Advanced Synthesis and Isomer Separation of Carboxyl-tetrachloro-fluorescein for Industrial Scale
Advanced Synthesis and Isomer Separation of Carboxyl-tetrachloro-fluorescein for Industrial Scale
The landscape of fluorescent dye manufacturing is undergoing a significant transformation driven by the urgent demand for high-purity reagents in DNA sequencing and molecular biology. Patent CN114736213A introduces a groundbreaking preparation method for carboxyl-tetrachloro-fluorescein, specifically addressing the longstanding bottlenecks associated with isomer separation and industrial scalability. This technology leverages a direct cyclization reaction followed by a novel recrystallization and acid-induced precipitation strategy to isolate high-purity 5-carboxyl-tetrachloro-fluorescein and 6-carboxyl-tetrachloro-fluorescein. For R&D directors and procurement managers seeking a reliable pharmaceutical intermediate supplier, this patent represents a pivotal shift away from labor-intensive chromatography towards robust, cost-effective chemical engineering solutions that ensure consistent supply chains for critical diagnostic reagents.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of tetrachloro-fluorescein derivatives has been plagued by inefficient routes that struggle to meet the rigorous purity standards required for modern biotechnology applications. The classical direct condensation method, often utilizing Lewis acids like zinc bromide (ZnBr2), necessitates harsh reaction conditions involving temperatures as high as 180°C or even melting states, which frequently results in low yields and significant byproduct formation. Furthermore, this route generates a mixture of 5-position and 6-position isomers that possess nearly identical physical and chemical properties, making their separation exceptionally difficult without resorting to preparative silica gel column chromatography. As illustrated in the reaction scheme below, the reliance on HPLC purification for isomer separation severely restricts production capacity to merely gram or milligram scales, rendering it economically unviable for industrial batch preparation.
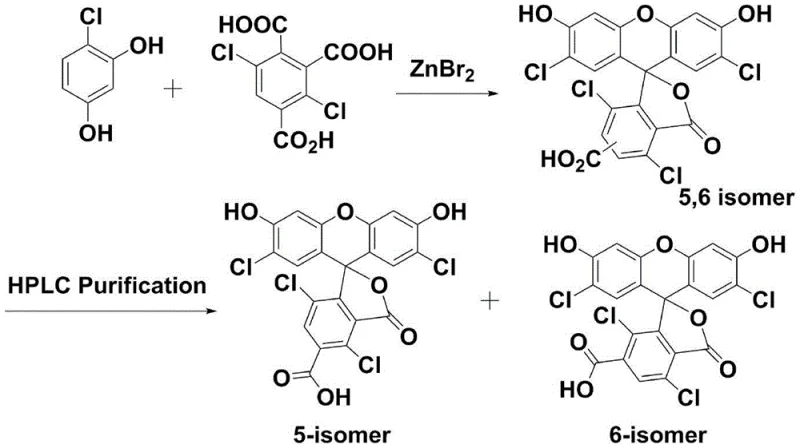
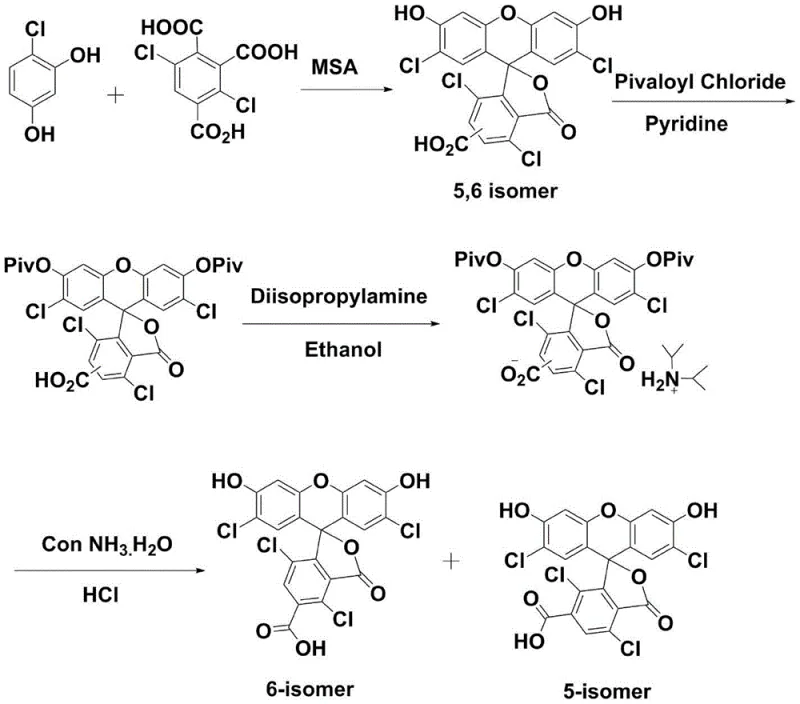
In an attempt to mitigate these issues, stepwise synthesis methods were developed, such as those utilizing methanesulfonic acid (MSA) followed by complex protection and deprotection sequences involving pivaloyl chloride and diisopropylamine. While this approach improved the initial cyclization yield to approximately 79%, it introduced a myriad of additional processing steps that complicated the workflow and increased material costs. The resulting isomer mixture still required resolution through salt formation and solvent differentiation, yet the final HPLC purity often plateaued around 90%, falling short of the >98% purity demanded by high-end sequencing applications. The intricate multi-step nature of this pathway, depicted in the following diagram, highlights the excessive operational burden and the inherent difficulty in scaling such fragile chemical transformations to a commercial level.
