Advanced Synthetic Route For 40000X Sweetener Enabling Commercial Scale-Up And Cost Efficiency
Advanced Synthetic Route For 40000X Sweetener Enabling Commercial Scale-Up And Cost Efficiency
The global demand for low-calorie, high-intensity sweeteners is driving significant innovation in the food additive sector, particularly for compounds that offer superior stability and sweetness profiles compared to traditional options like aspartame. Patent CN110818774A discloses a groundbreaking synthetic methodology for producing N-{N-[3-(3-hydroxy-4-methoxyphenyl)propyl]-L-α-aspartyl}-S-tert-butyl-L-cysteine-1-methyl ester, a dipeptide derivative exhibiting sweetness approximately 40,000 times that of sucrose. This technical breakthrough addresses critical limitations in current sweetener manufacturing by providing a route that avoids complex high-pressure hydrogenation steps often associated with earlier generations of dipeptide sweeteners. The structural integrity and functional groups of this molecule are critical for its metabolic stability and intense sweetening power, making it a highly attractive candidate for next-generation food formulations. ![Chemical structure of N-{N-[3-(3-hydroxy-4-methoxyphenyl)propyl]-L-α-aspartyl}-S-tert-butyl-L-cysteine-1-methyl ester showing the dipeptide backbone and aromatic side chain](/insights/img/high-potency-sweetener-synthesis-food-additive-supplier-20260306045857-01.webp)
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional synthesis pathways for high-potency dipeptide sweeteners often rely on cumbersome multi-step sequences that involve hazardous reagents or extreme reaction conditions which complicate industrial scale-up. Many existing processes for similar compounds require high-pressure hydrogenation equipment that poses significant safety risks and capital expenditure challenges for manufacturers attempting to enter the market. Furthermore, conventional routes frequently suffer from poor regioselectivity during the coupling of amino acid derivatives, leading to difficult-to-separate impurities that compromise the sensory profile of the final food additive. The reliance on unstable intermediates in older methodologies often results in lower overall yields and increased waste generation, which contradicts modern green chemistry principles essential for sustainable food ingredient production.
The Novel Approach
The methodology described in the patent introduces a streamlined convergent synthesis that strategically constructs the molecule from two key advanced intermediates, significantly enhancing process efficiency and control. By utilizing a Wittig reaction to establish the carbon chain length followed by a highly selective DIBAL-H reduction, the process ensures the precise formation of the critical aldehyde intermediate without over-reduction to the alcohol. This approach allows for the modular assembly of the complex dipeptide structure, enabling better quality control over stereochemistry and purity. The final coupling step utilizes mild reductive amination conditions which are far more operationally convenient than previous high-pressure alternatives, thereby reducing the technical barrier for commercial adoption. 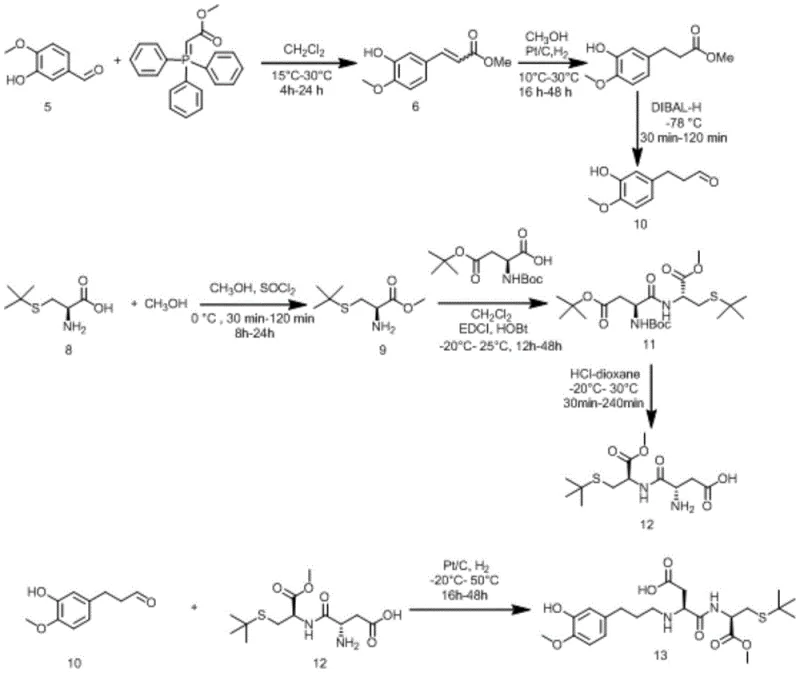
Mechanistic Insights into Peptide Coupling and Reductive Amination
