Advanced Rhodium-Catalyzed C-H Activation for Scalable Isoquinoline Pharmaceutical Intermediates Production
Advanced Rhodium-Catalyzed C-H Activation for Scalable Isoquinoline Pharmaceutical Intermediates Production
The landscape of pharmaceutical intermediate manufacturing is undergoing a significant transformation driven by the need for more sustainable and efficient synthetic methodologies. A pivotal advancement in this domain is detailed in patent CN111018779B, which discloses a novel synthetic route for 2-(3-isoquinolyl)-propionic acid ethyl ester derivatives. This technology leverages the power of transition metal catalysis to achieve direct ortho-C-H bond activation of aromatic rings, bypassing traditional multi-step functionalization processes. By utilizing easily accessible O-acetyl oximes and allenoic acid esters as starting materials, this method offers a streamlined pathway to construct complex isoquinoline scaffolds that are ubiquitous in bioactive molecules. For R&D directors and process chemists, this represents a critical opportunity to enhance synthetic efficiency while reducing the environmental footprint of API production.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of isoquinoline derivatives has relied heavily on classical cyclization strategies that often necessitate harsh reaction conditions and extensive pre-functionalization of substrates. Traditional routes typically require the introduction of specific leaving groups, such as halogens, onto the aromatic ring prior to cyclization, which inherently lowers the overall atom economy of the process. Furthermore, these conventional methods frequently suffer from narrow substrate scope, limiting the structural diversity that can be achieved without redesigning the entire synthetic sequence. The reliance on stoichiometric oxidants or reductants in older protocols also generates substantial chemical waste, posing challenges for waste management and increasing the cost of goods sold. Additionally, the sensitivity of many traditional reagents to moisture and air complicates scale-up operations, leading to inconsistent batch quality and extended production timelines.
The Novel Approach
In stark contrast, the methodology described in the patent introduces a robust rhodium-catalyzed cross-coupling reaction that operates under remarkably mild conditions. This innovative approach utilizes O-acetyl oximes, which serve as both the substrate and the directing group for the activation of the ortho-C-H bond. The reaction proceeds efficiently in the presence of a rhodium salt catalyst and a silver additive, facilitating the coupling with allenoic acid esters to form the isoquinoline core. As illustrated in the general reaction scheme below, this process eliminates the need for pre-functionalization, thereby simplifying the supply chain for raw materials.
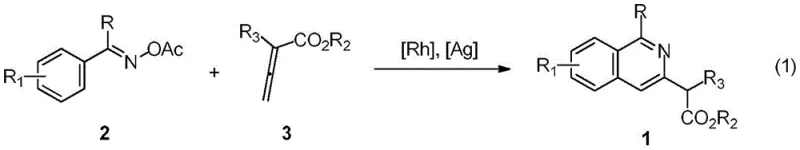
The operational simplicity is further enhanced by the use of common solvents like 1,2-dichloroethane and moderate temperatures around 60 °C. This shift from harsh, multi-step sequences to a direct catalytic coupling not only accelerates the synthesis but also significantly improves the purity profile of the final product by minimizing side reactions associated with aggressive reagents.
Mechanistic Insights into Rhodium-Catalyzed C-H Activation
The core of this technological breakthrough lies in the mechanism of rhodium(III)-catalyzed C-H bond activation. The process initiates with the coordination of the rhodium catalyst to the nitrogen atom of the O-acetyl oxime, which acts as a powerful directing group. This coordination brings the metal center into close proximity with the ortho-C-H bond of the aromatic ring, facilitating its cleavage through a concerted metalation-deprotonation (CMD) pathway assisted by the pivalic acid additive. Once the rhodacycle intermediate is formed, the allenoic acid ester inserts into the metal-carbon bond. The unique electronic structure of the allenoate allows for regioselective insertion, ensuring the formation of the desired isoquinoline skeleton with high fidelity. Subsequent reductive elimination or protonolysis releases the final product and regenerates the active catalytic species, completing the cycle.
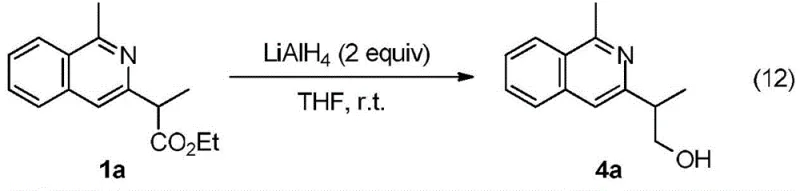
From an impurity control perspective, this mechanism offers distinct advantages over radical-based or high-temperature thermal cyclizations. The mild reaction temperature of 60 °C prevents the thermal degradation of sensitive functional groups that might be present on the aromatic ring or the ester side chain. Furthermore, the high selectivity of the rhodium catalyst minimizes the formation of regioisomers, which are often difficult to separate and can compromise the safety profile of pharmaceutical intermediates. The ability to further derivatize the product, such as the reduction of the ester group to an alcohol using lithium aluminum hydride as shown below, demonstrates the versatility of the scaffold for downstream medicinal chemistry applications.