The Novel Approach
The innovative methodology disclosed in the patent fundamentally reimagines the production workflow by eliminating the need for hydroxyl protection and column chromatography entirely. By employing parachlororesorcinol and dichlorotrimellitic anhydride as raw materials in the presence of a protonic acid catalyst, the process achieves direct cyclization with high conversion rates exceeding 99%. The crude 5,6-isomer mixture is subsequently purified through a straightforward recrystallization technique using organic solvents like ethyl acetate and n-hexane, which effectively removes impurities without the massive solvent consumption associated with chromatography. This streamlined approach not only simplifies the equipment requirements but also drastically enhances the atom economy, making it uniquely suitable for the commercial scale-up of complex fluorescent dyes needed by the global diagnostics industry.
Mechanistic Insights into Protonic Acid Catalyzed Cyclization
The core of this technological breakthrough lies in the precise control of the electrophilic aromatic substitution reactions facilitated by strong protonic acids such as methanesulfonic acid or trifluoromethanesulfonic acid. Unlike Lewis acids which can be corrosive and difficult to remove, protonic acids promote the formation of the xanthene skeleton through a mechanism that favors the generation of the 5,6-carboxyl-tetrachloro-fluorescein backbone with minimal side reactions. The rigid planar structure of the resulting fluorescein derivative is critical for its optical properties, ensuring long wavelength absorption and emission in the visible light region while maintaining resistance to photobleaching. By optimizing the feeding ratio of raw materials to between 2.05:1 and 2.5:1, the reaction kinetics are tuned to maximize the formation of the desired isomer mixture while suppressing the formation of polymeric byproducts that typically contaminate the crude product in traditional syntheses.
Furthermore, the separation mechanism exploits subtle differences in the solubility profiles of the 5-isomer and 6-isomer under specific acidic conditions in organic media. When the purified 5,6-mixture is dissolved in a solvent like acetonitrile and treated with an acid solution at controlled temperatures, the system undergoes a phase transition where one isomer preferentially precipitates as a yellow solid while the other remains in the filtrate. This phenomenon allows for the isolation of the 5-carboxyl-tetrachloro-fluorescein with an HPLC purity of 98.97% and the 6-isomer with 98.41% purity simply through filtration and washing. This solubility-driven resolution is a masterclass in process chemistry, replacing expensive resin-based separations with thermodynamic control, thereby ensuring that the final high-purity carboxyl-tetrachloro-fluorescein meets the stringent specifications required for sensitive DNA labeling applications.
How to Synthesize Carboxyl-tetrachloro-fluorescein Efficiently
The implementation of this synthesis route requires careful attention to temperature control and solvent selection to ensure optimal yield and purity throughout the three main stages of production. The process begins with the cyclization reaction in a glass-lined reactor under nitrogen protection, followed by a recrystallization step to upgrade the crude material, and concludes with the critical isomer separation via acid precipitation. Operators must adhere strictly to the cooling profiles and stirring times specified in the patent to maximize the crystallization efficiency of each isomer. For a detailed breakdown of the standardized operating procedures, including specific temperatures, stirring durations, and solvent volumes, please refer to the technical guide below.
- Conduct direct cyclization of parachlororesorcinol and dichlorotrimellitic anhydride using methanesulfonic acid at 160°C to form the crude 5,6-isomer mixture.
