Optimizing Dutasteride Production: A Novel Dehydrogenation and Amidation Strategy for Commercial Scale
Optimizing Dutasteride Production: A Novel Dehydrogenation and Amidation Strategy for Commercial Scale
The pharmaceutical landscape for Benign Prostatic Hyperplasia (BPH) treatment has been significantly shaped by the introduction of 5-alpha-reductase inhibitors, with Dutasteride standing out as a potent dual inhibitor of Type I and Type II enzymes. As demand for high-purity active pharmaceutical ingredients (APIs) grows, the efficiency of the synthetic route becomes a critical bottleneck for supply chains globally. Patent CN103254268A discloses a transformative process for preparing Dutasteride that addresses longstanding issues regarding safety, yield, and environmental impact. This technical insight report analyzes the shift from traditional multi-step syntheses to a streamlined approach utilizing DDQ-mediated dehydrogenation and triphosgene-based amidation. By leveraging commercially available 4-aza-5-alpha-androstane-3-ketone-17-beta-carboxylic acid as a starting material, this method offers a robust pathway for industrial manufacturing.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
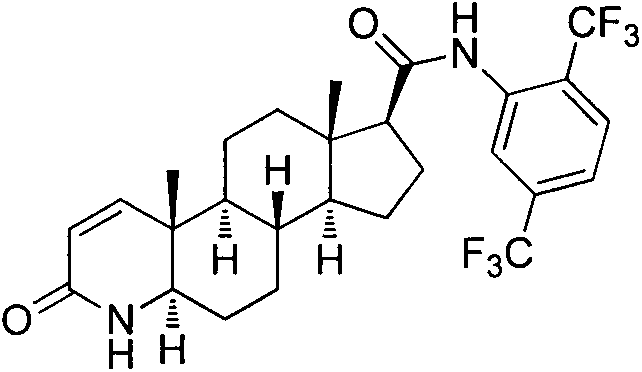
Historically, the synthesis of Dutasteride has been plagued by operational complexity and significant safety hazards inherent to early-generation protocols. As detailed in prior art such as WO95/07927, conventional routes often commence with pregnenolone acid derivatives, necessitating a tedious sequence of reactions including oxidation, ring-opening, azepine cyclization, and multiple reduction steps before reaching the target scaffold. These lengthy sequences not only depress overall yields due to cumulative losses at each stage but also introduce a wide array of difficult-to-remove impurities that compromise the quality of the final API. Furthermore, traditional amidation steps frequently rely on thionyl chloride (SOCl2) as a chlorinating agent. While chemically effective, thionyl chloride is highly corrosive to reactor vessels and generates substantial quantities of sulfur dioxide gas, a toxic asphyxiant that requires expensive and complex scrubbing systems to meet environmental regulations.
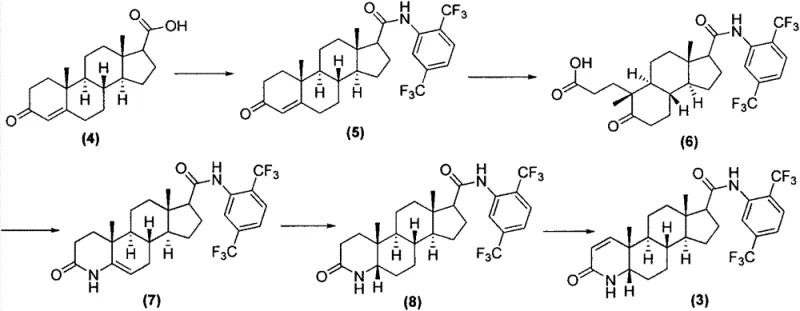
Additionally, the use of pyridine as a solvent or acid-binding agent in older methodologies presents severe occupational health risks. Pyridine is a known neurotoxin with a pervasive, unpleasant odor that is notoriously difficult to eliminate from the final crystal lattice, often leading to product defects and failed quality control specifications. The transportation and storage of liquid thionyl chloride are also strictly regulated due to its volatility and reactivity, adding logistical friction to the supply chain. These factors combined create a manufacturing environment that is costly to maintain, hazardous for operators, and environmentally unsustainable, driving the urgent need for the greener alternative presented in the patent data.
The Novel Approach
The methodology outlined in CN103254268A represents a paradigm shift by truncating the synthetic timeline and replacing hazardous reagents with safer, solid-state alternatives. Instead of building the steroid skeleton from scratch, this process utilizes a readily available 4-aza-androstane derivative (Compound 1) as the foundation. The innovation lies in a direct 1,2-dehydrogenation followed by a highly controlled amidation. By bypassing the ring-opening and reclosing steps required in prior art, the new route drastically reduces the number of unit operations, thereby minimizing solvent consumption and waste generation. The replacement of liquid thionyl chloride with solid triphosgene is particularly noteworthy; triphosgene acts as a stable source of phosgene equivalents but can be weighed and handled as a solid powder, significantly reducing the risk of accidental exposure and equipment corrosion. This strategic substitution not only enhances process safety but also simplifies the engineering controls required for commercial scale-up.
