Scalable Synthesis of Pyroxasulfone Intermediates Using Advanced BOC Protection Chemistry
Introduction to Next-Generation Agrochemical Intermediate Synthesis
The development of efficient and safe synthetic routes for key agrochemical intermediates is a critical priority for modern pharmaceutical and crop protection manufacturers. Patent CN116410138A introduces a groundbreaking methodology for the preparation of 5-difluoromethoxy-4-hydroxymethyl-3-trifluoromethyl-1-methylpyrazole, a vital precursor for the herbicide Pyroxasulfone. This innovation addresses long-standing safety and cost challenges by replacing hazardous methylhydrazine with inexpensive hydrazine hydrate, utilizing a strategic BOC protection sequence to ensure high regioselectivity. The structural integrity of the target molecule, essential for its biological activity as a VLCFA inhibitor, is maintained through mild reaction conditions that facilitate easier purification and higher overall throughput.
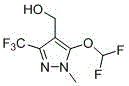
For R&D directors and process chemists, this patent represents a significant shift towards greener chemistry without compromising yield. The traditional reliance on toxic reagents often necessitates expensive containment systems and complex waste treatment protocols. By contrast, this new approach leverages standard organic transformations—cyclization, hydroxymethylation, and nucleophilic substitution—underpinned by a robust protecting group strategy. This not only enhances operator safety but also streamlines the supply chain by utilizing readily available bulk chemicals, positioning this route as a superior alternative for reliable agrochemical intermediate supplier networks seeking to optimize their manufacturing portfolios.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of pyrazole-based herbicide intermediates has been plagued by the use of methylhydrazine, a substance known for its high toxicity and carcinogenic potential. Prior art, such as that described in patent EP1990336, relies heavily on reacting ethyl trifluoroacetoacetate directly with aqueous methylhydrazine solutions. This conventional pathway presents severe operational hazards, requiring specialized equipment to handle volatile and dangerous vapors, which drastically increases capital expenditure and operational risk. Furthermore, the lack of a protecting group in these early stages often leads to selectivity issues during subsequent functionalization, resulting in complex impurity profiles that are difficult and costly to remove during downstream processing.
Additionally, the direct use of methylhydrazine limits the flexibility of the synthesis, as the nitrogen atom is already substituted before the ring is fully formed. This can lead to side reactions during the difluoromethylation step, where the nitrogen might compete with the oxygen for alkylation, reducing the overall yield of the desired 5-difluoromethoxy product. The environmental burden of disposing of hydrazine-containing waste streams further complicates the regulatory compliance landscape for manufacturers, making these legacy processes increasingly unsustainable in a market that demands stricter environmental, social, and governance (ESG) standards.
The Novel Approach
The innovative route disclosed in CN116410138A fundamentally reengineers the synthesis by introducing a BOC (tert-butoxycarbonyl) protection step at the very beginning. By reacting cheap hydrazine hydrate with BOC anhydride at controlled low temperatures (around -10°C to 0°C), the process generates a stable tert-butoxycarbonyl hydrazine intermediate. This protected species then undergoes a clean cyclization with ethyl trifluoroacetoacetate under reflux conditions to form the pyrazole core. This strategic modification effectively masks the nitrogen reactivity, allowing for precise control over subsequent substitutions and eliminating the formation of N-difluoromethylated byproducts.
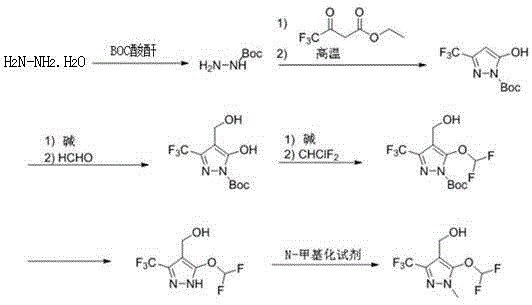
Following the ring closure, the process proceeds through a highly efficient sequence of hydroxymethylation using formaldehyde and difluoromethylation using difluorochloromethane in the presence of a base. The beauty of this novel approach lies in its modularity and safety; the harsh conditions associated with methylhydrazine are completely avoided. The final steps involve a straightforward acid-mediated deprotection to remove the BOC group, followed by a selective N-methylation. This results in a high-purity final product with a simplified workup procedure, demonstrating a clear advantage in cost reduction in herbicide manufacturing by minimizing waste and maximizing atom economy.
Mechanistic Insights into BOC-Protected Pyrazole Cyclization
The core mechanistic advantage of this synthesis lies in the temporal control of nitrogen reactivity provided by the BOC group. In the initial cyclization step, the protected hydrazine acts as a 1,3-dinucleophile, attacking the beta-keto ester functionality of ethyl trifluoroacetoacetate. The electron-withdrawing nature of the trifluoromethyl group activates the carbonyl for nucleophilic attack, while the BOC group ensures that the nitrogen remains non-nucleophilic towards external electrophiles like difluorochloromethane during the intermediate stages. This prevents the formation of N-CF2H bonds prematurely, which would be difficult to reverse and would constitute a major impurity. The cyclization typically proceeds via a condensation mechanism followed by dehydration, driven by the thermodynamic stability of the aromatic pyrazole ring system.
Furthermore, the selectivity during the final N-methylation step is critically dependent on the prior deprotection. Once the BOC group is removed using acids like trifluoroacetic acid or hydrochloric acid, the nitrogen becomes available for alkylation. However, because the 5-position is already occupied by the difluoromethoxy group and the 4-position by the hydroxymethyl group, the methylation reagent (such as dimethyl sulfate) reacts exclusively at the N1 position. This precise orchestration of reactivity ensures that the impurity profile remains exceptionally clean, a key metric for R&D directors evaluating the feasibility of scaling complex agrochemical intermediates. The use of mild bases like potassium carbonate in the final step further suppresses side reactions, ensuring high chemical fidelity.
