Advanced Manufacturing of 2,6-Dichloro-3-Fluoro Acetophenone for Global Pharma Supply Chains
Advanced Manufacturing of 2,6-Dichloro-3-Fluoro Acetophenone for Global Pharma Supply Chains
The pharmaceutical industry continuously demands higher purity standards for key intermediates, particularly those serving as the backbone for oncology therapeutics like Crizotinib. A pivotal advancement in this domain is documented in patent CN111892488A, which outlines a robust method for efficiently synthesizing 2,6-dichloro-3-fluoro acetophenone. This specific intermediate is critical for the production of protein kinase inhibitors, yet historical manufacturing routes have been plagued by low purity profiles and complex purification burdens that inflate costs. The disclosed technology introduces a refined Friedel-Crafts acylation strategy that leverages a dual-solvent system to enhance regioselectivity and yield. By optimizing the molar ratios of Lewis acid catalysts and implementing a novel recrystallization protocol, this method directly addresses the longstanding issues of impurity carryover that compromise drug safety. For global procurement teams, this represents a shift towards more reliable supply chains capable of delivering high-purity pharmaceutical intermediates without the traditional bottlenecks.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of halogenated acetophenones has been fraught with significant technical hurdles that impact both economic viability and product quality. Conventional routes often suffer from poor regiocontrol during the electrophilic aromatic substitution, leading to complex mixtures of isomers that are notoriously difficult to separate. Furthermore, traditional workup procedures frequently involve harsh conditions or extensive chromatographic steps, which are impractical for large-scale industrial applications due to high solvent consumption and waste generation. The presence of residual metal catalysts and organic impurities in the final product poses a severe risk to the downstream synthesis of Active Pharmaceutical Ingredients (APIs), potentially affecting the bioavailability and toxicity profile of the final drug. These inefficiencies result in elevated production costs and extended lead times, creating volatility in the supply of critical oncology intermediates. Consequently, manufacturers have struggled to meet the stringent purity specifications required by regulatory bodies without incurring prohibitive expenses.
The Novel Approach
The methodology presented in patent CN111892488A offers a transformative solution by re-engineering the reaction environment to favor the desired product formation inherently. By employing a specific mixed solvent system comprising nitrobenzene and dichloromethane, the process creates an optimal polarity environment that stabilizes the reactive acylium species while suppressing side reactions. The strategic adjustment of the aluminum trichloride to substrate ratio ensures complete conversion while minimizing the formation of poly-acylated byproducts. Moreover, the introduction of a targeted recrystallization step using a defined ratio of low and high polarity solvents allows for the precise removal of trace impurities without the need for complex distillation columns alone. This streamlined approach not only simplifies the post-treatment workflow but also significantly enhances the overall yield and purity of the 2,6-dichloro-3-fluoro acetophenone. Such innovations are essential for achieving cost reduction in pharmaceutical intermediates manufacturing while maintaining the highest quality standards.
Mechanistic Insights into Friedel-Crafts Acylation
The core of this synthetic breakthrough lies in the nuanced manipulation of electrophilic aromatic substitution dynamics on a highly deactivated benzene ring. The substrate, 2,4-dichlorofluorobenzene, presents a challenging electronic landscape due to the electron-withdrawing nature of the halogen substituents, which typically retards the rate of acylation. However, the use of anhydrous aluminum trichloride in a nitrobenzene-rich medium generates a super-electrophilic acetyl cation complex capable of overcoming this deactivation barrier. The nitrobenzene acts not merely as a solvent but as a polar coordinator that helps solvate the Lewis acid-base complex, thereby increasing the effective concentration of the active electrophile. This mechanistic advantage ensures that the acetylation occurs selectively at the position ortho to the fluorine atom, driven by the specific directing effects and steric accessibility of the ring positions. Understanding this interaction is vital for R&D directors aiming to replicate or scale this high-purity OLED material or pharmaceutical precursor synthesis.
