPLC to determine the exact endpoint, preventing over-oxidation. Detailed standardized synthesis steps are provided below to guide technical teams in replicating these results accurately.
- Perform bromination of N-methyl-4-piperidone to generate 3-bromo-N-methyl-4-piperidone under controlled acidic conditions.
- Execute thiolation using sodium sulfide to replace the bromine atom, forming the mercapto-ketone intermediate safely.
- Conduct cyclization with glyoxylic acid and ammonia, followed by esterification and oxidation to yield the final aromatic intermediate.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, this patented route offers substantial strategic advantages for organizations focused on cost reduction in API manufacturing. By eliminating expensive and hazardous reagents like n-butyl lithium, the raw material costs are significantly lowered, and the safety infrastructure requirements are reduced. The use of common solvents like methanol and ethanol, along with inexpensive inorganic salts, further drives down the variable cost per kilogram. This economic efficiency allows suppliers to offer more competitive pricing without compromising on quality, providing a buffer against market volatility in raw material prices. Additionally, the simplified workup procedures reduce solvent consumption and waste generation, aligning with modern sustainability goals and reducing disposal costs.
- Cost Reduction in Manufacturing: The elimination of cryogenic operations and pyrophoric reagents translates to lower capital expenditure on specialized equipment and reduced operational expenses. The high yield of each step minimizes material loss, ensuring that more input material is converted into saleable product. This efficiency directly impacts the bottom line, allowing for substantial cost savings that can be passed down the supply chain or reinvested in R&D. The avoidance of complex purification steps also reduces labor hours and utility consumption, further enhancing the overall economic viability of the process.
- Enhanced Supply Chain Reliability: Sourcing common chemicals like sodium sulfide and glyoxylic acid is far more reliable than procuring specialized organolithium solutions, which often have long lead times and strict transport regulations. This accessibility ensures continuous production capability, reducing the risk of stockouts that can disrupt downstream API synthesis. The stability of the intermediates allows for batch stocking, providing flexibility to meet sudden spikes in demand. This robustness makes the supply chain more resilient to external shocks, ensuring a steady flow of materials to pharmaceutical manufacturers.
- Scalability and Environmental Compliance: The mild reaction conditions and aqueous workups make this process inherently scalable from pilot plant to multi-ton production without significant re-engineering. The reduced generation of hazardous waste simplifies compliance with environmental regulations, lowering the administrative burden and potential liability. The use of ferric sulfate as an oxidant produces iron sludge which is easier to treat than heavy metal catalyst residues. This environmental friendliness enhances the corporate social responsibility profile of the manufacturer, appealing to eco-conscious partners and regulators alike.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis route. They are derived from the specific advantages and operational parameters outlined in the patent documentation. Understanding these details helps stakeholders assess the feasibility of adopting this technology for their specific production needs. The answers reflect the consensus on safety, quality, and scalability inherent in this novel methodology.
Q: How does this new route improve safety compared to conventional methods?
A: This protocol eliminates the use of hazardous n-butyl lithium and elemental sulfur, significantly reducing fire risks and handling complexities associated with pyrophoric reagents.
Q: What are the purity specifications for the final intermediate?
A: The process is designed to minimize side reactions through mild temperature controls, facilitating easier purification and ensuring high chemical purity suitable for API synthesis.
Q: Is this synthesis method scalable for industrial production?
A: Yes, the use of stable raw materials and avoidance of extreme low-temperature requirements makes this route highly adaptable for large-scale commercial manufacturing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Edoxaban Tosylate Intermediate Supplier
NINGBO INNO PHARMCHEM stands at the forefront of implementing such advanced synthetic technologies to serve the global pharmaceutical market. Our extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production ensures that we can seamlessly transition this patent-pending chemistry into robust manufacturing operations. We maintain stringent purity specifications and operate rigorous QC labs to guarantee that every batch meets the exacting standards required for anticoagulant drug synthesis. Our commitment to technical excellence means we do not just supply chemicals; we deliver validated processes that enhance your production efficiency.
We invite you to engage with our technical procurement team to discuss how this innovative route can benefit your specific supply chain. Request a Customized Cost-Saving Analysis to quantify the potential economic impact of switching to this safer, more efficient methodology. We are prepared to provide specific COA data and route feasibility assessments to support your decision-making process. Partner with us to secure a sustainable and cost-effective source of high-quality intermediates for your critical pharmaceutical projects.
