Advanced Dual-Enzyme Synthesis of (R)-Citronellol for Commercial Fragrance Manufacturing
The global demand for high-purity chiral intermediates in the fragrance industry is driving a significant shift towards sustainable biocatalytic solutions. Patent CN112662709A introduces a groundbreaking method for synthesizing (R)-citronellol through a novel dual-enzyme coupling system. This technology leverages the synergistic action of alcohol dehydrogenase YsADH and old yellow enzyme OYE2y-HG to convert geraniol into (R)-citronellol with exceptional efficiency. Unlike traditional chemical reductions that often struggle with chemoselectivity, this biological approach operates under mild conditions, typically between 25-55 deg.C, ensuring the integrity of sensitive functional groups. For R&D directors seeking robust pathways for terpene alcohol derivatives, this patent offers a compelling alternative to harsh chemical hydrogenation, promising a greener footprint without compromising on yield or optical purity.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional chemical synthesis of citronellol often relies on the reduction of citral or geraniol using metal catalysts under high pressure and temperature. These methods frequently suffer from poor chemoselectivity, leading to the formation of unwanted byproducts such as citronellal or fully saturated alcohols that require energy-intensive distillation to remove. Furthermore, achieving high enantiomeric excess in chemical routes typically necessitates expensive chiral ligands or resolution steps, which drastically increase the cost of goods sold. The reliance on transition metals also introduces regulatory hurdles regarding heavy metal residues, a critical concern for suppliers serving the food and fragrance sectors. Consequently, procurement teams face volatile pricing and supply chain risks associated with the complex purification infrastructure required to meet stringent purity specifications.
The Novel Approach
In contrast, the dual-enzyme coupling method described in the patent utilizes a sophisticated coenzyme self-circulation system that inherently resolves these selectivity issues. By employing engineered E. coli strains expressing specific genes (YsADH and OYE2y-HG), the process achieves a conversion rate of 95 percent at a substrate concentration of 40mM within just 6 hours. This biological route operates at ambient pressure and moderate temperatures, significantly reducing energy consumption and equipment stress. The elimination of external cosubstrates for coenzyme regeneration marks a pivotal advancement in atom economy, as the system internally recycles NADPH. For a reliable flavor & fragrance intermediate supplier, adopting this technology translates to a streamlined manufacturing process that minimizes waste generation and maximizes throughput efficiency.
Mechanistic Insights into Dual-Enzyme Coenzyme Self-Circulation
The core innovation lies in the precise orchestration of two distinct enzymatic activities within a single reaction vessel. The alcohol dehydrogenase YsADH initially catalyzes the dehydrogenation of geraniol to generate geranial and NADPH. Subsequently, the old yellow enzyme OYE2y-HG utilizes this in-situ generated NADPH to reduce geranial into (R)-citronellal, which is further reduced to the final alcohol product. This seamless handover of hydride equivalents ensures that the expensive cofactor NADP+ is continuously regenerated without the need for additional glucose or formate donors. Such a mechanism not only lowers raw material costs but also simplifies the downstream workup by reducing the load of organic impurities. Understanding this catalytic cycle is essential for technical teams aiming to optimize reaction parameters for cost reduction in synthetic flavors manufacturing.
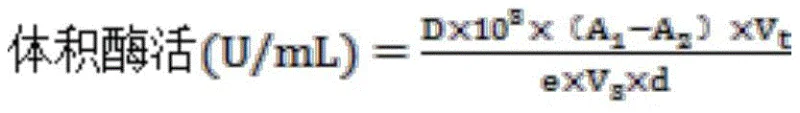
Furthermore, the system exhibits remarkable stereoselectivity, consistently producing (R)-citronellol with an e.e. value >99 percent. This high level of optical purity is paramount for the subsequent synthesis of cis-rose oxide, where the specific chirality dictates the olfactory profile of the final fragrance. The enzyme engineering ensures that side reactions are minimized, maintaining a clean impurity profile that facilitates easier purification. For quality assurance managers, this means fewer batches are rejected due to off-spec enantiomeric ratios, thereby enhancing overall supply chain reliability. The robustness of this biocatalytic platform allows for consistent performance across different scales, making it an ideal candidate for commercial scale-up of complex terpene alcohols.
How to Synthesize (R)-Citronellol Efficiently
Implementing this synthesis route requires careful preparation of the biocatalysts and precise control over reaction conditions to maximize yield. The process begins with the fermentation of engineered bacterial strains to produce wet thalli, which are then mixed in a specific mass ratio to balance the enzymatic activities. Operators must maintain the reaction environment at an optimal pH of 7.5 and a temperature of 40 deg.C to ensure peak enzyme performance. Detailed standard operating procedures regarding substrate feeding rates and agitation speeds are critical to prevent mass transfer limitations. The detailed standardized synthesis steps are outlined in the guide below for technical reference.
