Advanced Cobalt-Catalyzed Synthesis of Butyrolactones: A Breakthrough for Commercial Scale-Up
Advanced Cobalt-Catalyzed Synthesis of Butyrolactones: A Breakthrough for Commercial Scale-Up
The pharmaceutical and fine chemical industries are constantly seeking more efficient, sustainable, and cost-effective pathways to construct essential heterocyclic scaffolds. A pivotal advancement in this domain is detailed in patent CN111393394B, which discloses a novel synthetic method for butyrolactone compounds. This technology leverages a cobalt-catalyzed carbonyl insertion ring-expansion reaction, transforming readily available oxetane derivatives into valuable butyrolactones under a syngas atmosphere. Unlike traditional approaches that rely on stoichiometric oxidants or scarce precious metals, this invention utilizes earth-abundant cobalt complexes to achieve high atom economy and exceptional substrate tolerance. For R&D directors and procurement managers alike, this represents a paradigm shift towards greener, more economical manufacturing of key pharmaceutical intermediates such as those found in Baclofen and Pregabalin precursors.
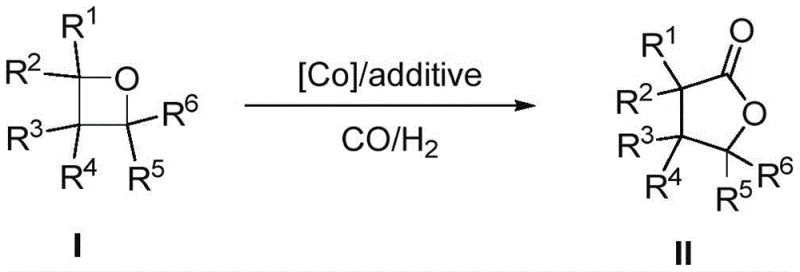
The core innovation lies in the specific reaction conditions that enable the cleavage of the strained oxetane ring and the subsequent insertion of a carbonyl group. As illustrated in the general reaction scheme, the process accommodates a wide variety of substituents (R1-R6), ranging from simple alkyl groups to complex aryl and heteroaryl moieties. This structural flexibility is critical for medicinal chemists who require diverse libraries of analogues for structure-activity relationship (SAR) studies. By establishing a reliable protocol that functions effectively with base metal catalysis, this patent addresses the long-standing challenge of balancing reactivity with cost-efficiency in lactone synthesis.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of γ-butyrolactones has been plagued by significant operational and economic hurdles. Traditional routes often involve the Baeyer-Villiger oxidation of cyclobutanones, a process that necessitates the use of hazardous, stoichiometric oxidizing agents like hydrogen peroxide or peracids. These reagents not only generate substantial waste streams, complicating environmental compliance, but also pose safety risks during scale-up due to their exothermic nature. Alternatively, nucleophilic additions to furanones catalyzed by rhodium complexes have been employed; however, the reliance on rhodium, a precious metal with volatile pricing and supply chain vulnerabilities, renders these methods economically unattractive for large-volume production. Furthermore, earlier attempts at oxetane carbonylation, such as those reported by Alper in the late 1980s, required extreme conditions—temperatures exceeding 165°C and pressures up to 60 atm—along with dual catalytic systems involving ruthenium. Such harsh parameters limit the functional group tolerance and increase the capital expenditure required for specialized high-pressure reactors.
The Novel Approach
In stark contrast, the methodology described in CN111393394B offers a streamlined solution that mitigates these drawbacks through the strategic use of a cobalt catalyst under syngas conditions. By introducing hydrogen gas alongside carbon monoxide, the reaction achieves superior catalytic activity at significantly milder temperatures, typically around 100°C. This reduction in thermal demand not only lowers energy consumption but also preserves sensitive functional groups that might degrade under harsher conditions. The elimination of precious metals like rhodium and ruthenium is a game-changer for cost reduction in pharmaceutical intermediate manufacturing, as it decouples production costs from the fluctuating markets of noble metals. Moreover, the system demonstrates remarkable versatility, successfully converting a broad spectrum of oxetane substrates—including those with electron-withdrawing and electron-donating groups—into the corresponding lactones with high selectivity.
