Scaling High-Optical Purity Chiral Alcohol Production with Engineered Carbonyl Reductase CgKR1
Introduction to Patent CN102618513B
The pharmaceutical industry continuously seeks robust methodologies for the synthesis of optically active chiral alcohols, which serve as critical building blocks for numerous blockbuster drugs. Patent CN102618513B introduces a groundbreaking advancement in this domain by disclosing a novel carbonyl reductase, designated as CgKR1, derived from Candida glabrata. This intellectual property represents a significant leap forward in biocatalytic engineering, specifically addressing the longstanding challenges of low substrate loading and high cofactor costs associated with traditional enzymatic reductions. The patent details not only the wild-type enzyme but also a series of rationally designed mutants that exhibit drastically improved catalytic efficiency and thermal stability. For R&D directors and procurement strategists, this technology offers a viable pathway to manufacture high-value intermediates for drugs such as Clopidogrel and various ACE inhibitors with superior economic and environmental profiles.
The core innovation lies in the enzyme's ability to catalyze the asymmetric reduction of prochiral carbonyl compounds under mild conditions without the necessity for excessive external cofactor supplementation. Unlike conventional chemical reduction methods that often rely on hazardous reducing agents and generate significant waste, this biocatalytic approach leverages the inherent stereoselectivity of the CgKR1 enzyme. The patent explicitly highlights the successful application of this system in producing (R)-methyl o-chloromandelate, (R)-ethyl 2-hydroxy-4-phenylbutyrate, and (R)-ethyl 4-chloro-3-hydroxybutyrate. These molecules are indispensable precursors in the synthesis of antiplatelet agents, antihypertensives, and metabolic supplements, respectively. By integrating this technology into existing production lines, manufacturers can achieve a reliable pharmaceutical intermediate supplier status while adhering to increasingly stringent green chemistry regulations.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial production of chiral alcohols has been plagued by significant technical and economic bottlenecks. Traditional chemical synthesis routes often employ stoichiometric amounts of metal hydrides, such as sodium borohydride or lithium aluminum hydride, which necessitate the use of chiral auxiliaries or resolution steps to achieve optical purity. These processes are not only costly due to the price of reagents but also generate substantial quantities of toxic waste, complicating disposal and increasing the overall environmental footprint. Furthermore, chemical methods frequently struggle to achieve enantiomeric excess (ee) values above 95% without multiple recrystallization steps, leading to yield losses. In the realm of biocatalysis, earlier generations of carbonyl reductases suffered from poor substrate tolerance, typically limiting reaction concentrations to below 20 g/L to maintain selectivity. Additionally, many prior art enzymes required the continuous addition of expensive nicotinamide cofactors like NADP+, which significantly inflated the operational expenditure (OpEx) for large-scale manufacturing.
The Novel Approach
The technology disclosed in CN102618513B fundamentally disrupts these limitations through protein engineering and process optimization. The inventors utilized site-directed mutagenesis to modify the active center and hydrophobic channels of the wild-type CgKR1 enzyme. Specific mutations, such as F92L, F94V, I99Y, and G174A, were introduced to enhance substrate binding affinity and thermal stability. The resulting quadruple mutant, CgKR1 F92L/F94V/I99Y/G174A, exhibits a catalytic activity up to 12 times higher than the wild type. Crucially, this novel approach enables reactions at substrate concentrations as high as 2.0 mol/L (approximately 412 g/L for certain substrates) while maintaining ee values exceeding 98%. Moreover, the process design incorporates a cofactor regeneration system using glucose dehydrogenase, allowing the reaction to proceed with minimal to zero external addition of NADP+. This eliminates a major cost driver and simplifies the downstream purification process, making it an ideal solution for cost reduction in API manufacturing.