Mechanistic Insights into DDQ-Mediated Dehydrogenation and Triphosgene Activation
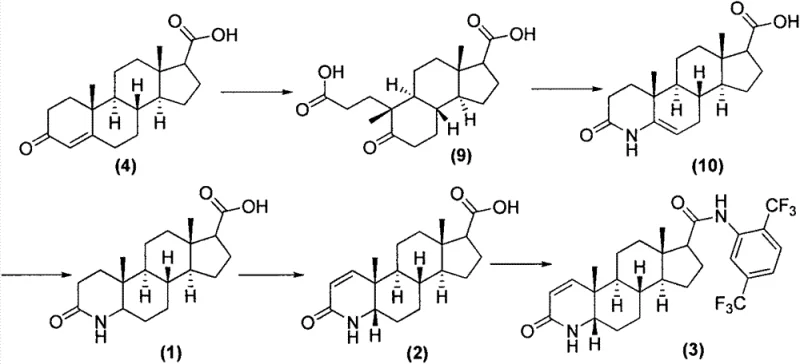
The core chemical transformation in this novel process is the introduction of the 1,2-double bond into the A-ring of the steroid nucleus, a critical structural feature for biological activity. This is achieved through the synergistic action of 2,3-dichloro-5,6-dicyano-1,4-benzoquinone (DDQ) and N,O-bis(trimethylsilyl)trifluoroacetamide (BSTFA). In this mechanism, BSTFA likely serves to silylate the amide nitrogen or the enolizable positions, increasing the electron density and facilitating the hydride abstraction by DDQ. The reaction proceeds in toluene, a solvent chosen for its ability to sustain the elevated temperatures (120-130°C) required to drive the dehydrogenation to completion without decomposing the sensitive steroid backbone. This specific reagent combination avoids the use of heavy metal catalysts or harsh acidic conditions that could lead to epimerization or degradation of the chiral centers, ensuring the stereochemical integrity of the 5-alpha configuration is preserved throughout the synthesis.
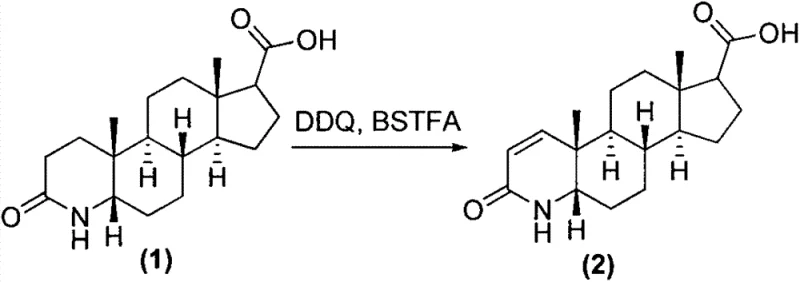
Following dehydrogenation, the activation of the C17-carboxylic acid is executed using triphosgene in the presence of a catalytic amount of dimethylformamide (DMF). The DMF catalyzes the formation of the Vilsmeier-Haack type intermediate, which rapidly converts the carboxylic acid into the corresponding acyl chloride in situ. Unlike the violent exotherm associated with thionyl chloride, the reaction with solid triphosgene can be carefully modulated by controlling the addition rate under ice-bath conditions, keeping the temperature below 60°C. Subsequent addition of 2,5-bis(trifluoromethyl)aniline in the presence of sodium carbonate allows for a clean nucleophilic attack on the acyl chloride. Sodium carbonate acts as a mild, inorganic acid scavenger, neutralizing the HCl byproduct without introducing organic amine impurities that are difficult to purge. This mechanistic elegance ensures that the final crude product contains minimal side products, streamlining the downstream purification process.
How to Synthesize Dutasteride Efficiently
The implementation of this synthesis requires precise control over reaction parameters to maximize yield and purity. The process is divided into two distinct stages: the oxidative dehydrogenation of the saturated precursor and the subsequent amide bond formation. Operators must adhere to strict temperature profiles, particularly during the reflux stage of the dehydrogenation and the exothermic activation phase of the amidation. The use of toluene as a primary solvent across both steps allows for potential telescoping or solvent recycling, further enhancing process economics. Detailed standard operating procedures regarding reagent stoichiometry, addition rates, and workup protocols are essential for reproducibility. For a comprehensive breakdown of the standardized synthesis steps, please refer to the technical guide below.