- Prepare wet cells of engineered E. coli expressing alcohol dehydrogenase YsADH and old yellow enzyme OYE2y-HG, mixing them at a mass ratio of 4: 1.
- Establish the reaction system with 40mM geraniol substrate and 0.4mM NADP+ cofactor in 50mM Tris-HCl buffer at pH 7.5.
- Maintain reaction at 40°C and 400rpm for 6 hours to achieve >95% conversion, followed by ethyl acetate extraction and purification.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, this biocatalytic process offers substantial strategic advantages for organizations focused on sustainability and cost efficiency. The elimination of expensive transition metal catalysts and the reduction in solvent usage directly contribute to a lower environmental impact, aligning with modern green chemistry mandates. Supply chain leaders will appreciate the simplified logistics, as the process does not require hazardous high-pressure hydrogen gas or specialized containment systems. This inherent safety profile reduces insurance costs and regulatory compliance burdens, facilitating smoother operations across global manufacturing sites. Additionally, the high specificity of the enzymes reduces the need for complex chromatographic separations, shortening the overall production cycle time.
- Cost Reduction in Manufacturing: The self-circulating coenzyme system removes the necessity for purchasing large quantities of external reducing agents or cosubstrates, which traditionally account for a significant portion of biocatalytic operational expenses. By internally regenerating NADPH, the process drastically lowers the variable cost per kilogram of product. Furthermore, the high conversion efficiency minimizes raw material waste, ensuring that the majority of the geraniol substrate is converted into valuable product rather than discarded byproducts. This efficiency gain allows manufacturers to offer more competitive pricing structures while maintaining healthy margins in a volatile market.
- Enhanced Supply Chain Reliability: The use of robust whole-cell catalysts simplifies the storage and handling requirements compared to isolated enzymes, which often require cold chain logistics. The stability of the engineered strains ensures consistent batch-to-batch performance, reducing the risk of production delays caused by catalyst variability. This reliability is crucial for reducing lead time for high-purity fragrance ingredients, allowing customers to plan their inventory with greater confidence. Moreover, the scalability of the fermentation process ensures that supply can be rapidly ramped up to meet surges in market demand without significant capital investment in new reactor types.
- Scalability and Environmental Compliance: Operating under mild aqueous conditions significantly reduces the generation of hazardous organic waste streams, simplifying wastewater treatment protocols. The absence of heavy metal residues eliminates the need for costly scavenging steps, further streamlining the purification workflow. This environmentally friendly profile supports corporate sustainability goals and facilitates easier regulatory approval in regions with strict environmental laws. The process is inherently designed for commercial scale-up of complex polymer additives and fine chemicals, proving its versatility beyond just fragrance applications.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this dual-enzyme technology. These insights are derived directly from the experimental data and beneficial effects reported in the patent documentation. They serve to clarify the operational feasibility and strategic value of adopting this biocatalytic route for industrial production. Stakeholders are encouraged to review these points to understand the full scope of the technology's capabilities.
Q: How does the dual-enzyme system improve atom economy compared to traditional biocatalysis?
A: The patented coenzyme self-circulation system eliminates the need for excessive auxiliary substrates. By coupling YsADH and OYE2y-HG, the NADPH generated by dehydrogenation is immediately consumed for reduction, driving the cycle without external cost drivers.
Q: What is the optical purity achievable with this enzymatic route?
A: The process demonstrates exceptional enantioselectivity, consistently delivering an e.e. value of >99%. This high stereochemical control is critical for downstream synthesis of cis-rose oxide.
Q: Is this biocatalytic method suitable for large-scale industrial production?
A: Yes, the mild reaction conditions (40°C, atmospheric pressure) and use of whole-cell catalysts simplify reactor requirements. The high substrate tolerance up to 40mM supports efficient commercial scale-up.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable (R)-Citronellol Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of this dual-enzyme coupling technology for the fine chemical sector. As a dedicated CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that laboratory innovations are successfully translated into industrial reality. Our facilities are equipped with rigorous QC labs and adhere to stringent purity specifications, guaranteeing that every batch of (R)-citronellol meets the highest international standards. We are committed to providing our partners with a secure and consistent supply of high-quality intermediates that drive their own product excellence.
We invite you to collaborate with us to leverage this advanced synthesis method for your specific application needs. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your volume requirements and quality targets. Please contact us to request specific COA data and route feasibility assessments, and let us demonstrate how our expertise can optimize your supply chain for the future.