The core of this synthesis relies on the efficient formation of the peptide bond between the aspartic acid derivative and the cysteine moiety, facilitated by carbodiimide chemistry using EDCI and HOBt. This activation strategy minimizes racemization of the chiral centers, which is paramount for maintaining the specific receptor binding affinity required for the intense sweet taste. The subsequent deprotection of the tert-butyl groups under acidic conditions must be carefully controlled to prevent hydrolysis of the methyl ester, demonstrating the need for precise pH management during the workup phase. The mechanistic pathway ensures that the sensitive thioether linkage in the cysteine residue remains intact throughout the synthesis, preserving the structural features necessary for the molecule's metabolic stability and lack of caloric contribution.
Furthermore, the final reductive amination step represents a critical junction where the aromatic aldehyde and the free amine of the dipeptide are joined under catalytic hydrogenation conditions. The use of palladium on carbon (Pd/C) at ambient pressure and temperature allows for the in situ formation of the Schiff base followed by immediate reduction, driving the equilibrium towards the desired secondary amine product. This one-pot transformation eliminates the need for isolating the unstable imine intermediate, thereby reducing processing time and solvent consumption. The selectivity of the catalyst ensures that other reducible functional groups, such as the aromatic ring or the ester moieties, remain unaffected, resulting in a clean reaction profile that simplifies downstream purification and enhances the overall yield of the high-value sweetener.
How to Synthesize N-{N-[3-(3-hydroxy-4-methoxyphenyl)propyl]-L-α-aspartyl}-S-tert-butyl-L-cysteine-1-methyl ester Efficiently
The synthesis protocol outlined in the patent provides a robust framework for producing this high-intensity sweetener with high fidelity and reproducibility suitable for pilot and commercial scales. The process begins with the preparation of the aromatic aldehyde fragment via a Wittig olefination of isovanillin, followed by catalytic hydrogenation and selective reduction to the aldehyde, ensuring a steady supply of the key electrophile. Parallel to this, the amino acid fragment is constructed through esterification and peptide coupling, requiring strict anhydrous conditions to maximize the efficiency of the EDCI-mediated bond formation. Detailed standardized synthesis steps see the guide below for specific molar ratios and temperature controls.
- Preparation of 3-(3-hydroxy-4-methoxyphenyl)propionaldehyde via Wittig reaction followed by hydrogenation and DIBAL-H reduction.
- Synthesis of N-(L-α-aspartyl)-S-tert-butyl-L-cysteine-1-methyl ester through esterification and EDCI/HOBt mediated peptide coupling.
- Final condensation of the aldehyde and amine intermediates via palladium-catalyzed reductive amination to yield the target sweetener.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this synthetic route offers tangible benefits regarding raw material security and operational cost structures. The starting materials, such as isovanillin and protected amino acids, are commodity chemicals available from multiple global suppliers, mitigating the risk of supply chain disruptions common with exotic or proprietary precursors. The elimination of specialized high-pressure reactor requirements lowers the barrier to entry for contract manufacturing organizations, allowing for a broader base of qualified suppliers and fostering competitive pricing dynamics in the market. Additionally, the simplified workup procedures reduce the consumption of solvents and energy, aligning with corporate sustainability goals while simultaneously lowering the variable costs associated with utility consumption and waste disposal.
