Revolutionizing (S)-NBHP Production: High-Efficiency Biocatalysis for Commercial Scale-Up
The pharmaceutical industry is constantly seeking robust, scalable, and environmentally sustainable methods for producing chiral intermediates, particularly for oncology treatments. A significant breakthrough in this domain is detailed in patent CN110777125B, which discloses a highly efficient preparation method for heterocyclic drug intermediates, specifically focusing on the synthesis of (S)-N-Boc-3-hydroxypiperidine ((S)-NBHP). This compound serves as a critical chiral building block for Ibrutinib, a potent medication used in the treatment of lymphoma. The core innovation lies in the rational design of an alcohol dehydrogenase mutant, designated as Y127W, where the tyrosine at position 127 is mutated to tryptophan. This specific molecular modification fundamentally alters the enzyme's interaction with the substrate, enabling a dramatic increase in catalytic efficiency and stability. Unlike traditional biocatalytic methods that often struggle with low substrate tolerance and product inhibition, this novel mutant allows for operation in a single aqueous phase system without any organic cosolvents. The result is a process capable of achieving substrate concentrations as high as 600 g·L⁻¹ with conversion rates exceeding 99% within just 12 hours. For global procurement and R&D teams, this represents a paradigm shift towards greener, more cost-effective manufacturing of high-value pharmaceutical intermediates.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the biosynthesis of chiral piperidine derivatives like (S)-NBHP has been plagued by significant technical and economic bottlenecks that hinder industrial scale-up. Conventional enzymatic routes typically suffer from severe substrate and product inhibition, necessitating the use of low substrate concentrations, often below 100 g·L⁻¹, which drastically reduces reactor throughput and increases solvent usage per unit of product. To mitigate solubility issues and improve conversion, prior art methods frequently rely on the addition of organic cosolvents such as isopropanol, methanol, or ethyl caprylate. While these solvents can enhance substrate solubility, they often compromise enzyme stability and activity over time, leading to inconsistent batch quality. Furthermore, many existing processes require the continuous addition of expensive exogenous cofactors like NAD(P)H, or complex two-phase systems that complicate downstream separation and purification. These factors collectively inflate the cost of goods sold (COGS) and create substantial waste disposal challenges, making them less attractive for cost reduction in API manufacturing when compared to modern, engineered solutions.
The Novel Approach
The technology described in patent CN110777125B overcomes these historical limitations through a sophisticated protein engineering strategy combined with an optimized cofactor regeneration system. By mutating the 127th amino acid residue from tyrosine to tryptophan, the resulting Y127W mutant exhibits a remarkably enhanced affinity for the bulky N-Boc protected substrate. This structural change effectively alleviates product inhibition, allowing the reaction to proceed efficiently in a pure water phase without the need for harmful organic additives. The process couples the Y127W alcohol dehydrogenase with glucose dehydrogenase (BmGDH), creating a self-sustaining cycle that regenerates the necessary cofactor in situ using inexpensive glucose. This eliminates the need for costly external cofactor supplementation. The outcome is a streamlined, single-pot reaction that achieves unprecedented substrate loading capacities of up to 600 g·L⁻¹. This high-density biotransformation not only maximizes space-time yield but also simplifies the workup procedure, as there are no organic solvents to remove before extraction, thereby offering a compelling solution for the commercial scale-up of complex pharmaceutical intermediates.
Mechanistic Insights into Y127W Mutant Catalysis
The superior performance of the Y127W mutant is rooted in precise stereoelectronic modifications at the enzyme's active site. Structural analysis reveals that the substitution of tyrosine with the larger, more hydrophobic tryptophan residue at position 127 optimizes the binding pocket for the N-Boc-3-piperidone substrate. This mutation significantly enhances the catalytic efficiency, quantified by the $k_{cat}/K_m$ value, which is improved by approximately 21.5 times compared to the wild-type enzyme. The tryptophan side chain likely provides better hydrophobic stacking interactions with the Boc group of the substrate, stabilizing the transition state and facilitating faster hydride transfer. Additionally, the mutant demonstrates exceptional thermal stability, with a half-life of 147 hours at 30°C, which is nearly double that of the wild type. This robustness is critical for maintaining high activity over extended reaction periods required for high-conversion batches. The kinetic data underscores the mutant's ability to maintain high turnover numbers even under high substrate loads, ensuring consistent production quality.