Mechanistic Insights into CgKR1-Catalyzed Asymmetric Reduction
The mechanistic superiority of the CgKR1 system stems from precise structural modifications that optimize the enzyme-substrate interaction. The patent describes the active center as a spherical space near the substrate binding site, where specific amino acid residues dictate stereoselectivity. By substituting bulky phenylalanine residues at positions 92 and 94 with smaller leucine and valine residues, the steric hindrance within the hydrophobic channel is reduced. This modification facilitates easier access for bulky substrates like ethyl 2-carbonyl-4-phenylbutyrate, thereby explaining the dramatic increase in catalytic turnover. Simultaneously, the substitution of isoleucine at position 99 with tyrosine introduces polar interactions that enhance the soluble expression of the recombinant protein in E. coli hosts. These structural insights provide R&D teams with a clear understanding of how enzyme engineering can be leveraged to overcome mass transfer limitations often encountered in high-concentration biotransformations.
Furthermore, the reaction mechanism relies on a coupled enzyme system that ensures thermodynamic favorability and economic efficiency. The carbonyl reductase catalyzes the hydride transfer from the reduced cofactor NADPH to the prochiral ketone, generating the chiral alcohol and oxidized NADP+. To sustain this cycle without prohibitive costs, glucose dehydrogenase (GDH) is employed to regenerate NADPH from NADP+ using glucose as a sacrificial electron donor. This regeneration loop allows the cofactor to function catalytically rather than stoichiometrically. The patent data indicates that with this system, the concentration of added NADP+ can be reduced to as low as 0.1 mmol/L or even omitted entirely when using resting cells containing endogenous cofactors. This dual-enzyme strategy ensures high conversion rates (>99% for many substrates) and exceptional optical purity, effectively controlling the impurity profile by minimizing side reactions associated with non-enzymatic reduction.
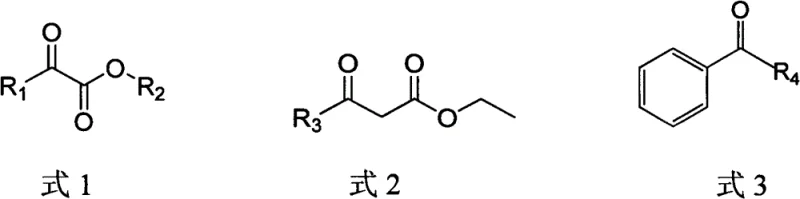
How to Synthesize Chiral Alcohol Intermediates Efficiently
The implementation of this biocatalytic route involves a streamlined workflow designed for industrial scalability. The process begins with the fermentation of recombinant E. coli strains harboring the optimized CgKR1 genes, followed by cell harvesting and optional lysis to obtain crude enzyme preparations. The actual biotransformation is conducted in an aqueous phosphate buffer system at a controlled pH of 5.5 to 7.0 and temperatures ranging from 20°C to 35°C. Glucose is fed into the system to drive the cofactor regeneration cycle, ensuring continuous reduction of the carbonyl substrate. Detailed standardized synthetic steps for the preparation of the recombinant strain and the execution of the reduction reaction are provided in the guide below.
- Prepare recombinant E. coli expressing CgKR1 mutants (e.g., F92L/F94V/I99Y/G174A) and cultivate in LB medium with IPTG induction.
- Conduct asymmetric reduction in phosphate buffer (pH 6.0) with glucose dehydrogenase for cofactor regeneration at 25°C.
- Extract the resulting chiral alcohol product using ethyl acetate and purify via distillation to achieve >98% ee.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of the CgKR1 technology translates into tangible strategic advantages beyond mere technical performance. The primary value proposition lies in the drastic simplification of the supply chain for critical reagents. By eliminating the dependency on stoichiometric amounts of expensive external cofactors, the variable cost of goods sold (COGS) is significantly reduced. This cost structure improvement is further amplified by the enzyme's ability to operate at high substrate concentrations. Higher concentration means that for a given production target, the required reactor volume is substantially smaller, leading to lower capital expenditure (CapEx) for equipment and reduced utility consumption for heating, cooling, and agitation. These factors collectively contribute to a more resilient and cost-effective manufacturing model.
