Scalable Production of 2,5-Furandicarboxylic Acid Diesters Using Earth-Abundant Catalysts
Introduction to Advanced Bio-Based Polymer Precursors
The global shift towards sustainable materials has intensified the demand for bio-based alternatives to petroleum-derived polymers, specifically targeting the replacement of polyethylene terephthalate (PET) with polyethylene furanoate (PEF). Central to this transition is the efficient production of 2,5-furandicarboxylic acid (FDCA) and its derivatives. Patent CN110590721B introduces a groundbreaking methodology for synthesizing 2,5-furandicarboxylic acid diester compounds directly from 5-hydroxymethylfurfural (HMF). This technology leverages a novel class of nitrogen-doped carbon material-supported cobalt-based bimetallic catalysts, which operate under remarkably mild conditions using inexpensive oxygen or air as the oxidant. By circumventing the need for precious metals and harsh reaction environments, this invention addresses critical bottlenecks in the commercialization of renewable polymer monomers, offering a pathway to high-purity intermediates with superior economic viability.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the oxidative esterification of HMF to FDCA diesters has relied heavily on noble metal catalysts such as Palladium (Pd) and Gold (Au). While these systems demonstrate catalytic activity, they suffer from prohibitive costs that severely limit their scalability for industrial applications. Furthermore, existing base-metal alternatives, particularly Cobalt-Nitrogen-Carbon (Co-N-C) systems, often necessitate the addition of expensive cocatalysts or require excessively long reaction times to achieve acceptable yields. These inefficiencies not only inflate the operational expenditure but also complicate the downstream purification processes due to potential metal leaching and byproduct formation. The reliance on high-pressure hydrogenation or aggressive oxidants in older methodologies further exacerbates safety concerns and energy consumption, creating a significant barrier for reliable bio-based polymer intermediate suppliers aiming to compete with established petrochemical value chains.
The Novel Approach
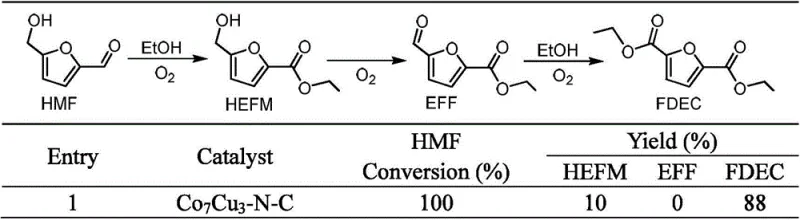
The patented methodology revolutionizes this landscape by employing earth-abundant bimetallic systems, such as Cobalt-Copper or Cobalt-Potassium, supported on nitrogen-doped carbon matrices derived from ZIF-8 precursors. This approach eliminates the dependency on precious metals while achieving near-quantitative conversion rates and exceptional selectivity. The process is highly adaptable, functioning efficiently with various lower alcohols including methanol, ethanol, and propanol to produce corresponding diesters like dimethyl 2,5-furandicarboxylate (FDMC) or diethyl 2,5-furandicarboxylate (FDEC). By utilizing ambient air or low-pressure oxygen at moderate temperatures ranging from 60 to 120°C, the technology drastically simplifies reactor design and operational protocols. This versatility ensures that manufacturers can tailor the solvent system to specific downstream polymerization requirements without compromising yield or purity.
Mechanistic Insights into Co-Bimetallic Catalyzed Oxidative Esterification
The core of this technological advancement lies in the synergistic interaction between the cobalt center and the secondary active metal within the nitrogen-doped carbon framework. The catalytic cycle initiates with the activation of molecular oxygen on the metal sites, facilitating the selective oxidation of the hydroxymethyl group and the aldehyde functionality of HMF. Unlike monometallic systems that may struggle with over-oxidation or incomplete conversion, the bimetallic configuration optimizes the electronic environment, enhancing the adsorption of reactants and the desorption of products. This precise tuning prevents the formation of unwanted carboxylic acid precipitates, keeping the intermediate species soluble in the alcohol medium and allowing for higher substrate concentrations. The nitrogen doping in the carbon support further stabilizes the metal nanoparticles, preventing agglomeration and ensuring long-term catalyst stability throughout the reaction duration.