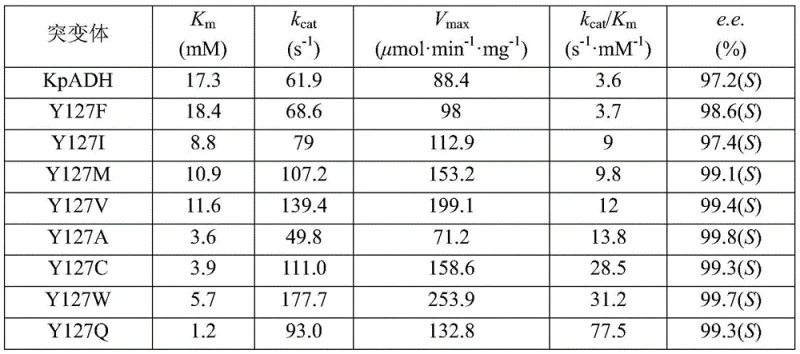
Furthermore, the mechanism involves a tightly coupled cofactor regeneration cycle that is essential for economic viability. As illustrated in the reaction scheme, the Y127W alcohol dehydrogenase reduces the ketone substrate to the corresponding chiral alcohol while oxidizing NAD(P)H to NAD(P)+. Simultaneously, the co-immobilized or co-present glucose dehydrogenase (GDH) oxidizes glucose to gluconolactone, reducing NAD(P)+ back to NAD(P)H. This cyclic regeneration ensures that only a catalytic amount of cofactor is needed, effectively removing one of the major cost drivers in biocatalysis. The system operates optimally at a neutral pH of 7.0 and moderate temperatures (25-35°C), conditions that are mild enough to prevent substrate degradation yet energetic enough to drive the reaction to completion. The high enantioselectivity (>99.4% e.e.) is maintained throughout the reaction, indicating that the mutation does not compromise the stereochemical control of the enzyme, a vital attribute for producing high-purity pharmaceutical intermediates required for regulatory approval.

How to Synthesize (S)-N-Boc-3-hydroxypiperidine Efficiently
The implementation of this biocatalytic route is designed for straightforward integration into existing manufacturing infrastructure. The process begins with the preparation of a reaction buffer, typically phosphate-buffered saline (PBS) at pH 7.0, which serves as the sole reaction medium. The substrate, N-Boc-3-piperidone, is added directly to this aqueous phase along with glucose, which acts as the sacrificial hydrogen donor. The key to success lies in the specific ratio of the biocatalysts; the Y127W mutant enzyme powder and the GDH enzyme powder are added in a optimized mass ratio, typically around 1.5:1 to 2:1, depending on the specific activity of the batches. The reaction is initiated by mechanical stirring at 30°C. Due to the high stability of the Y127W mutant, the reaction tolerates high substrate concentrations without the need for fed-batch strategies, although gradual addition can be employed for even larger scales. Monitoring the reaction progress via HPLC confirms that conversion exceeds 99% within 12 hours. Post-reaction, the mixture is simply extracted with an organic solvent like dichloromethane, and the product is isolated after solvent removal, yielding a pale yellow solid with high purity. For detailed operational parameters and safety guidelines, please refer to the standardized protocol below.
- Prepare a single aqueous reaction system containing PBS buffer (pH 7.0), substrate N-Boc-3-piperidone, and glucose as the hydrogen donor.
- Add the lyophilized enzyme powders of the Y127W alcohol dehydrogenase mutant and BmGDH (glucose dehydrogenase) directly to the reaction mixture without organic cosolvents.
- Maintain the reaction at 30°C with mechanical stirring for 12 hours to achieve >99% conversion, followed by extraction and purification.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of the Y127W biocatalytic process offers transformative advantages that directly impact the bottom line and operational resilience. The most significant benefit is the drastic simplification of the reaction medium. By eliminating the need for organic cosolvents like isopropanol or two-phase systems, the process reduces the volume of hazardous waste generated, lowering disposal costs and aligning with increasingly strict environmental regulations. This "green chemistry" approach also simplifies the downstream processing train, as there is no need to recover or distill large volumes of organic solvents before product isolation. Consequently, the overall energy consumption of the manufacturing process is significantly reduced, contributing to a lower carbon footprint for the final API.
