Scalable Enzymatic Resolution of R-3-Aminobutanol for High-Purity HIV Integrase Inhibitor Synthesis
Introduction to Advanced Biocatalytic Resolution Technologies
The pharmaceutical industry's relentless pursuit of high-purity chiral intermediates has led to significant innovations in biocatalysis, particularly for critical antiretroviral agents. Patent CN110358804B discloses a groundbreaking enzymatic production process for R-3-aminobutanol, a pivotal chiral building block used in the synthesis of Dolutegravir, a next-generation HIV integrase inhibitor. Unlike traditional chemical methods that often struggle with environmental impact and selectivity issues, this invention leverages the exquisite stereospecificity of omega-transaminases derived from microorganisms such as Ochrobactrum anthropi and Brucella neotomae. By utilizing a kinetic resolution strategy where the enzyme selectively consumes the S-enantiomer of the racemic substrate, the process leaves behind the desired R-enantiomer with exceptional optical purity. This technological leap addresses the critical demand for reliable pharmaceutical intermediate suppliers who can deliver complex chiral molecules with consistent quality and reduced environmental footprint.
The significance of this patent extends beyond mere academic interest; it represents a viable industrial solution for cost reduction in API manufacturing. Dolutegravir has demonstrated superior efficacy and resistance profiles compared to earlier generations of HIV medications, driving massive market demand. However, the production cost and optical purity of its key precursor, R-3-aminobutanol, have historically been bottlenecks. The disclosed method overcomes the dynamic equilibrium limitations common in transaminase reactions by coupling the resolution with pyruvic acid, effectively driving the reaction towards completion for the unwanted isomer. This ensures that the recovered R-3-aminobutanol meets the stringent purity specifications required for GMP-grade drug substance production, positioning this biocatalytic route as a preferred choice for forward-thinking supply chain heads.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the production of optically pure R-3-aminobutanol has relied heavily on chemical synthesis routes that are fraught with inefficiencies and operational hazards. One prevalent method involves the kinetic resolution of racemic 3-aminobutanol using chiral resolving agents, a process notoriously plagued by a maximum theoretical yield of 50% and the generation of substantial waste streams containing the unwanted S-enantiomer salts. Furthermore, these chemical resolutions often require harsh reaction conditions, including extreme temperatures or the use of corrosive acids and bases, which complicate reactor material selection and increase safety risks in large-scale manufacturing. Another conventional approach utilizes expensive chiral pool starting materials, necessitating multi-step synthetic sequences that accumulate impurities and drastically inflate the cost of goods sold. These legacy methods fail to meet the modern standards of green chemistry and economic viability required by today's competitive generic and branded pharmaceutical markets.
The Novel Approach
In stark contrast, the novel enzymatic approach detailed in the patent utilizes omega-transaminases to achieve a highly efficient kinetic resolution under mild, aqueous conditions. The core innovation lies in the discovery that specific microbial transaminases exhibit absolute stereospecificity for the S-enantiomer of 3-aminobutanol when paired with pyruvic acid as an amino acceptor. This reaction converts the S-isomer into L-alanine and 4-hydroxy-2-butanone, leaving the desired R-3-aminobutanol untouched in the reaction mixture. 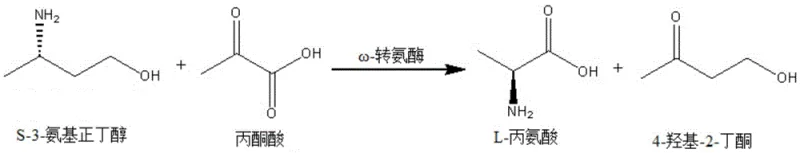 This mechanism bypasses the thermodynamic equilibrium issues often seen in asymmetric synthesis because the byproduct formation drives the resolution forward. The ability to operate at neutral pH and moderate temperatures (30-40°C) not only preserves the integrity of the sensitive amino alcohol but also simplifies the downstream processing, allowing for straightforward recovery via crystallization or extraction. This shift from stoichiometric chemical reagents to catalytic biological systems marks a paradigm shift in how high-value chiral intermediates are manufactured.