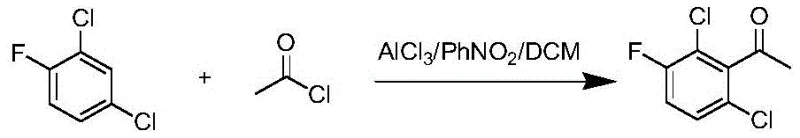
Controlling the impurity profile in such halogenated systems requires a deep understanding of the thermodynamic and kinetic factors governing the reaction pathway. The patent specifies a careful temperature control regime, initiating the reaction at room temperature to manage the exotherm and prevent thermal degradation or rearrangement of the product. Following the reaction, the quenching process with ice water is critical to hydrolyze the aluminum complexes without generating excessive heat that could promote hydrolysis of the sensitive ketone product. The subsequent purification via recrystallization exploits the differential solubility of the target ketone versus its isomers in a mixed solvent of dichloromethane and ethanol. This physical separation mechanism is far more efficient than chemical scavenging, ensuring that the final product meets the rigorous specifications needed for clinical applications. By mastering these mechanistic details, manufacturers can consistently produce commercial scale-up of complex pharmaceutical intermediates with minimal batch-to-batch variation.
How to Synthesize 2,6-Dichloro-3-Fluoro Acetophenone Efficiently
Implementing this synthesis route requires strict adherence to the optimized parameters regarding solvent ratios and reagent addition rates to ensure safety and reproducibility. The process begins with the preparation of the catalyst solution, where anhydrous aluminum trichloride is dispersed in the nitrobenzene/dichloromethane mixture before the slow introduction of acetyl chloride to form the active complex. Once the electrophile is generated, the 2,4-dichlorofluorobenzene is added dropwise to maintain thermal equilibrium, preventing runaway reactions that could compromise the structural integrity of the molecule. After the reaction reaches completion, indicated by the consumption of starting materials, the mixture is carefully quenched and extracted to isolate the crude organic layer. The detailed standardized synthesis steps below outline the precise operational protocols required to achieve the reported yields and purity levels.
- Prepare the catalyst system by dissolving anhydrous aluminum trichloride in a mixed solvent of nitrobenzene and dichloromethane, followed by the slow addition of acetyl chloride at room temperature.
- Introduce 2,4-dichlorofluorobenzene dissolved in dichloromethane to the reaction mixture, maintaining strict temperature control, and quench with ice water upon completion.
- Purify the crude product through fractional distillation followed by a specialized recrystallization process using a mixed organic solvent system to achieve high purity.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this novel synthesis route translates into tangible strategic advantages that extend beyond simple unit price considerations. The elimination of complex chromatographic purification steps significantly reduces the consumption of high-grade silica and vast quantities of elution solvents, which are major cost drivers in fine chemical manufacturing. Furthermore, the use of common industrial solvents like dichloromethane and nitrobenzene ensures that raw material sourcing remains stable and unaffected by niche supply shortages. The simplified workup procedure also shortens the overall cycle time per batch, allowing for increased throughput within existing reactor infrastructure without the need for capital-intensive equipment upgrades. These factors collectively contribute to a more resilient supply chain capable of meeting the fluctuating demands of the global oncology drug market. Ultimately, this process optimization supports reducing lead time for high-purity pharmaceutical intermediates while enhancing the overall reliability of the supplier base.
- Cost Reduction in Manufacturing: The primary economic benefit stems from the drastic simplification of the downstream processing workflow, which removes the need for expensive and time-consuming column chromatography. By relying on crystallization and distillation, the process minimizes solvent waste and reduces the energy load associated with solvent recovery systems. Additionally, the high selectivity of the reaction reduces the loss of valuable starting materials to byproduct formation, thereby improving the overall atom economy of the synthesis. These efficiencies allow for a significant reduction in the cost of goods sold (COGS), enabling more competitive pricing structures for long-term supply agreements. Such cost optimizations are critical for maintaining margins in the highly competitive generic and branded pharmaceutical sectors.