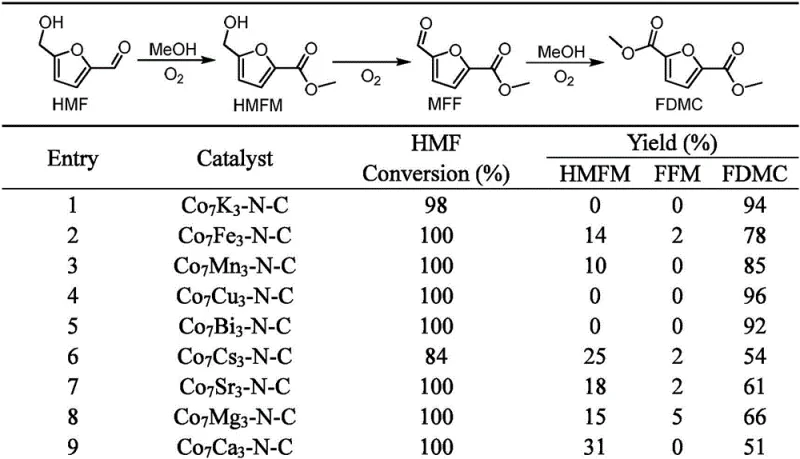
Impurity control is meticulously managed through the specific selection of the second metal component, such as Copper, Iron, or Potassium, which modulates the redox potential of the catalyst. This modulation is critical for suppressing side reactions that typically lead to ring-opening of the furan moiety or the formation of humins, which are common pitfalls in HMF chemistry. The result is a clean reaction profile where the primary byproduct is water, and the target diester is obtained with high purity, often exceeding 90% yield in optimized conditions. For R&D directors, this implies a streamlined purification workflow, potentially requiring only simple distillation rather than complex chromatographic separations, thereby preserving the integrity of the furan ring which is essential for the mechanical properties of the final PEF polymer.
How to Synthesize Dimethyl 2,5-Furandicarboxylate Efficiently
The synthesis protocol outlined in the patent provides a robust framework for laboratory and pilot-scale production of FDMC. The process begins by dissolving 5-hydroxymethylfurfural in methanol to achieve a substrate concentration between 0.5 and 20 wt%, followed by the addition of the optimized Co7Cu3-N-C catalyst. The mixture is then subjected to an oxygen or air atmosphere at a pressure of 1 to 5 bar and heated to 80°C for approximately 4 hours. This standardized procedure ensures reproducible results and high throughput, making it an ideal candidate for technology transfer. Detailed standardized synthesis steps see the guide below.
- Mix 5-hydroxymethylfurfural (HMF) with a lower alcohol solvent such as methanol or ethanol in a reaction vessel.
- Add the nitrogen-doped carbon material-loaded cobalt-based bimetallic catalyst (e.g., Co7Cu3-N-C) to the mixture.
- React under closed conditions with air or oxygen at 60-120°C and 1-5 bar pressure for 1-6 hours to obtain the diester.
Commercial Advantages for Procurement and Supply Chain Teams
From a procurement perspective, the substitution of noble metals with iron-group metals represents a paradigm shift in cost structure for fine chemical manufacturing. The elimination of Palladium and Gold from the catalyst formulation removes a major source of price volatility and supply risk, as these precious metals are subject to geopolitical fluctuations and limited geological availability. Furthermore, the ability to use air as an oxidant rather than pure oxygen or stoichiometric chemical oxidants significantly reduces utility costs and simplifies gas handling infrastructure. This translates to substantial cost savings in renewable polymer manufacturing, allowing buyers to secure long-term contracts with more predictable pricing models. The mild reaction conditions also reduce energy consumption, contributing to a lower carbon footprint which is increasingly a prerequisite for supplying major consumer goods corporations.
