Scalable Semi-Synthesis of Natural Brassinolide via Strategic Side-Chain Engineering
Introduction to Advanced Brassinolide Manufacturing
The global demand for high-efficiency plant growth regulators has intensified the search for scalable production methods of natural brassinolide, the most biologically active member of the brassinosteroid family. Patent CN113912666A introduces a groundbreaking semi-synthetic methodology that addresses the historical bottlenecks of supply and cost. Unlike previous attempts that relied on impractical extraction from vast quantities of rape pollen or prohibitively complex total synthesis, this invention utilizes epibrassinolide as a strategic starting material. By employing a concise four-step sequence involving selective protection, oxidation, stereoselective methyl transposition, and reduction, the process effectively reconstructs the critical 24-beta methyl stereochemistry. This technical breakthrough represents a significant leap forward for manufacturers seeking a reliable agrochemical intermediate supplier capable of delivering high-purity materials at an industrial scale.
The significance of this patent lies in its ability to bypass the most challenging aspect of brassinolide synthesis: the construction of the chiral side chain. Traditional routes often struggle with low diastereoselectivity and excessive step counts, leading to poor overall yields and high waste generation. The disclosed method streamlines this by leveraging the existing steroid skeleton of epibrassinolide, modifying only the necessary functional groups to achieve the natural configuration. This approach not only enhances the economic feasibility of producing natural brassinolide but also aligns with modern green chemistry principles by minimizing solvent usage and reaction steps. For R&D directors and procurement specialists, this route offers a tangible pathway to secure a stable supply chain for this high-value agricultural input.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the acquisition of natural brassinolide has been plagued by severe inefficiencies that hindered its widespread agricultural application. The initial discovery relied on extraction from plant sources, specifically rape pollen, but the concentration of the active compound is infinitesimally low. Reports indicate that obtaining merely 4mg of high-activity crystal required processing 227kg of pollen, a ratio that renders extraction commercially impossible for large-scale farming needs. Consequently, the industry turned to chemical synthesis, but early total synthesis routes were fraught with difficulties. As illustrated in prior art, such as the route published in Can. J. Chem. Vol. 71 (1993), synthesizing the molecule from basic building blocks involves numerous steps, each presenting chiral selectivity problems.
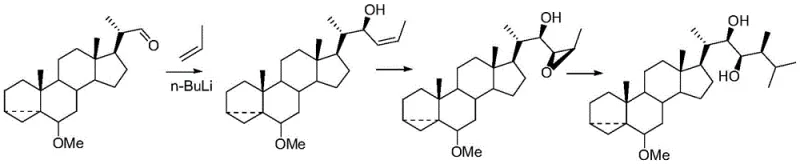
These conventional total synthesis pathways suffer from cumulative yield losses, where the efficiency of each individual step compounds to result in a negligible final output. Furthermore, the construction of the side chain, particularly the establishment of the 24-position beta-methyl group, requires sophisticated chiral auxiliaries or resolution techniques that are expensive and difficult to scale. The separation of diastereomers at multiple stages increases processing time and solvent consumption, driving up the cost of goods sold (COGS) to levels that are unsustainable for commodity agrochemicals. These structural and economic barriers have long prevented natural brassinolide from realizing its full potential in the global market, creating a persistent supply gap that this new patent aims to fill.
The Novel Approach
In stark contrast to the arduous total synthesis methods, the novel approach detailed in CN113912666A adopts a semi-synthetic strategy that drastically simplifies the molecular assembly. By starting with epibrassinolide, a commercially accessible analog that already possesses the correct tetracyclic steroid core, the synthesis focuses exclusively on modifying the side chain stereochemistry. The process is elegantly divided into four logical stages: protection of the 2,3-dihydroxy system, oxidation of the 23-hydroxyl, transposition of the 24-methyl group, and final reduction. This strategic shortcut eliminates the need to build the molecule from scratch, thereby reducing the step count and significantly improving the overall mass balance of the operation.