How to Synthesize 2-(3-isoquinolyl)-propionic Acid Ethyl Ester Efficiently
The practical implementation of this synthesis is designed for reproducibility and ease of handling, making it suitable for both laboratory discovery and pilot-scale production. The protocol involves a straightforward mixing of reagents under an inert atmosphere, followed by a standard heating period. Detailed standard operating procedures regarding exact stoichiometry, safety precautions for handling rhodium catalysts, and specific workup parameters are critical for maintaining batch-to-batch consistency. The following section outlines the generalized workflow derived from the patent examples, serving as a foundational guide for process development teams looking to adopt this technology.
- Weigh O-acetyl oxime (0.3 mmol), [RuCp*Cl2]2 catalyst (0.006 mmol), silver hexafluoroantimonate (0.03 mmol), and pivalic acid (0.06 mmol) in a glove box and add to a reaction vessel.
- Add 1,2-dichloroethane (3 mL) under nitrogen atmosphere, followed by the diacrylate ester (0.6 mmol), and heat the mixture at 60 °C for 24 hours.
- Upon completion, remove solvent via rotary distillation and purify the crude product using silica gel column chromatography with petroleum ether and ethyl acetate.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain leaders, the adoption of this rhodium-catalyzed technology translates into tangible strategic benefits beyond mere chemical elegance. The elimination of pre-functionalized starting materials means that the supply chain can rely on commodity chemicals like simple acetophenone oximes and commercially available allenoates, rather than expensive, custom-synthesized halogenated precursors. This simplification of the bill of materials reduces exposure to price volatility in the specialty chemical market and shortens the lead time for raw material acquisition. Moreover, the high atom economy of the reaction ensures that a greater proportion of the input mass is converted into the valuable product, directly impacting the cost of goods sold by reducing raw material waste.
- Cost Reduction in Manufacturing: The removal of stoichiometric oxidants and reductors from the process equation leads to significant cost savings in reagent procurement and waste disposal. By avoiding the use of heavy metal oxidants often required in traditional cyclizations, the downstream purification process is simplified, reducing the consumption of silica gel and solvents during chromatography. Although the rhodium catalyst is a precious metal, its usage at low catalytic loadings (e.g., 2 mol%) combined with the potential for recovery makes the overall process economically viable compared to the cumulative costs of multi-step traditional routes.
- Enhanced Supply Chain Reliability: The robustness of the reaction conditions, specifically the tolerance for various functional groups and the use of stable solvents like DCE, ensures high process reliability. This stability minimizes the risk of batch failures due to sensitive reaction parameters, thereby guaranteeing a consistent supply of high-purity intermediates. The broad substrate scope demonstrated in the patent implies that a single manufacturing platform can produce a wide array of derivatives, allowing for flexible production scheduling to meet fluctuating market demands without requiring extensive equipment changes.
- Scalability and Environmental Compliance: The mild thermal requirements (60 °C) and the absence of hazardous oxidizing agents make this process inherently safer and easier to scale from kilogram to tonne quantities. From an environmental compliance standpoint, the reduction in chemical waste aligns with increasingly stringent global regulations on industrial emissions. The high selectivity of the reaction reduces the burden on wastewater treatment facilities, positioning manufacturers who adopt this technology as leaders in sustainable chemical production.
Frequently Asked Questions (FAQ)
Understanding the technical nuances of this new synthetic route is essential for stakeholders evaluating its integration into existing production lines. The following questions address common inquiries regarding catalyst selection, substrate compatibility, and product isolation. These answers are derived directly from the experimental data and technical disclosures within the patent documentation, providing a reliable basis for decision-making.
Q: What are the optimal reaction conditions for this Rhodium-catalyzed coupling?
A: The optimal conditions involve using [RuCp*Cl2]2 as the catalyst with AgSbF6 as an additive in 1,2-dichloroethane (DCE) solvent at 60 °C for 24 hours, utilizing pivalic acid as a crucial promoter.
Q: Does this method require pre-functionalized substrates?
A: No, a key advantage of this protocol is that it utilizes direct C-H bond activation, eliminating the need for pre-functionalization such as halogenation, thereby improving atom economy.
Q: Can the resulting isoquinoline esters be further derivatized?
A: Yes, the ester group on the isoquinoline ring can be readily reduced to alcohols using reagents like LiAlH4, allowing for diverse downstream functionalization for drug discovery.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 2-(3-isoquinolyl)-propionic Acid Ethyl Ester Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of advanced C-H activation technologies in modern drug synthesis. Our team of expert process chemists has extensively evaluated the scalability of rhodium-catalyzed pathways, ensuring that we can deliver these complex pharmaceutical intermediates with the highest standards of quality and consistency. We possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, leveraging our state-of-the-art facilities to maintain stringent purity specifications. Our rigorous QC labs employ advanced analytical techniques to verify the identity and purity of every batch, ensuring that our clients receive materials that meet the exacting requirements of global regulatory bodies.
We invite you to collaborate with us to optimize your supply chain for isoquinoline-based APIs. By partnering with our technical procurement team, you can request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We encourage you to contact us today to obtain specific COA data and route feasibility assessments, allowing you to make informed decisions that drive efficiency and innovation in your drug development programs.