- Cost Reduction in Manufacturing: The deployment of non-precious metal catalysts fundamentally alters the economic equation of FDCA diester production. By removing the need for expensive noble metals, the initial capital expenditure for catalyst loading is drastically reduced, and the risk of metal theft or loss during recycling is eliminated. Additionally, the high selectivity of the bimetallic system minimizes waste generation, reducing the costs associated with waste treatment and raw material loss. This efficiency allows for a more competitive pricing strategy against petroleum-based terephthalates, facilitating market penetration for bio-based alternatives without sacrificing margin.
- Enhanced Supply Chain Reliability: The reliance on earth-abundant metals such as Cobalt, Copper, and Iron ensures a stable and resilient supply chain, free from the bottlenecks often associated with rare earth or precious metal sourcing. The catalyst preparation method, utilizing commercially available precursors like zinc nitrate and 2-methylimidazole, further decentralizes the supply risk. For supply chain heads, this means reduced lead time for high-purity bio-based polymer intermediates, as the raw materials are globally sourced and not subject to the same export restrictions or mining constraints as platinum group metals. The robustness of the catalyst also implies longer cycle life, reducing the frequency of catalyst replenishment orders.
- Scalability and Environmental Compliance: The process operates at low pressures (1-5 bar) and moderate temperatures, which significantly lowers the engineering requirements for reactor vessels compared to high-pressure hydrogenation processes. This ease of scale-up facilitates the commercial scale-up of complex furan derivatives from kilogram to multi-ton scales with minimal retrofitting of existing facilities. Moreover, the use of molecular oxygen generates water as the only byproduct, aligning with stringent environmental regulations and green chemistry principles. This compliance reduces the regulatory burden and permitting time for new production lines, accelerating time-to-market for new sustainable material initiatives.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this catalytic system. These insights are derived directly from the experimental data and claims within the patent documentation, providing a factual basis for decision-making. Understanding these nuances is essential for evaluating the feasibility of integrating this technology into existing production workflows.
Q: What are the advantages of using non-noble metal catalysts for FDCA diester production?
A: Using non-noble metal catalysts like Cobalt-Copper systems significantly reduces raw material costs compared to traditional Palladium or Gold catalysts, while maintaining high conversion rates and selectivity under mild conditions.
Q: Can this process utilize different alcohol solvents?
A: Yes, the method is versatile and supports various lower alcohols including methanol, ethanol, propanol, and butanol, allowing for the synthesis of different diester derivatives like FDMC, FDEC, and FDPC.
Q: What are the typical reaction conditions for this oxidative esterification?
A: The reaction typically operates at temperatures between 60-120°C and pressures of 1-5 bar using air or oxygen, which simplifies equipment requirements and enhances operational safety.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Bio-Based Polymer Intermediate Supplier
NINGBO INNO PHARMCHEM stands at the forefront of translating advanced academic research into commercial reality, possessing extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production. Our technical team is adept at optimizing the cobalt-bimetallic catalyst formulations described in CN110590721B to meet stringent purity specifications required for polymerization-grade monomers. With rigorous QC labs and a commitment to process safety, we ensure that every batch of 2,5-furandicarboxylic acid diester delivered meets the highest international standards, supporting your transition to a sustainable supply chain.
We invite you to engage with our technical procurement team to discuss how this innovative oxidative esterification technology can be tailored to your specific volume requirements. By requesting a Customized Cost-Saving Analysis, you can quantify the potential economic benefits of switching to this non-noble metal catalytic route. We encourage potential partners to contact us directly to obtain specific COA data and route feasibility assessments, ensuring a seamless integration of these high-performance bio-based intermediates into your manufacturing portfolio.