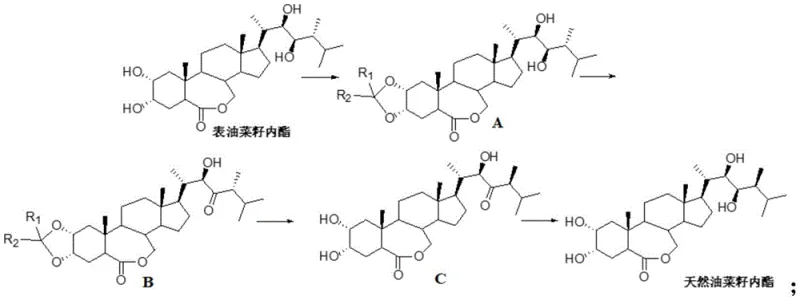
The core innovation of this route is the clever utilization of enolization chemistry to invert the stereochemistry at the C-24 position. Instead of attempting to install the methyl group with the correct stereochemistry de novo, the method starts with the incorrect isomer (alpha) and uses thermodynamic or kinetic control to transpose it to the desired beta configuration. This transformation is achieved under mild conditions using standard reagents, avoiding the need for exotic catalysts or extreme temperatures. The result is a robust, scalable process that transforms a lower-value starting material into a premium product with high efficiency. For supply chain managers, this translates to a more predictable production timeline and reduced dependency on complex, multi-vendor precursor sourcing strategies.
Mechanistic Insights into Stereoselective Methyl Transposition
The heart of this synthetic achievement lies in the mechanistic execution of the 24-methyl transposition, a step that resolves the primary stereochemical discrepancy between epibrassinolide and natural brassinolide. The process begins with the selective protection of the 2 and 3-hydroxyl groups on the A-ring using a ketal reagent. This protection is crucial because it masks these reactive sites, preventing them from interfering with subsequent reactions on the side chain. Without this safeguard, the oxidants and bases used later could indiscriminately react with the A-ring hydroxyls, leading to a complex mixture of byproducts and severely compromising the purity of the final API intermediate. The choice of ketal reagent and acid catalyst is optimized to ensure high conversion while maintaining the integrity of the sensitive lactone ring.
Following protection, the 23-hydroxyl group is selectively oxidized to a ketone using Dess-Martin periodinane. This specific oxidant is chosen for its mildness and high chemoselectivity; it effectively converts the secondary alcohol to a ketone without over-oxidizing other parts of the molecule or cleaving the acid-sensitive protecting groups. Once the 23-ketone is formed, the stage is set for the critical transposition. Under basic conditions, typically using potassium tert-butoxide, the alpha-proton at the C-24 position is abstracted to form an enolate intermediate. This enolization allows the 24-methyl group to equilibrate from the less stable alpha orientation to the thermodynamically favored beta orientation. Subsequent protonation locks the methyl group into the natural configuration. Finally, the 23-carbonyl is reduced back to a hydroxyl group using a borohydride reagent, restoring the side chain functionality while preserving the newly established stereochemistry. This sequence demonstrates a profound understanding of physical organic chemistry to solve a complex stereochemical problem with minimal operational complexity.
How to Synthesize Natural Brassinolide Efficiently
The synthesis of natural brassinolide via this patented route offers a streamlined alternative to traditional methods, focusing on high-yield transformations and easily manageable reaction conditions. The protocol is designed for reproducibility, utilizing common laboratory solvents and reagents that are readily available in bulk quantities. The initial protection step sets the foundation for selectivity, followed by precise oxidation and the pivotal methyl transposition that defines the molecule's biological activity. The final reduction and deprotection steps yield the target compound with high fidelity. For process chemists looking to implement this technology, the detailed standardized synthesis steps are outlined below to ensure consistent quality and yield across batches.
- Protect the 2,3-dihydroxy groups of epibrassinolide using a ketal reagent and acid catalyst to form Compound A.
- Oxidize the 23-hydroxyl group of Compound A to a ketone using Dess-Martin periodinane to obtain Compound B.
- Perform enolization on Compound B using a base like potassium tert-butoxide to transpose the 24-alpha methyl to the 24-beta position, yielding Compound C.
- Reduce the 23-carbonyl group of Compound C using a borohydride reducing agent to finalize the natural brassinolide structure.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this semi-synthetic route offers compelling economic and logistical benefits that directly impact the bottom line. The primary advantage is the drastic reduction in manufacturing complexity. By shortening the synthetic route from over twenty steps in total synthesis to just four key transformations, the process significantly lowers the consumption of raw materials, solvents, and energy. This reduction in process intensity translates directly into cost reduction in plant growth regulator manufacturing, allowing for more competitive pricing in the agricultural sector. Furthermore, the reliance on epibrassinolide as a starting material leverages an existing, mature supply chain, mitigating the risk of raw material shortages that often plague novel synthetic pathways dependent on obscure precursors.