How to Synthesize 5-difluoromethoxy-4-hydroxymethyl-3-trifluoromethyl-1-methylpyrazole Efficiently
The synthesis protocol outlined in the patent provides a robust framework for laboratory and pilot-scale production, emphasizing temperature control and stoichiometric precision. The process begins with the exothermic protection of hydrazine, requiring careful thermal management to prevent decomposition. Subsequent steps leverage common organic solvents such as isopropanol, xylene, and acetonitrile, which are easily recovered and recycled, enhancing the economic viability of the route. The detailed standardized synthesis steps below outline the critical parameters for achieving optimal yields and purity, serving as a practical guide for process engineers looking to implement this technology.
- Protect hydrazine hydrate with BOC anhydride at low temperature (-10°C to 0°C) to form tert-butoxycarbonyl hydrazine.
- Perform ring closure with ethyl trifluoroacetoacetate under reflux to generate the protected pyrazole core (Compound I).
- Execute sequential hydroxymethylation with formaldehyde and difluoromethylation with difluorochloromethane in alkaline conditions.
- Remove the BOC protecting group using acid (e.g., TFA) and finalize with N-methylation using dimethyl sulfate.
Commercial Advantages for Procurement and Supply Chain Teams
From a procurement perspective, the shift from methylhydrazine to hydrazine hydrate represents a substantial reduction in raw material costs and logistical complexity. Methylhydrazine is a controlled substance in many jurisdictions due to its toxicity, often requiring specialized transport and storage facilities that drive up overheads. Hydrazine hydrate, conversely, is a commodity chemical with a stable global supply chain, ensuring consistent availability and price stability. This transition not only lowers the direct cost of goods sold but also mitigates the risk of supply disruptions caused by regulatory changes or safety incidents at supplier facilities, thereby enhancing supply chain reliability for long-term production contracts.
- Cost Reduction in Manufacturing: The elimination of toxic methylhydrazine removes the need for expensive containment infrastructure and specialized personal protective equipment, leading to significant operational expenditure savings. Additionally, the high selectivity of the BOC-protected route reduces the burden on purification units, lowering solvent consumption and energy usage associated with distillation and chromatography. The overall process efficiency is improved by the ability to run reactions at atmospheric pressure and moderate temperatures, which reduces utility costs and extends the lifespan of reactor vessels.
- Enhanced Supply Chain Reliability: By relying on widely available starting materials like hydrazine hydrate and ethyl trifluoroacetoacetate, manufacturers can diversify their supplier base and reduce dependency on single-source vendors of hazardous reagents. The robustness of the synthetic route means that production schedules are less likely to be impacted by safety audits or environmental shutdowns, ensuring a steady flow of high-purity intermediates to downstream formulation plants. This stability is crucial for meeting the just-in-time delivery requirements of major agrochemical companies.
- Scalability and Environmental Compliance: The process is designed with industrial scale-up in mind, avoiding high-pressure hydrogenation or cryogenic conditions that are difficult to replicate in large reactors. The waste streams generated are less hazardous, simplifying effluent treatment and reducing the environmental footprint of the manufacturing site. This alignment with green chemistry principles facilitates easier permitting and regulatory approval, accelerating the time-to-market for new herbicide formulations derived from this intermediate.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthetic route. These insights are derived directly from the experimental data and comparative examples provided in the patent documentation, offering clarity on yield expectations, safety protocols, and scalability factors. Understanding these nuances is essential for technical teams evaluating the adoption of this methodology for commercial production.
Q: Why is hydrazine hydrate preferred over methylhydrazine in this synthesis?
A: Hydrazine hydrate is significantly less toxic and cheaper than methylhydrazine. The novel route uses BOC protection to control selectivity, eliminating the high safety risks associated with handling toxic methylhydrazine while maintaining high yield.
Q: What is the role of the BOC protecting group in this process?
A: The BOC group protects the nitrogen atom during the initial cyclization and subsequent functionalization steps. This prevents unwanted N-difluoromethylation and ensures that methylation occurs selectively at the final stage, improving overall purity and yield.
Q: Is this process suitable for large-scale industrial production?
A: Yes, the patent explicitly states the method avoids excessive temperatures and high pressure, uses common solvents like isopropanol and acetonitrile, and involves simple post-treatment steps, making it highly suitable for commercial scale-up.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 5-difluoromethoxy-4-hydroxymethyl-3-trifluoromethyl-1-methylpyrazole Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of adopting safer and more efficient synthetic routes for key agrochemical building blocks. Our technical team has extensively analyzed the BOC-protection strategy described in CN116410138A and is well-equipped to translate this laboratory-scale innovation into commercial reality. We possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from pilot plant to full-scale manufacturing is seamless. Our state-of-the-art facilities are designed to handle fluorinated intermediates with the highest safety standards, and our rigorous QC labs enforce stringent purity specifications to meet the exacting demands of global crop protection leaders.
We invite procurement managers and R&D directors to collaborate with us to leverage this advanced technology for your supply chain. By partnering with us, you gain access to a Customized Cost-Saving Analysis that quantifies the economic benefits of switching to this hydrazine hydrate-based route. We encourage you to contact our technical procurement team today to request specific COA data and route feasibility assessments tailored to your volume requirements, ensuring a secure and cost-effective supply of this vital herbicide intermediate.
