Advanced Synthesis of 3-Cyano-4-Halogenated Quinoline Derivatives for Commercial API Production
Advanced Synthesis of 3-Cyano-4-Halogenated Quinoline Derivatives for Commercial API Production
The pharmaceutical industry's relentless pursuit of effective treatments for chronic myelocytic leukemia (CML) has placed a premium on the efficient synthesis of key kinase inhibitor intermediates. Patent CN102070524A introduces a transformative methodology for preparing 3-cyano-4-halogenated quinoline derivatives, which serve as critical building blocks for next-generation tyrosine kinase inhibitors. This novel approach addresses the longstanding challenges of low yields and difficult purification associated with traditional routes, offering a robust pathway for the commercial production of high-purity intermediates. By leveraging a streamlined six-step sequence that includes condensation, thermal cyclization, and strategic halogenation, this technology enables manufacturers to achieve superior atom economy and operational simplicity. For R&D directors and supply chain leaders, understanding the nuances of this patented process is essential for securing a competitive advantage in the oncology drug market.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of 3-cyano-4-halogenated quinoline derivatives has been plagued by significant inefficiencies that hinder large-scale production. As documented in prior art such as J. Med. Chem. (2004), earlier routes often relied on cyclization steps that resulted in excessively low yields and generated substantial amounts of difficult-to-remove byproducts. These impurities not only complicate downstream purification but also drastically reduce the overall throughput of the manufacturing line, leading to increased waste and higher operational costs. Furthermore, alternative methods reported in patents like WO2005047259 necessitated the use of expensive activated carboxylic acid reagents, such as carbonyldiimidazole (CDI), which inflate the raw material costs and introduce supply chain vulnerabilities. The reliance on such costly reagents makes these conventional processes economically unviable for the mass production required to meet global demand for affordable cancer therapies.
The Novel Approach
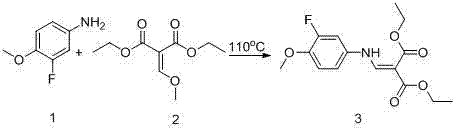
In stark contrast, the method disclosed in CN102070524A revolutionizes the synthesis landscape by employing a series of mild yet highly effective reaction conditions that maximize yield and purity. The process initiates with a condensation reaction between aniline derivatives and activated methylene compounds under controlled heating, establishing a stable foundation for subsequent transformations. Crucially, the cyclization step utilizes high-boiling solvents like thermal oil or phenyl ether derivatives at elevated temperatures, facilitating efficient ring closure without the need for exotic catalysts. This is followed by a clever dual-function step where thionyl chloride simultaneously effects carboxylic acid halogenation and ring chlorination, thereby consolidating two synthetic operations into one. This consolidation not only reduces the number of unit operations but also minimizes solvent usage and waste generation, presenting a clear path toward greener and more cost-effective manufacturing.
Mechanistic Insights into Thermal Cyclization and Halogenation
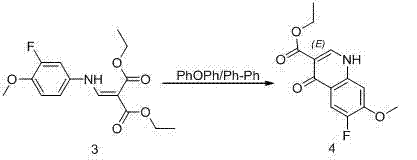
The core of this synthetic breakthrough lies in the mechanistic efficiency of the thermal cyclization and the subsequent halogenation steps. During the cyclization phase, the enamine intermediate undergoes an intramolecular nucleophilic attack facilitated by the high thermal energy provided by the solvent system, typically maintained around 260°C. This thermal activation overcomes the kinetic barriers associated with forming the quinoline core, ensuring that the reaction proceeds to completion with minimal formation of regioisomers or polymeric byproducts. The use of thermal oil or biphenyl mixtures as the reaction medium provides a stable thermal environment that prevents localized overheating, which is critical for maintaining the integrity of the sensitive cyano and ester functionalities present in the molecule. This precise control over reaction thermodynamics is what allows the process to achieve yields exceeding 90% in the cyclization step, a marked improvement over the sluggish conversions seen in older methodologies.
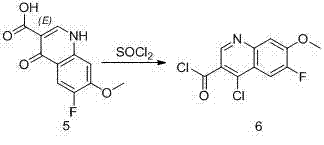
Furthermore, the halogenation strategy employed in this patent demonstrates exceptional chemical ingenuity by utilizing thionyl chloride (SOCl2) to perform a dual transformation. In this single operational step, the carboxylic acid moiety is converted into a reactive acid chloride, while simultaneously, a chlorine atom is introduced onto the quinoline ring at the 4-position. This tandem reaction mechanism eliminates the need for separate halogenation and activation steps, significantly reducing the processing time and the accumulation of intermediate impurities. The resulting acid chloride is then seamlessly subjected to ammonolysis to form the amide, which is subsequently dehydrated to yield the final nitrile product. This cohesive flow of chemical transformations ensures that the impurity profile remains clean throughout the synthesis, ultimately delivering a final product with purity levels suitable for direct use in API synthesis without extensive recrystallization.
