Advanced Synthesis of Tebipenem Pivoxil Impurity I for Pharmaceutical Quality Control
Introduction to Advanced Impurity Profiling in Carbapenems
The rigorous demand for pharmaceutical quality control has driven significant innovation in the synthesis of reference standards for complex antibiotics. Patent CN102584812B introduces a groundbreaking preparation method for Tebipenem Pivoxil impurities, specifically targeting the open-ring degradation products that are critical for safety assessments. As oral carbapenems like Tebipenem Pivoxil gain traction for treating resistant bacterial strains, the ability to accurately quantify degradation products becomes paramount for regulatory compliance. This patent addresses a major bottleneck in the industry by providing a route that eliminates the need for sophisticated freeze-drying and column chromatography, traditionally required to handle these unstable beta-lactam derivatives. By leveraging controlled hydrolysis and strategic crystallization, manufacturers can now access high-purity reference substances that are essential for validating the quality of bulk drug substances.
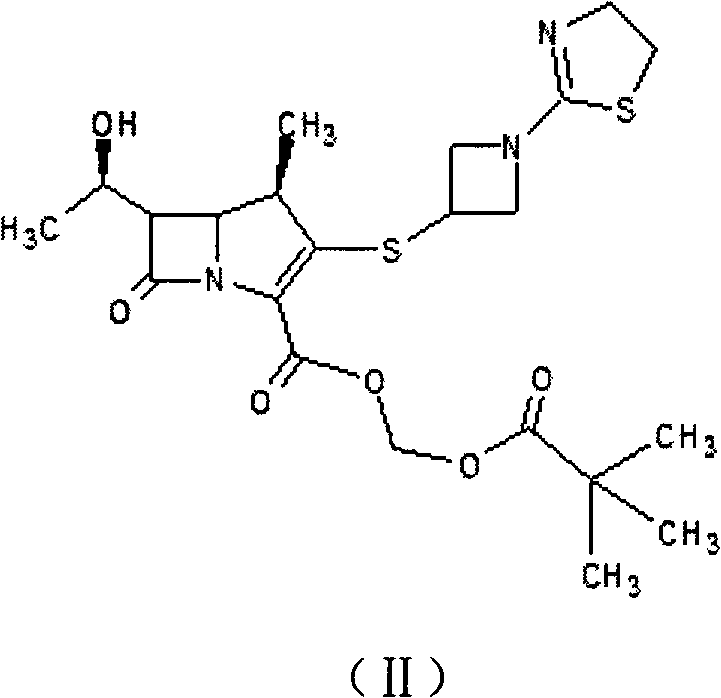
The structural integrity of carbapenems is notoriously fragile, particularly in acidic or alkaline environments where the beta-lactam ring is prone to opening. This vulnerability, while a challenge for drug stability, offers a mechanistic pathway for generating specific impurities needed for analytical method development. The patent outlines a process where the parent compound is subjected to mild hydrolytic conditions to selectively generate Impurity I, a key degradation product. This approach not only simplifies the supply chain for reference materials but also ensures that the impurities produced are chemically identical to those formed during the actual shelf-life of the pharmaceutical product, thereby enhancing the reliability of stability studies.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the isolation of beta-lactam impurities has been a resource-intensive endeavor plagued by low yields and high operational costs. Conventional workflows typically necessitate the use of preparative high-performance liquid chromatography (HPLC) or flash column chromatography to separate the target impurity from the reaction matrix. These techniques are not only expensive due to the consumption of large volumes of high-grade solvents and stationary phases but are also difficult to scale beyond gram quantities. Furthermore, the final isolation step often requires lyophilization (freeze-drying) to remove residual solvents without thermally degrading the sensitive beta-lactam core. This reliance on specialized equipment creates a significant barrier to entry for many chemical suppliers and results in long lead times for obtaining certified reference materials.