- Cost Reduction in Manufacturing: The economic model of this process is strengthened by the elimination of expensive exogenous cofactors. The efficient coupling with glucose dehydrogenase allows for the recycling of NAD(P)H, meaning the costly cofactor does not need to be purchased in stoichiometric amounts. Additionally, the ability to run reactions at extremely high substrate concentrations (up to 600 g·L⁻¹) means that the same reactor volume can produce significantly more product per batch compared to conventional low-concentration methods. This intensification of the process leads to substantial savings in capital expenditure (CapEx) and operational expenditure (OpEx) by maximizing asset utilization. The removal of organic solvents further cuts raw material costs and reduces the complexity of solvent recovery systems.
- Enhanced Supply Chain Reliability: Reliance on complex multi-step chemical syntheses or unstable biocatalysts often introduces volatility into the supply chain. The Y127W mutant's exceptional stability (half-life of 147 hours) ensures consistent batch-to-batch performance, reducing the risk of failed runs and production delays. The raw materials required—glucose and buffer salts—are commodity chemicals with stable, global supply chains, unlike specialized organometallic catalysts that may be subject to geopolitical supply risks. This robustness ensures reducing lead time for high-purity pharmaceutical intermediates and guarantees a steady flow of materials to downstream API manufacturers, securing the continuity of drug production.
- Scalability and Environmental Compliance: Scaling biocatalytic processes is often challenging due to oxygen transfer limitations or heat generation, but this single aqueous phase system is inherently easier to manage. The absence of flammable organic solvents reduces safety hazards associated with large-scale reactors, simplifying compliance with safety standards. The high space-time yield (~1400 g·L⁻¹·d⁻¹) demonstrates that the process is not just a laboratory curiosity but a viable industrial solution. The simplified waste stream, primarily consisting of aqueous buffer and gluconolactone, is easier to treat than solvent-heavy waste, facilitating smoother regulatory approvals and environmental audits for reliable pharmaceutical intermediate supplier facilities.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this patented biocatalytic technology. These insights are derived directly from the experimental data and beneficial effects reported in the patent documentation, providing clarity on how this method compares to legacy processes. Understanding these specifics is crucial for technical teams evaluating the feasibility of integrating this route into their current production pipelines.
Q: What are the advantages of the Y127W mutant over wild-type enzymes?
A: The Y127W mutant exhibits a 21.5-fold improvement in catalytic efficiency (kcat/Km) compared to the wild type and demonstrates superior stability with a half-life of 147 hours at 30°C. Crucially, it eliminates product inhibition in single aqueous phases, allowing for substrate concentrations up to 600 g/L.
Q: Does this process require expensive cofactors or organic solvents?
A: No. The process utilizes a coupled enzyme system with glucose dehydrogenase (GDH) to regenerate NAD(P)H in situ using glucose. Furthermore, it operates efficiently in a single aqueous phase without the need for toxic organic cosolvents, significantly reducing environmental impact and downstream processing costs.
Q: What is the optical purity of the final product?
A: The biocatalytic reduction using the Y127W mutant yields (S)-N-Boc-3-hydroxypiperidine with an enantiomeric excess (e.e.) value exceeding 99.4%, meeting the stringent purity requirements for chiral pharmaceutical intermediates used in oncology drug synthesis.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable (S)-NBHP Supplier
The technological advancements detailed in patent CN110777125B highlight the immense potential of enzyme engineering in modern pharmaceutical manufacturing. At NINGBO INNO PHARMCHEM, we recognize the critical importance of translating such innovative laboratory discoveries into robust, industrial-scale realities. As a premier CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production. Our state-of-the-art facilities are equipped to handle complex biocatalytic processes, ensuring that the high efficiency and purity demonstrated in the patent are maintained at scale. We adhere to stringent purity specifications and operate rigorous QC labs to guarantee that every batch of (S)-NBHP meets the exacting standards required for oncology drug synthesis.
We invite pharmaceutical companies and research institutions to collaborate with us to leverage this cutting-edge technology for your supply chain. By partnering with us, you gain access to a Customized Cost-Saving Analysis tailored to your specific volume requirements. We encourage you to contact our technical procurement team today to request specific COA data and route feasibility assessments. Let us help you secure a sustainable, cost-effective, and high-quality supply of this vital chiral intermediate, ensuring your drug development projects proceed without interruption.