- Perform 1,2-dehydrogenation of the starting steroid acid using DDQ and BSTFA in toluene at elevated temperatures to form the unsaturated intermediate.
- Activate the carboxylic acid group of the intermediate using solid triphosgene and a catalytic amount of DMF in toluene under ice-bath conditions.
- React the activated acyl chloride species with 2,5-bis(trifluoromethyl)aniline in the presence of sodium carbonate to yield the final Dutasteride product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the transition to this novel synthetic route offers tangible benefits that extend beyond mere chemical yield. The elimination of hazardous liquid reagents like thionyl chloride and pyridine fundamentally alters the cost structure of manufacturing by reducing the need for specialized corrosion-resistant equipment and complex waste gas treatment facilities. The use of solid triphosgene allows for more precise dosing and inventory management, reducing raw material waste and lowering the total cost of ownership for the production line. Furthermore, the simplified workflow reduces the overall cycle time per batch, enabling manufacturers to respond more agilely to market fluctuations in API demand without compromising on safety or regulatory compliance standards.
- Cost Reduction in Manufacturing: The substitution of expensive and hazardous reagents with cost-effective alternatives like sodium carbonate and solid triphosgene drives significant operational savings. By removing the need for extensive scrubbing systems required for sulfur dioxide emissions, capital expenditure on plant infrastructure is minimized. Additionally, the higher selectivity of the DDQ/BSTFA system reduces the burden on purification units, lowering solvent consumption and energy costs associated with distillation and recrystallization steps.
- Enhanced Supply Chain Reliability: Utilizing commercially available starting materials such as 4-aza-5-alpha-androstane-3-ketone-17-beta-carboxylic acid ensures a stable supply base, mitigating the risk of raw material shortages. The robustness of the reaction conditions, which tolerate standard industrial solvents like toluene and dichloromethane, means that production is less susceptible to disruptions caused by the scarcity of exotic reagents. This reliability is crucial for maintaining continuous production schedules and meeting delivery commitments to downstream pharmaceutical partners.
- Scalability and Environmental Compliance: The process is inherently designed for green chemistry principles, generating fewer toxic byproducts and utilizing reagents that are easier to contain and dispose of safely. The absence of heavy metals and the reduction of volatile organic amine emissions simplify the environmental permitting process for new manufacturing sites. This scalability ensures that production can be ramped up from pilot batches to multi-ton commercial volumes without encountering the engineering bottlenecks typical of more complex, multi-step synthetic routes.
Frequently Asked Questions (FAQ)
The following questions address common technical inquiries regarding the implementation of this synthesis route. These answers are derived directly from the experimental data and comparative analysis provided in the patent literature, focusing on the practical implications for process chemistry and quality assurance teams.
Q: Why is triphosgene preferred over thionyl chloride in this synthesis?
A: Triphosgene is a solid crystalline reagent that is stable at room temperature and easier to measure accurately compared to liquid thionyl chloride. It significantly reduces equipment corrosion and eliminates the release of sulfur dioxide gas, simplifying waste gas treatment and improving operator safety.
Q: What are the advantages of using sodium carbonate over pyridine as an acid binding agent?
A: Sodium carbonate is an inorganic base that avoids the use of pyridine, which is neurotoxic, has a strong unpleasant odor, and is difficult to remove completely from the final product. Using sodium carbonate results in cleaner products with fewer impurities and reduces environmental pollution.
Q: How does the DDQ/BSTFA system improve the dehydrogenation step?
A: The combination of DDQ (2,3-dichloro-5,6-dicyano-1,4-benzoquinone) and BSTFA (N,O-bis(trimethylsilyl)trifluoroacetamide) facilitates a mild and efficient 1,2-dehydrogenation. This system operates under relatively safe conditions compared to harsher oxidative methods, yielding high purity intermediates suitable for subsequent amidation.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Dutasteride Supplier
At NINGBO INNO PHARMCHEM, we recognize that the successful commercialization of complex pharmaceutical intermediates requires more than just a patent; it demands deep process engineering expertise. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory bench to industrial reactor is seamless. We are committed to delivering high-purity Dutasteride intermediates that meet stringent purity specifications, supported by our rigorous QC labs equipped with state-of-the-art analytical instrumentation. Our facility is optimized to handle the specific safety requirements of reagents like triphosgene, guaranteeing a secure and compliant supply chain for our global partners.
We invite you to collaborate with us to optimize your supply chain for BPH therapeutics. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements. Contact us today to request specific COA data and route feasibility assessments, and let us demonstrate how our advanced manufacturing capabilities can enhance your project's bottom line while ensuring the highest standards of quality and reliability.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