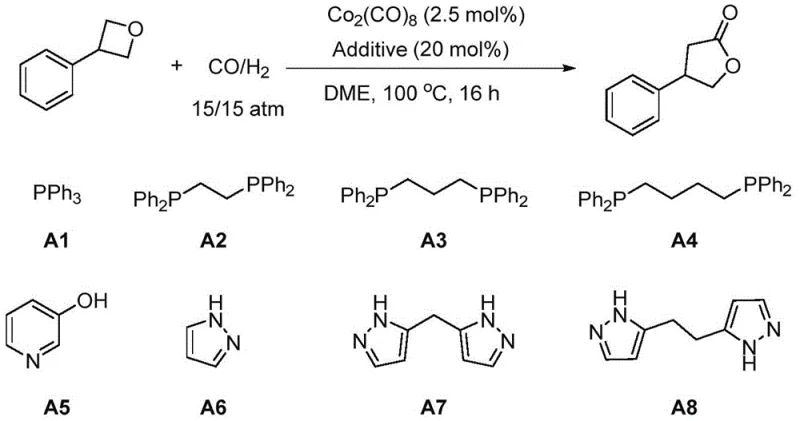
Mechanistic Insights into Cobalt-Catalyzed Carbonylative Ring Expansion
Understanding the mechanistic underpinnings of this transformation is vital for optimizing process parameters and ensuring consistent quality. The reaction proceeds via a cobalt-mediated cycle where the active catalytic species, likely a cobalt hydride or cobalt carbonyl complex generated in situ from precursors like Co2(CO)8, coordinates with the oxygen atom of the oxetane ring. This coordination weakens the C-O bond, facilitating ring opening. The presence of hydrogen is crucial; it assists in the generation of the active hydride species and helps maintain the catalyst in its reduced, active state, preventing the formation of inactive cobalt clusters. Following ring opening, carbon monoxide inserts into the cobalt-carbon bond, extending the carbon chain by one unit. The cycle concludes with reductive elimination or nucleophilic attack by the oxygen to close the five-membered lactone ring, regenerating the catalyst.
Impurity control is another area where this method excels, largely due to the specific choice of additives. The patent highlights the efficacy of nitrogen-containing ligands, particularly bis-pyrazole derivatives (labeled as A8 in the experimental data), which act as promoters to enhance both conversion rates and selectivity. These additives stabilize the cobalt center and modulate its electronic properties, suppressing side reactions such as polymerization or over-reduction that could lead to alcohol byproducts. By fine-tuning the ligand environment, manufacturers can achieve high purity profiles, minimizing the burden on downstream purification steps like chromatography or recrystallization. This level of control is essential for meeting the stringent impurity specifications required for API intermediates.
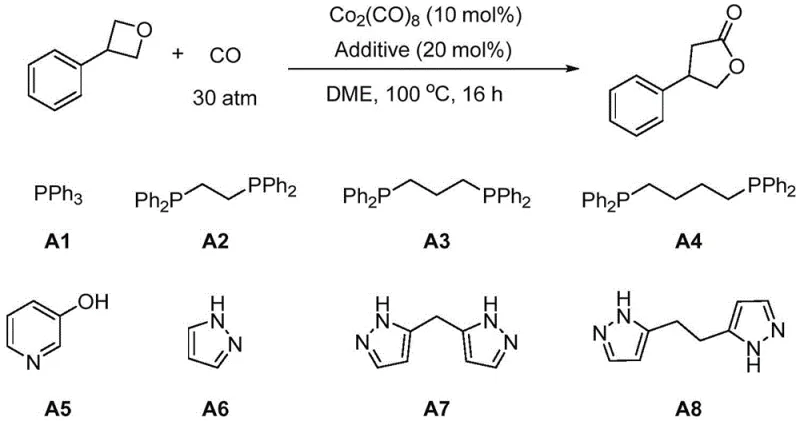
How to Synthesize 4-Phenylbutyrolactone Efficiently
Implementing this synthesis route requires careful attention to reaction setup and safety protocols, given the use of pressurized gases. The standard procedure involves charging a high-pressure autoclave with the cobalt precursor and the optimized additive under an inert atmosphere to prevent catalyst deactivation by oxygen. Once the substrate and solvent are added, the vessel is pressurized with the specific CO/H2 ratio and heated. While the general principles are straightforward, precise control over temperature and pressure is necessary to maximize yield. For detailed operational parameters and safety guidelines, please refer to the standardized synthesis steps provided below.