- Cost Reduction in Manufacturing: The elimination of complex chiral resolution steps and the reduction in total unit operations lead to substantial cost savings. Traditional methods require expensive chiral columns and multiple recrystallizations to separate diastereomers, which are both time-consuming and costly. This new method achieves the desired stereochemistry through a chemical transposition, effectively turning a purification cost into a simple reaction step. Additionally, the use of mild reagents like Dess-Martin periodinane and sodium borohydride avoids the need for heavy metal catalysts, which often require expensive removal processes to meet regulatory limits for agrochemicals. The overall simplified workflow reduces labor hours and equipment occupancy time, enhancing the overall asset utilization of the production facility.
- Enhanced Supply Chain Reliability: Sourcing strategies are greatly simplified by using epibrassinolide, a commercially available bulk chemical, rather than synthesizing the steroid core from petrochemical feedstocks. This shifts the supply risk away from the manufacturer and onto established suppliers of the starting material, ensuring greater continuity of supply. The robustness of the reaction conditions, which tolerate standard industrial solvents like dichloromethane and tetrahydrofuran, means that the process can be easily transferred between different manufacturing sites without requiring specialized equipment. This flexibility is crucial for maintaining supply continuity in the face of regional disruptions or capacity constraints, making the manufacturer a more reliable partner for long-term contracts.
- Scalability and Environmental Compliance: The process is inherently scalable due to its reliance on homogeneous reactions and standard workup procedures like extraction and crystallization. Unlike heterogeneous catalytic processes that can suffer from mass transfer limitations at large scales, this solution-phase chemistry scales linearly from kilogram to tonne production. From an environmental perspective, the shorter route generates less chemical waste per kilogram of product, reducing the burden on wastewater treatment facilities. The avoidance of toxic heavy metals and the use of recyclable solvents align with increasingly stringent environmental regulations, reducing the risk of compliance-related shutdowns and enhancing the sustainability profile of the final product for eco-conscious customers.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production of natural brassinolide using this advanced semi-synthetic methodology. These answers are derived directly from the technical specifications and experimental data provided in the patent documentation, offering clarity on the feasibility and advantages of the process. Understanding these details is essential for stakeholders evaluating the integration of this technology into their existing supply chains or product portfolios.
Q: Why is the 24-position methyl transposition critical in this synthesis?
A: The defining structural difference between epibrassinolide and natural brassinolide is the stereochemistry at the C-24 position. Natural brassinolide requires a 24-beta methyl configuration, whereas the readily available epibrassinolide has a 24-alpha methyl. This transposition step is the core innovation that converts a cheap, abundant starting material into the high-value natural isomer without requiring complex total synthesis from scratch.
Q: How does this method improve upon traditional extraction or total synthesis?
A: Traditional extraction from pollen yields negligible amounts (approx. 4mg from 227kg), making it commercially unviable. Total synthesis involves over 20 steps with difficult chiral control. This semi-synthetic route reduces the process to just four high-yielding steps, leveraging the existing steroid backbone of epibrassinolide to bypass the most difficult stereochemical constructions.
Q: What are the key purity considerations for the final product?
A: Purity is managed through selective protection of the A-ring hydroxyls to prevent side reactions during side-chain modification. The use of mild oxidants like Dess-Martin periodinane prevents over-oxidation, and the final recrystallization step ensures the removal of any remaining isomers or unreacted intermediates, meeting stringent specifications for agrochemical applications.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Natural Brassinolide Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of this patented synthesis route for the global agrochemical market. As a premier CDMO partner, we possess the technical expertise to translate this laboratory-scale innovation into a robust commercial reality. Our facilities are equipped to handle the specific requirements of steroid chemistry, including the precise temperature control needed for the enolization and reduction steps. We bring extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your demand for high-purity natural brassinolide is met with consistency and reliability. Our rigorous QC labs enforce stringent purity specifications, guaranteeing that every batch meets the highest standards for biological efficacy and safety.
We invite you to collaborate with us to leverage this cutting-edge technology for your product lines. By partnering with our technical procurement team, you can access a Customized Cost-Saving Analysis that quantifies the economic benefits of switching to this semi-synthetic route for your specific volume requirements. We encourage you to reach out today to request specific COA data and route feasibility assessments tailored to your project needs. Let us help you secure a sustainable and cost-effective supply of this critical plant growth regulator, driving value and efficiency in your agricultural solutions.