The Novel Approach
In stark contrast, the methodology described in CN102584812B utilizes a clever manipulation of solubility and pH to achieve purification without chromatography. The process begins with a controlled hydrolysis of Tebipenem, followed by a precise neutralization step that exploits the amphoteric nature of the product. By adjusting the pH to a narrow window between 4 and 6, the process ensures the product remains in its free acid form rather than converting to a salt, which is crucial for accurate molecular weight determination. The subsequent addition of acetone induces a salting-out effect, separating inorganic byproducts from the organic phase. This is followed by a recrystallization step using isopropyl ether, which yields a dry, free-flowing solid with purity exceeding 97%. 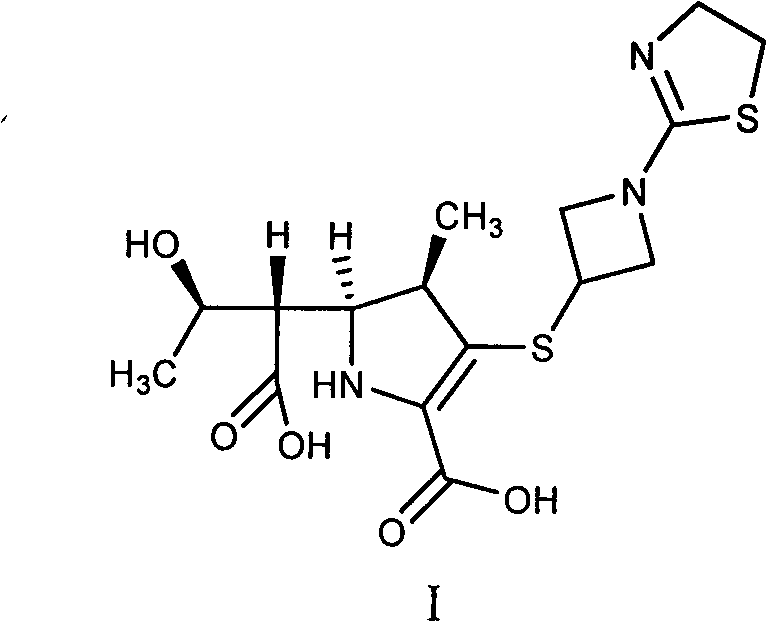 This elimination of chromatographic purification represents a paradigm shift in how reference standards are manufactured, drastically reducing both capital expenditure and operational complexity.
This elimination of chromatographic purification represents a paradigm shift in how reference standards are manufactured, drastically reducing both capital expenditure and operational complexity.
Mechanistic Insights into Controlled Hydrolytic Ring Opening
The core chemical transformation in this process is the nucleophilic attack on the beta-lactam carbonyl, leading to ring opening. Under the specified conditions of 2N NaOH or HCl at low temperatures (-5 to 10°C), the hydroxide or hydronium ions catalyze the cleavage of the strained four-membered ring. The low temperature is a critical kinetic control parameter; it slows down the reaction rate sufficiently to prevent over-degradation or the hydrolysis of the pivotal ester side chain, which would lead to different impurities like P9. The reaction is quenched rapidly by neutralizing the pH, effectively stopping the hydrolysis at the desired stage. This precision allows for the selective generation of the open-ring acid (Impurity I) while minimizing the formation of polymeric byproducts that often complicate carbapenem chemistry.
Following the reaction, the purification mechanism relies on the differential solubility of the organic product versus inorganic salts in mixed solvent systems. The addition of acetone to the aqueous reaction mixture reduces the dielectric constant of the solvent, causing the inorganic salts (NaCl) to precipitate out while keeping the organic impurity in solution or as a sticky solid that can be separated. The final crystallization from ethanol/methanol and isopropyl ether leverages the poor solubility of the zwitterionic open-ring structure in non-polar ethers. This drives the formation of a highly ordered crystal lattice, excluding remaining trace impurities and solvent molecules. The result is a thermodynamically stable solid form that does not require the energy-intensive removal of water via freeze-drying, ensuring the material is ready for immediate analytical use.
How to Synthesize Tebipenem Pivoxil Impurity I Efficiently
The synthesis protocol detailed in the patent offers a robust framework for producing this critical reference standard with high reproducibility. The procedure is designed to be scalable, moving away from the artisanal nature of column chromatography towards a more unit-operation-based chemical manufacturing process. Key to success is the strict adherence to temperature profiles and pH endpoints, which dictate the speciation of the molecule. Operators must ensure that the neutralization step hits the target pH of 5.6 precisely to avoid salt formation, which would alter the physical properties of the final product. The detailed standardized synthesis steps below outline the exact sequence of reagent addition and phase separation required to achieve the reported purity levels.
- React Tebipenem with 2N NaOH or HCl solution at -5 to 10°C for 10-30 minutes to initiate ring opening.
- Neutralize the reaction mixture to pH 4-6 using 2N HCl or NaOH, then add acetone to precipitate salts and separate the supernatant.
- Dissolve the residue in ethanol or methanol, filter, and induce crystallization by adding isopropyl ether at low temperatures (-20 to 0°C).