- Cost Reduction in Manufacturing: The economic impact of this technology is profound due to the removal of costly input materials. Traditional biocatalytic processes often require the addition of grams of NADP+ per kilogram of product, which is financially unsustainable at scale. The CgKR1 system, utilizing an efficient glucose-driven regeneration cycle, reduces this requirement to trace levels. Additionally, the high catalytic activity of the mutants means less enzyme protein is needed to achieve the same throughput, lowering the fermentation burden. The combination of reduced reagent costs, smaller reactor footprints, and simplified downstream processing creates a compelling business case for switching from chemical to biological synthesis, driving substantial cost savings without compromising quality.
- Enhanced Supply Chain Reliability: Supply continuity is a critical metric for pharmaceutical production, and the CgKR1 platform enhances this through robustness. The engineered mutants exhibit improved thermal stability, with T50 values reaching up to 45.4°C, which allows the process to tolerate minor temperature fluctuations during large-scale operation without loss of activity. Furthermore, the enzyme is expressed in standard E. coli hosts using common plasmids like pET28a, ensuring that the biological catalyst can be produced reliably by any contract development and manufacturing organization (CDMO) with standard fermentation capabilities. This universality reduces the risk of supply bottlenecks associated with proprietary or difficult-to-culture organisms, ensuring a steady flow of high-purity pharmaceutical intermediates.
- Scalability and Environmental Compliance: As regulatory pressure mounts to reduce the environmental impact of chemical manufacturing, this biocatalytic route offers a clear compliance advantage. The process operates in aqueous media at mild temperatures, avoiding the use of volatile organic solvents and heavy metal catalysts typical of chemical reduction. The waste stream is primarily composed of biodegradable organic matter (gluconic acid from glucose oxidation), which is far easier and cheaper to treat than heavy metal sludge. The ease of scale-up is demonstrated by the patent's success in moving from milligram-scale screening to mole-level reactions without loss of selectivity. This scalability ensures that the technology can meet the demands of commercial scale-up of complex pharmaceutical intermediates, from pilot batches to multi-ton annual production.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of the CgKR1 biocatalytic system. These answers are derived directly from the experimental data and claims presented in patent CN102618513B, providing a factual basis for decision-making. Understanding these specifics helps stakeholders evaluate the feasibility of integrating this technology into their current manufacturing portfolios for chiral alcohol production.
Q: Does the CgKR1 process require expensive external cofactors?
A: No, when using resting cells or crude enzyme extracts, the process utilizes intracellular cofactors or a glucose dehydrogenase regeneration system, eliminating the need for costly external NADP+ addition.
Q: What is the substrate tolerance of the engineered CgKR1 mutants?
A: The engineered mutants, particularly CgKR1 F92L/F94V/I99Y/G174A, demonstrate exceptional substrate tolerance, supporting concentrations up to 2.0 mol/L while maintaining high optical purity.
Q: Which pharmaceutical intermediates can be synthesized using this technology?
A: This technology is specifically validated for key intermediates such as (R)-methyl o-chloromandelate for Clopidogrel, (R)-ethyl 2-hydroxy-4-phenylbutyrate for ACE inhibitors, and (R)-ethyl 4-chloro-3-hydroxybutyrate for L-carnitine.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Chiral Alcohol Intermediate Supplier
The technological potential of the CgKR1 enzyme system represents a paradigm shift in the synthesis of high-value chiral building blocks. NINGBO INNO PHARMCHEM stands at the forefront of this innovation, leveraging our extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production. Our state-of-the-art facilities are equipped with rigorous QC labs capable of verifying stringent purity specifications, ensuring that every batch of chiral alcohol intermediate meets the exacting standards required by global regulatory bodies. We understand that transitioning to a new biocatalytic route requires confidence in both the science and the supply partner, and we are committed to providing that assurance through transparent communication and proven execution.
We invite potential partners to engage with our technical procurement team to discuss how this technology can be tailored to your specific production needs. By requesting a Customized Cost-Saving Analysis, you can quantify the economic benefits of switching to our enzymatic process for your specific API intermediate. We encourage you to contact us to obtain specific COA data for our reference standards and to receive detailed route feasibility assessments. Let us collaborate to optimize your supply chain, reduce costs, and secure a sustainable future for your pharmaceutical manufacturing operations.