How to Synthesize 3-Cyano-4-Halogenated Quinoline Derivatives Efficiently
The implementation of this synthesis route requires careful attention to temperature control and reagent stoichiometry to replicate the high yields reported in the patent. The process begins with the condensation of the aniline precursor, followed by the critical thermal cyclization which demands robust heating equipment capable of sustaining temperatures up to 260°C safely. Subsequent steps involve standard aqueous workups and solvent exchanges that are well within the capabilities of modern multipurpose chemical plants. For a detailed breakdown of the specific operating parameters, stoichiometric ratios, and isolation techniques required to execute this synthesis successfully, please refer to the standardized guide below.
- Condense aniline derivatives with activated methylene compounds at 110°C to form the enamine intermediate.
- Perform thermal cyclization using thermal oil or phenyl ether solvents at 260°C to construct the quinoline core.
- Execute simultaneous ring chlorination and acid chloride formation using thionyl chloride, followed by ammonolysis and dehydration.
Commercial Advantages for Procurement and Supply Chain Teams
From a procurement and supply chain perspective, the adoption of this patented methodology offers profound strategic benefits that extend beyond mere technical feasibility. The elimination of expensive coupling reagents like CDI fundamentally alters the cost structure of the intermediate, making the final API more price-competitive in the global market. Additionally, the use of commodity chemicals such as thionyl chloride and thermal oil ensures that raw material sourcing is resilient against market fluctuations, providing a stable foundation for long-term production planning. The high yields observed at each stage of the synthesis mean that less starting material is required to produce the same amount of final product, effectively lowering the cost of goods sold (COGS) and reducing the environmental footprint associated with waste disposal.
- Cost Reduction in Manufacturing: The strategic replacement of high-cost activated reagents with inexpensive halogenating agents like thionyl chloride results in a drastic reduction in raw material expenditures. By consolidating the ring chlorination and acid activation into a single step, the process also reduces labor costs and utility consumption associated with running additional reaction vessels. This streamlined approach minimizes the loss of valuable intermediates during transfer and purification, ensuring that the theoretical yield is closely matched by the actual isolated yield, which translates directly to improved profit margins for the manufacturer.
- Enhanced Supply Chain Reliability: The reliance on widely available bulk chemicals rather than specialized, low-volume reagents mitigates the risk of supply disruptions. Thermal oil and phenyl ether solvents are standard industrial commodities with established global supply chains, ensuring that production schedules can be maintained without delay. Furthermore, the robustness of the reaction conditions means that the process is less sensitive to minor variations in raw material quality, allowing for greater flexibility in vendor selection and reducing the need for stringent incoming quality control testing that can slow down production intake.
- Scalability and Environmental Compliance: The simplicity of the workup procedures, which primarily involve filtration and pH adjustment, facilitates easy scale-up from pilot plant to commercial tonnage production. The reduction in the number of synthetic steps and the avoidance of heavy metal catalysts simplify the waste stream, making it easier to treat effluents and comply with increasingly strict environmental regulations. This environmental compatibility not only reduces disposal costs but also enhances the corporate sustainability profile of the manufacturing entity, a factor of growing importance to downstream pharmaceutical partners.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis route. These answers are derived directly from the experimental data and beneficial effects described in the patent documentation, providing clarity on the operational realities of the process. Understanding these details is crucial for technical teams evaluating the feasibility of adopting this method for their own production lines.
Q: How does this method improve upon previous synthesis routes for quinoline derivatives?
A: Unlike prior art methods that suffer from low yields in cyclization or require expensive reagents like CDI, this patented process utilizes cost-effective thermal cyclization and thionyl chloride, achieving significantly higher total yields and purity suitable for industrial scale-up.
Q: What are the critical reaction conditions for the cyclization step?
A: The cyclization step requires high-temperature conditions, specifically heating the intermediate in solvents like phenyl ether, biphenyl, or thermal oil to approximately 260°C, which ensures efficient ring closure with minimal byproduct formation.
Q: Is this process suitable for large-scale manufacturing of oncology intermediates?
A: Yes, the process is explicitly designed for industrial application, utilizing common solvents and reagents while avoiding complex purification steps, thereby ensuring consistent quality and supply continuity for pharmaceutical production.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 3-Cyano-4-Halogenated Quinoline Derivative Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-quality intermediates play in the development of life-saving oncology medications. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that we can meet the rigorous demands of global pharmaceutical supply chains. We are committed to delivering products with stringent purity specifications, supported by our rigorous QC labs that utilize state-of-the-art analytical instrumentation to verify every batch. Our expertise in handling complex heterocyclic chemistry allows us to optimize processes like the one described in CN102070524A, guaranteeing consistent quality and reliable delivery schedules for our partners.
We invite you to collaborate with us to leverage this advanced synthesis technology for your next project. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We are ready to provide specific COA data and comprehensive route feasibility assessments to demonstrate how our manufacturing capabilities can enhance your supply chain efficiency and reduce your overall production costs.