- Prepare the reaction mixture by adding dicobalt octacarbonyl (Co2(CO)8) and a specific nitrogen-based additive (such as bis-pyrazole ligands) into a high-pressure reactor under inert atmosphere.
- Introduce the oxetane substrate and solvent (e.g., DME or THF) into the reactor, then seal and pressurize with a mixture of Carbon Monoxide and Hydrogen (syngas) to 15-30 atm.
- Heat the reaction mixture to 100°C for approximately 16 hours, then cool, depressurize, and purify the resulting butyrolactone product via distillation or chromatography.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this cobalt-catalyzed technology translates into tangible strategic benefits beyond mere chemical efficiency. The shift from precious metal catalysis to base metal systems fundamentally alters the cost structure of the supply chain. By eliminating the need for rhodium or ruthenium, companies can avoid the significant capital lock-up associated with purchasing and recovering expensive catalysts. This change leads to substantial cost savings in raw material procurement and simplifies the logistics of catalyst management. Furthermore, the mild reaction conditions reduce the wear and tear on production equipment, extending the lifecycle of reactors and lowering maintenance costs over time.
- Cost Reduction in Manufacturing: The most immediate impact is the drastic reduction in catalyst costs. Cobalt salts and carbonyls are orders of magnitude cheaper than rhodium complexes, and they do not require the same level of rigorous recovery processes to be economically viable. Additionally, the high atom economy of the carbonylation reaction means that fewer raw materials are wasted, further driving down the cost per kilogram of the final product. The ability to run the reaction at lower temperatures also results in significant energy savings, contributing to a leaner manufacturing budget.
- Enhanced Supply Chain Reliability: Relying on abundant base metals insulates the supply chain from the geopolitical and market volatilities that often plague precious metal supplies. Cobalt is widely available and its supply chain is more robust compared to the concentrated sources of platinum group metals. This stability ensures consistent production schedules and reduces the risk of delays caused by catalyst shortages. Moreover, the broad substrate scope allows for the sourcing of diverse oxetane starting materials, providing flexibility in raw material procurement.
- Scalability and Environmental Compliance: The process is inherently scalable, having been demonstrated to work effectively in batch reactors suitable for pilot and commercial scales. The absence of stoichiometric oxidants eliminates the generation of hazardous waste streams associated with Baeyer-Villiger oxidations, simplifying waste treatment and disposal. This aligns with modern green chemistry principles and helps facilities meet increasingly strict environmental regulations, avoiding potential fines and reputational damage while fostering a sustainable corporate image.
Frequently Asked Questions (FAQ)
To assist technical teams in evaluating this technology for their specific applications, we have compiled answers to common questions regarding the process capabilities and limitations. These insights are derived directly from the experimental data and technical specifications outlined in the patent documentation, ensuring accuracy and relevance for process development planning.
Q: What are the primary advantages of this cobalt-catalyzed method over traditional rhodium catalysis?
A: The primary advantage is the replacement of expensive precious metals like rhodium or ruthenium with abundant and cost-effective cobalt. Additionally, this method operates under milder temperatures (100°C) compared to historical methods requiring up to 190°C, significantly reducing energy consumption and safety risks.
Q: Does this synthesis method require high pressures of carbon monoxide?
A: No, the process is optimized to work efficiently at moderate pressures ranging from 15 to 30 atmospheres. The introduction of hydrogen gas alongside carbon monoxide (syngas) enhances catalytic activity, allowing for lower pressure operation compared to pure CO protocols.
Q: Is this method suitable for large-scale industrial production?
A: Yes, the use of stable cobalt catalysts and common solvents like DME or THF makes this process highly scalable. The avoidance of sensitive oxidizing agents and the robustness of the catalyst system support commercial scale-up from kilogram to multi-ton production.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Butyrolactone Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of advanced catalytic methodologies like the one described in CN111393394B. As a leading CDMO partner, we possess the technical expertise and infrastructure to translate such innovative laboratory protocols into robust commercial processes. Our team has extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your transition from R&D to market is seamless. We are committed to delivering high-purity butyrolactone intermediates that meet stringent purity specifications, supported by our rigorous QC labs and state-of-the-art analytical capabilities.
We invite you to collaborate with us to leverage this cost-effective synthesis route for your next project. Whether you require custom synthesis services or bulk supply of pharmaceutical intermediates, our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your volume requirements. Contact us today to request specific COA data and route feasibility assessments, and let us help you optimize your supply chain with superior chemical solutions.