- Cost Reduction in Manufacturing: The process achieves significant material efficiency through high-yielding steps, such as the initial Wittig reaction which reportedly achieves yields up to 97 percent in experimental examples, minimizing the loss of expensive chiral starting materials. By avoiding the need for cryogenic conditions in all but one specific reduction step, the process reduces the energy load on manufacturing facilities, leading to lower overhead costs per kilogram of produced sweetener. The use of standard heterogeneous catalysts like Pd/C allows for easy recovery and potential recycling of the precious metal, further contributing to long-term cost optimization strategies without compromising reaction kinetics or product quality.
- Enhanced Supply Chain Reliability: The modular nature of the synthesis allows for the stocking of key intermediates, such as the protected dipeptide and the aromatic aldehyde, which can be produced independently and stored until needed for the final coupling. This decoupling of production stages enhances flexibility in responding to fluctuating market demands, ensuring that finished goods inventory can be ramped up quickly without waiting for long linear synthesis cycles. Furthermore, the stability of the intermediates under standard storage conditions reduces the risk of spoilage or degradation during transit, ensuring consistent quality upon arrival at the final formulation site.
- Scalability and Environmental Compliance: The synthetic route is designed with industrial scalability in mind, utilizing reaction conditions that are easily transferable from laboratory glassware to large-scale stainless steel reactors without significant re-engineering. The absence of heavy metal catalysts other than the recoverable palladium, and the use of relatively benign solvents like methanol and dichloromethane (which can be managed with standard abatement systems), simplifies regulatory compliance and environmental permitting. This green chemistry profile not only reduces the environmental footprint but also future-proofs the supply chain against increasingly stringent global regulations on chemical manufacturing emissions and waste.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production and application of this novel sweetener, derived directly from the patent specifications and industry standards. Understanding these details is crucial for R&D teams evaluating the feasibility of incorporating this ingredient into new product lines and for quality assurance teams establishing specification limits. The answers reflect the specific advantages of the disclosed synthetic method over prior art technologies.
Q: What distinguishes this 40000x sweetener from existing options like Neotame?
A: This compound features a unique S-tert-butyl-L-cysteine moiety which enhances thermal stability and sweetness potency significantly beyond third-generation sweeteners, offering superior performance in acidic food matrices.
Q: Is the synthesis route scalable for industrial production?
A: Yes, the patent outlines a route using commercially available raw materials like isovanillin and standard reagents (EDCI, Pd/C), avoiding exotic catalysts, which facilitates scaling from kilogram to metric ton quantities.
Q: How does the process ensure high purity for food grade application?
A: The method employs rigorous purification steps including column chromatography and controlled crystallization, alongside specific reaction conditions like low-temperature DIBAL-H reduction to minimize side products and ensure high chemical purity.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable N-{N-[3-(3-hydroxy-4-methoxyphenyl)propyl]-L-α-aspartyl}-S-tert-butyl-L-cysteine-1-methyl ester Supplier
As a leader in fine chemical manufacturing, NINGBO INNO PHARMCHEM possesses the technical expertise to translate this patented laboratory methodology into a robust commercial reality. Our facility is equipped with the necessary infrastructure to handle sensitive reactions such as low-temperature reductions and hydrogenations safely and efficiently, ensuring consistent batch-to-batch quality. We have extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, leveraging our rigorous QC labs to maintain stringent purity specifications required for food-grade ingredients. Our commitment to process optimization ensures that we can deliver this high-value sweetener at a price point that makes it viable for mass-market applications.
We invite potential partners to engage with our technical procurement team to discuss how this innovative sweetener can enhance your product portfolio. By requesting a Customized Cost-Saving Analysis, you can gain insights into how our optimized manufacturing process can reduce your overall ingredient costs while improving product performance. We encourage you to contact us today to obtain specific COA data and route feasibility assessments tailored to your specific volume requirements and application needs.