This mechanism bypasses the thermodynamic equilibrium issues often seen in asymmetric synthesis because the byproduct formation drives the resolution forward. The ability to operate at neutral pH and moderate temperatures (30-40°C) not only preserves the integrity of the sensitive amino alcohol but also simplifies the downstream processing, allowing for straightforward recovery via crystallization or extraction. This shift from stoichiometric chemical reagents to catalytic biological systems marks a paradigm shift in how high-value chiral intermediates are manufactured.
Mechanistic Insights into Omega-Transaminase Catalyzed Resolution
The success of this process hinges on the precise molecular recognition capabilities of the omega-transaminase enzyme, which acts as a biological filter for chirality. The enzyme's active site is geometrically constrained to bind only the S-configured substrate, facilitating the transfer of an amino group to pyruvic acid while rejecting the R-configured molecule. This selectivity is further enhanced by the addition of pyridoxal phosphate (PLP) as a cofactor, typically at concentrations between 0.1 to 1 mmol/L, which stabilizes the transition state and accelerates the turnover rate. The patent highlights that enzymes from sources like Ochrobactrum anthropi (OATA) and Brucella neotomae (BNTA) possess unique amino acid sequences (SEQ ID NO: 1 and 3) that confer robustness against high substrate concentrations. This structural stability is crucial because it allows the reaction to proceed efficiently even when the loading of racemic 3-aminobutanol is increased to industrially relevant levels, such as 100 g/L, without significant loss of enzyme activity or selectivity.
From an impurity control perspective, this enzymatic mechanism offers a distinct advantage by minimizing the formation of side products common in chemical synthesis. Since the reaction is highly specific, the primary byproduct is L-alanine, a benign amino acid that is easily separated from the target R-3-aminobutanol due to differences in solubility and polarity. The process avoids the use of heavy metal catalysts or toxic organic solvents often required in asymmetric hydrogenation or reduction steps, thereby simplifying the purification workflow. Moreover, the optical purity of the final product is directly correlated with the conversion rate of the substrate; by controlling the reaction time and enzyme loading, manufacturers can tune the ee value of the recovered R-enantiomer to exceed 98%, meeting the rigorous quality standards demanded by regulatory agencies for antiviral drug intermediates.
How to Synthesize R-3-Aminobutanol Efficiently
The implementation of this enzymatic resolution process requires a systematic approach to strain engineering and reaction optimization to ensure maximum yield and purity. The patent outlines a clear pathway starting from gene synthesis to the final isolation of the product, emphasizing the importance of selecting the right host organism and expression conditions. While the specific laboratory protocols involve detailed steps regarding plasmid construction and fermentation parameters, the general workflow is designed to be adaptable for commercial scale-up. For a comprehensive understanding of the operational parameters, the detailed standardized synthesis steps are provided in the guide below.
- Construct recombinant E. coli strains expressing specific omega-transaminases (e.g., OATA, BNTA) and cultivate in TB medium with IPTG induction.
- Prepare crude enzyme solution via cell lysis and mix with racemic 3-aminobutanol and sodium pyruvate in a buffered aqueous system.
- Maintain reaction at pH 7.0-7.5 and 37°C to selectively convert S-enantiomer, then recover unreacted R-3-aminobutanol via crystallization or extraction.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the transition to this enzymatic process offers tangible strategic benefits that extend beyond simple technical metrics. The elimination of expensive chiral resolving agents and the reduction in synthetic steps directly translate to a more streamlined cost structure, making the supply of R-3-aminobutanol more resilient to raw material price fluctuations. By utilizing recombinant E. coli strains that can be fermented at high densities, the manufacturing process becomes less dependent on scarce natural resources and more reliant on scalable biotechnology infrastructure. This shift enhances supply chain reliability by reducing the number of external vendors needed for specialized reagents, thereby mitigating the risk of disruptions that often plague complex chemical supply chains. Furthermore, the aqueous nature of the reaction significantly lowers the volume of hazardous organic solvents required, aligning with increasingly strict environmental regulations and reducing waste disposal costs.