- Enhanced Supply Chain Reliability: The reliance on commodity chemicals and standard reaction conditions mitigates the risks associated with sourcing specialized reagents that may have limited availability. Since the process operates at near-ambient temperatures and uses robust catalysts, it is less susceptible to variations in utility supplies or minor equipment fluctuations. This robustness ensures consistent batch quality and on-time delivery performance, which are key metrics for supply chain heads managing just-in-time inventory models. Moreover, the scalability of the method means that production volumes can be ramped up quickly to accommodate sudden spikes in demand without compromising product integrity. This flexibility provides a crucial buffer against market volatility and ensures continuity of supply for critical drug manufacturing pipelines.
- Scalability and Environmental Compliance: From an environmental perspective, the reduction in solvent usage and waste generation aligns with increasingly stringent global regulations on chemical manufacturing emissions. The ability to recycle the mixed solvent system further enhances the sustainability profile of the process, reducing the carbon footprint associated with the production of this key intermediate. Scalability is inherently built into the design, as the exothermic nature of the reaction is well-managed by the dropwise addition protocol, making it safe to transfer from pilot plants to multi-ton reactors. This ease of scale-up facilitates the commercial production of complex pharmaceutical intermediates without the typical teething problems associated with process intensification. Consequently, partners can rely on a manufacturing process that is both environmentally responsible and industrially robust.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production and application of 2,6-dichloro-3-fluoro acetophenone. These insights are derived directly from the technical disclosures in patent CN111892488A and reflect the practical realities of industrial organic synthesis. Understanding these nuances helps stakeholders make informed decisions regarding vendor selection and process integration. The answers provided below clarify the specific advantages of this method over legacy technologies.
Q: What are the critical purity challenges in synthesizing 2,6-dichloro-3-fluoro acetophenone?
A: Traditional methods often struggle with regioselectivity and the removal of isomeric impurities, which can severely impact the safety and efficacy of the final Crizotinib API. The novel process addresses this through a specific solvent polarity balance and recrystallization protocol.
Q: How does the new solvent system improve reaction efficiency?
A: By utilizing a mixed solvent system of nitrobenzene and dichloromethane, the process stabilizes the acylium ion intermediate more effectively than single-solvent systems, leading to higher conversion rates and simplified downstream processing.
Q: Is this synthesis method suitable for large-scale industrial production?
A: Yes, the process avoids exotic reagents and extreme conditions, relying on standard industrial solvents and manageable temperature ranges (room temperature to 65°C), making it highly adaptable for metric-ton scale manufacturing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 2,6-Dichloro-3-Fluoro Acetophenone Supplier
At NINGBO INNO PHARMCHEM, we recognize that the successful development of next-generation kinase inhibitors depends heavily on the quality and consistency of key building blocks like 2,6-dichloro-3-fluoro acetophenone. Our technical team has extensively analyzed the patented routes to ensure that our manufacturing processes align with the highest standards of efficiency and purity. We possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that we can meet your volume requirements regardless of the project phase. Our facilities are equipped with rigorous QC labs and adhere to stringent purity specifications to guarantee that every batch meets the demanding criteria of the pharmaceutical industry. By leveraging our expertise in Friedel-Crafts chemistry and purification technologies, we deliver intermediates that facilitate smoother downstream synthesis for our clients.
We invite you to collaborate with us to optimize your supply chain and reduce your overall manufacturing costs through our advanced production capabilities. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume needs and quality requirements. We encourage you to contact us to request specific COA data and route feasibility assessments that demonstrate our commitment to excellence. Partnering with us ensures access to a reliable source of high-quality intermediates that support the rapid development and commercialization of life-saving therapies. Let us help you secure your supply chain with a partner dedicated to innovation and reliability.