- Purify the crude mixture via recrystallization in ethyl acetate and n-hexane to obtain high-purity 5,6-carboxyl-tetrachloro-fluorescein.
- Separate the 5-isomer and 6-isomer by exploiting solubility differences in acetonitrile with trifluoromethanesulfonic acid addition and temperature control.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this novel preparation method offers transformative benefits that directly impact the bottom line and operational resilience. By completely eliminating the need for silica gel column chromatography and preparative HPLC, the process removes a major bottleneck that has historically limited the availability of these critical intermediates to small research quantities. The removal of these purification steps translates to a drastic reduction in solvent consumption and waste generation, aligning with modern environmental compliance standards while simultaneously lowering the variable costs associated with raw material usage. This efficiency gain ensures that manufacturers can respond more agilely to market demand fluctuations without being constrained by the slow throughput of traditional purification technologies.
- Cost Reduction in Manufacturing: The elimination of expensive protecting groups like pivaloyl chloride and the associated reagents for deprotection significantly lowers the raw material bill of materials. Additionally, avoiding the use of Lewis acids such as ZnBr2 reduces the need for specialized corrosion-resistant equipment and extensive wastewater treatment for heavy metal removal. These cumulative savings allow for a substantial reduction in the overall cost of goods sold, making high-purity fluorescein derivatives more accessible for large-scale diagnostic kit production without compromising on quality standards.
- Enhanced Supply Chain Reliability: The simplified workflow, which relies on readily available commodity chemicals like methanesulfonic acid and common organic solvents, mitigates the risk of supply disruptions associated with specialty reagents. The robustness of the direct cyclization method ensures consistent batch-to-batch reproducibility, which is critical for maintaining the quality assurance protocols of downstream pharmaceutical customers. This reliability enables supply chain planners to forecast inventory levels with greater accuracy and reduce the safety stock requirements typically needed to buffer against the variability of complex multi-step syntheses.
- Scalability and Environmental Compliance: The process is designed for easy translation from laboratory benchtop to multi-ton industrial reactors, as it avoids the thermal hazards associated with melting-point reactions and the handling of pyrophoric catalysts. The reduction in solvent volume and the absence of silica waste streams simplify the environmental permitting process and lower the operational overhead related to hazardous waste disposal. This scalability ensures that the production of high-purity carboxyl-tetrachloro-fluorescein can be expanded rapidly to meet the growing global demand for next-generation sequencing reagents and fluorescent probes.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production and application of these advanced fluorescent intermediates. The answers are derived directly from the experimental data and technical disclosures within the patent documentation to provide accurate guidance for potential partners. Understanding these details is essential for evaluating the feasibility of integrating this technology into existing manufacturing lines or sourcing strategies.
Q: Why is the separation of 5-TET and 6-TET isomers challenging in traditional methods?
A: Traditional methods rely on silica gel column chromatography due to the similar physical and chemical properties of the isomers, which is costly, difficult to scale, and limits production to gram levels.
Q: How does the new protonic acid method improve purity compared to stepwise synthesis?
A: The new method avoids complex protection and deprotection steps, utilizing direct cyclization and recrystallization to achieve HPLC purity exceeding 98%, compared to roughly 90% in older stepwise routes.
Q: What are the key advantages for large-scale manufacturing of these fluorescent dyes?
A: The process eliminates expensive column chromatography and harsh Lewis acids like ZnBr2, significantly reducing equipment requirements, solvent waste, and operational complexity for ton-scale production.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Carboxyl-tetrachloro-fluorescein Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-performance fluorescent dyes play in advancing genomic research and diagnostic capabilities. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that we can meet the rigorous volume requirements of global life science companies. We are committed to delivering products with stringent purity specifications, supported by our rigorous QC labs that utilize state-of-the-art analytical instrumentation to verify every batch against the highest industry standards. Our capability to implement the advanced protonic acid catalyzed cyclization and recrystallization techniques described in recent patents positions us as a leader in the supply of complex pharmaceutical intermediates.
We invite you to collaborate with us to optimize your supply chain for fluorescein derivatives and achieve significant operational efficiencies. Please contact our technical procurement team to request a Customized Cost-Saving Analysis tailored to your specific volume needs. We are ready to provide specific COA data and route feasibility assessments to demonstrate how our manufacturing expertise can support your R&D and commercial goals with reliable, high-quality chemical solutions.