- Cost Reduction in Manufacturing: The enzymatic route fundamentally alters the economics of production by replacing stoichiometric chemical reagents with reusable biocatalysts. Since the enzyme acts catalytically rather than being consumed, the overall mass intensity of the process is drastically improved, leading to substantial cost savings in raw material procurement. Additionally, the ability to run the reaction at high substrate concentrations means that smaller reactor volumes can be used to produce the same amount of product, effectively increasing asset utilization and lowering capital expenditure requirements for new production lines. The simplified downstream processing, which avoids complex salt formation and breaking steps, further reduces labor and utility costs associated with purification.
- Enhanced Supply Chain Reliability: Adopting a biocatalytic process diversifies the sourcing strategy away from petrochemical-derived chiral pools towards fermentation-based production, which is generally more stable and predictable. The microbial strains used, such as those derived from Ochrobactrum and Brucella species, can be preserved and propagated indefinitely, ensuring a consistent supply of the biocatalyst without the batch-to-batch variability seen in natural extraction methods. This consistency is vital for maintaining long-term contracts with pharmaceutical clients who require guaranteed availability of critical intermediates for their own drug manufacturing schedules. The robustness of the enzyme under mild conditions also reduces the risk of production failures due to equipment malfunction or temperature excursions.
- Scalability and Environmental Compliance: The process is inherently designed for scale-up, utilizing standard fermentation and aqueous reaction technologies that are well-understood in the fine chemical industry. Operating at near-neutral pH and moderate temperatures reduces the corrosion burden on manufacturing equipment, extending the lifespan of reactors and piping systems. From an environmental standpoint, the reduction in organic solvent usage and the generation of biodegradable byproducts like alanine significantly lower the facility's environmental footprint. This compliance with green chemistry principles not only avoids potential regulatory fines but also enhances the corporate sustainability profile, which is increasingly important for securing partnerships with major multinational pharmaceutical companies.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the enzymatic production of R-3-aminobutanol, based on the specific data and embodiments found in the patent literature. These insights are intended to clarify the feasibility and advantages of adopting this biocatalytic route for industrial applications. Understanding these details is essential for technical teams evaluating the integration of this technology into existing manufacturing portfolios.
Q: What is the primary advantage of using omega-transaminase over chemical resolution for R-3-aminobutanol?
A: The enzymatic method offers superior stereospecificity under mild conditions (pH 7.0-7.5, 37°C), avoiding the harsh reagents and low yields associated with traditional chemical kinetic resolution.
Q: Can this biocatalytic process handle high substrate concentrations for industrial scale-up?
A: Yes, the patent demonstrates that specific omega-transaminases maintain high activity even at substrate concentrations up to 100g/L, significantly reducing solvent usage and improving volumetric productivity.
Q: Which microbial sources provide the most effective transaminases for this resolution?
A: Enzymes derived from Ochrobactrum anthropi (OATA), Brucella neotomae (BNTA), and Novosphingobium acidiphilum (NATA) exhibit the highest catalytic activity and optical purity retention.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable R-3-Aminobutanol Supplier
As the global demand for antiretroviral therapies continues to rise, securing a stable and high-quality supply of key intermediates like R-3-aminobutanol is paramount for pharmaceutical manufacturers. NINGBO INNO PHARMCHEM stands at the forefront of this sector, leveraging advanced biocatalytic technologies to deliver superior chiral building blocks. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that we can meet your volume requirements without compromising on quality. We operate stringent purity specifications and maintain rigorous QC labs to guarantee that every batch of R-3-aminobutanol meets the exacting standards required for HIV drug synthesis, providing you with peace of mind and supply security.
We invite you to collaborate with us to optimize your supply chain and reduce your overall manufacturing costs through our innovative enzymatic processes. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your specific production needs, demonstrating exactly how switching to our biocatalytic grade intermediates can improve your bottom line. Please contact us today to request specific COA data and route feasibility assessments, and let us partner with you to drive efficiency and excellence in your pharmaceutical manufacturing operations.