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this synthetic route offers tangible benefits in terms of cost structure and supply reliability. The most significant advantage lies in the drastic simplification of the downstream processing train. By removing the requirement for preparative chromatography columns and freeze-dryers, the capital intensity of the manufacturing process is significantly lowered. This reduction in equipment dependency translates directly into lower overhead costs per kilogram of product, allowing for more competitive pricing models for high-value reference standards. Additionally, the use of commodity solvents like acetone, ethanol, and isopropyl ether ensures that raw material sourcing is straightforward and immune to the supply chain disruptions often associated with specialized chromatographic resins.
- Cost Reduction in Manufacturing: The elimination of column chromatography removes one of the most expensive steps in fine chemical purification. Chromatographic media are costly consumables that require frequent replacement and generate substantial hazardous waste. By replacing this with a crystallization-based purification, the process achieves a substantial reduction in variable costs. Furthermore, the avoidance of lyophilization saves significant energy costs, as freeze-drying is an extremely energy-intensive unit operation. These cumulative savings allow for a more efficient allocation of resources, potentially lowering the total cost of ownership for pharmaceutical companies sourcing these critical quality control materials.
- Enhanced Supply Chain Reliability: Traditional methods for isolating beta-lactam impurities are often limited by batch size due to the capacity constraints of chromatography columns. This new method, relying on reactor volume and crystallization tanks, is inherently more scalable. It allows manufacturers to produce larger batches in a single run, thereby reducing the frequency of production campaigns and mitigating the risk of stockouts. The robustness of the process, with its wide operating windows for temperature and pH, also ensures consistent batch-to-batch quality, reducing the likelihood of failed quality control tests that could delay shipments to end-users.
- Scalability and Environmental Compliance: From an environmental perspective, this process generates less hazardous waste compared to traditional chromatographic methods. The solvents used are common and easily recoverable through distillation, facilitating a closed-loop solvent recovery system that aligns with green chemistry principles. The absence of silica gel or polymer-based stationary phases means there is no solid waste disposal issue related to spent chromatography media. This streamlined waste profile simplifies regulatory compliance and reduces the environmental footprint of the manufacturing facility, a key metric for modern sustainable supply chains.
Frequently Asked Questions (FAQ)
The following questions address common technical inquiries regarding the implementation and application of this synthesis method. Understanding the nuances of pH control and solvent selection is vital for laboratories aiming to replicate this process for internal reference standard production. The answers provided are derived directly from the experimental data and claims within the patent documentation, ensuring technical accuracy.
Q: Why is pH control critical in the synthesis of Tebipenem Impurity I?
A: Precise pH control between 4 and 6 is essential because the target molecule contains both acidic and basic centers. Deviating from this range can lead to the formation of hydrochloride salts or sodium salts, altering the molecular weight and preventing the isolation of the pure free acid form required for reference standards.
Q: How does this method improve upon traditional purification techniques?
A: Traditional methods often rely on expensive freeze-drying equipment and complex column chromatography to isolate unstable beta-lactam impurities. This patented process utilizes a simple salting-out and recrystallization technique using common solvents like acetone and isopropyl ether, significantly reducing equipment costs and operational complexity while achieving purities above 97%.
Q: What are the optimal temperature conditions for this reaction?
A: The reaction requires strict temperature control, typically between -5°C and 10°C, to prevent excessive degradation or the formation of secondary impurities. Similarly, the crystallization step is optimized at temperatures between -20°C and 0°C to maximize yield and ensure the formation of a dry, stable solid product without the need for lyophilization.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Tebipenem Pivoxil Impurity Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-quality impurities play in ensuring the safety and efficacy of life-saving antibiotics. Our technical team has extensively analyzed the pathways described in CN102584812B and possesses the expertise to scale this chemistry from laboratory benchtops to commercial production. We offer extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your supply of reference standards is never interrupted. Our facilities are equipped with rigorous QC labs capable of verifying stringent purity specifications, guaranteeing that every batch of Tebipenem Pivoxil Impurity I meets the exacting standards required for pharmaceutical regulatory filings.
We invite you to collaborate with us to optimize your supply chain for carbapenem intermediates. Our team is prepared to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements, demonstrating how our streamlined manufacturing processes can reduce your overall procurement costs. Please contact our technical procurement team today to request specific COA data and route feasibility assessments. Let us partner with you to secure a reliable, high-quality source of critical pharmaceutical impurities that supports your commitment to global health.
