Advanced Synthesis of N-Salicyloyl-8-Aminocaprylic Acid Sodium for Oral Drug Delivery Systems
Advanced Synthesis of N-Salicyloyl-8-Aminocaprylic Acid Sodium for Oral Drug Delivery Systems
The pharmaceutical industry continuously seeks robust and efficient synthetic routes for critical absorption enhancers that enable the oral delivery of macromolecular drugs. Patent CN1446795A introduces a refined methodology for synthesizing N-salicyloyl-8-aminocaprylic acid sodium, commonly known as SNAC, which serves as a pivotal promoter for increasing gastrointestinal permeability of therapeutic agents such as insulin and heparin. This technical insight report dissects the patented process, highlighting its strategic advantages over legacy methods through a detailed analysis of reaction mechanisms and operational parameters. By leveraging a direct acylation strategy starting from salicylic acid, the disclosed method achieves superior yield profiles while maintaining mild reaction conditions that are essential for preserving the integrity of sensitive functional groups. The structural complexity of the target molecule requires precise control over stoichiometry and temperature to minimize impurity formation, a challenge that this new protocol addresses effectively through optimized reagent selection and catalytic enhancement.
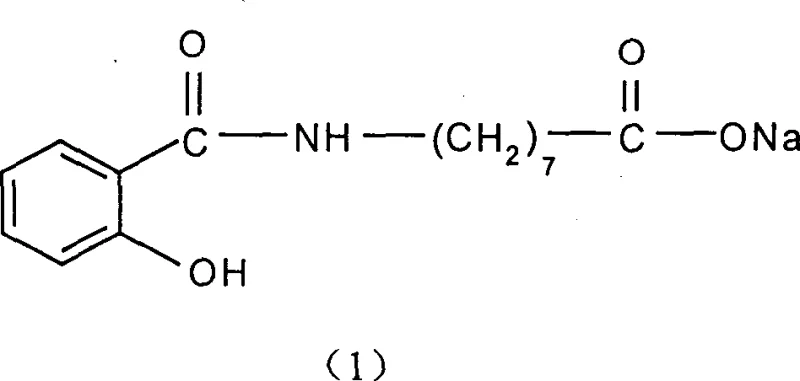
For R&D directors evaluating process feasibility, the transition from multi-step protection-deprotection sequences to a streamlined acyl chloride pathway represents a significant reduction in process mass intensity. The ability to generate the target compound with yields exceeding 68% without requiring extreme thermal energy input underscores the potential for this technology to redefine cost structures in the manufacturing of pharmaceutical intermediates. Furthermore, the compatibility of this synthesis with standard industrial equipment reduces the barrier to entry for commercial scale-up, ensuring a reliable supply chain for downstream drug formulation developers who depend on high-purity excipients.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Prior art methodologies for producing N-salicyloyl derivatives often suffered from severe thermodynamic and kinetic limitations that hindered their commercial viability. For instance, the polysalicylic acid system described in earlier literature necessitates reaction temperatures approaching 200°C, which not only consumes excessive energy but also promotes thermal degradation and the formation of complex polymeric byproducts that are difficult to separate. Another prevalent historical approach involved the use of acetylsalicylic acid as a starting material, which required an initial acylation followed by a hydrolysis step to reveal the phenolic hydroxyl group, thereby extending the synthetic timeline and introducing additional unit operations that cumulatively reduced the overall process yield to approximately 57%. These inefficiencies translate directly into higher production costs and increased waste generation, creating substantial bottlenecks for procurement teams aiming to secure cost-effective raw materials for large-scale API production.
The Novel Approach
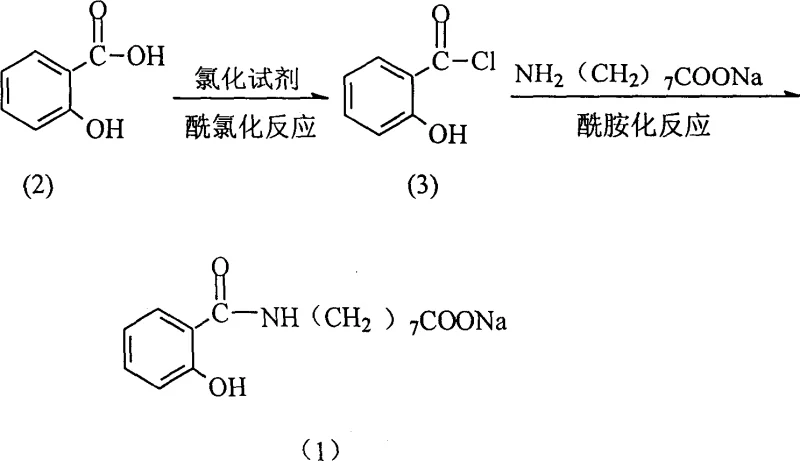
In stark contrast, the novel approach detailed in the patent utilizes a direct activation of salicylic acid via conversion to salicyloyl chloride, followed by immediate amidation with omega-aminocaprylic acid sodium. This two-step sequence eliminates the need for high-temperature polymerization or tedious deprotection cycles, allowing the reaction to proceed efficiently at temperatures ranging from 0°C to 80°C depending on the specific stage. The strategic selection of chlorinating agents such as thionyl chloride or oxalyl chloride ensures rapid conversion of the carboxylic acid group into a highly reactive electrophile, which then undergoes nucleophilic attack by the amine functionality of the amino acid derivative. This streamlined pathway not only simplifies the purification workflow but also significantly enhances the atom economy of the process, providing a clear competitive advantage for manufacturers seeking to optimize their production lines for high-value pharmaceutical intermediates.
Mechanistic Insights into Acyl Chloride Mediated Amidation
The core of this synthesis lies in the efficient generation of the acyl chloride intermediate, which serves as the activated species for the subsequent amide bond formation. When salicylic acid reacts with thionyl chloride, the hydroxyl group of the carboxylic acid attacks the sulfur atom, leading to the displacement of chloride ions and the eventual release of sulfur dioxide and hydrogen chloride gases. This gas evolution drives the equilibrium forward, ensuring near-quantitative conversion of the starting material into salicyloyl chloride, provided that the reaction is conducted under an inert atmosphere to prevent hydrolysis by atmospheric moisture. The presence of the ortho-hydroxyl group on the aromatic ring adds a layer of complexity, as it can potentially participate in intramolecular hydrogen bonding or side reactions, but the controlled temperature range of 30-40°C mitigates these risks while maintaining sufficient kinetic energy for the transformation.
Following the formation of the acyl chloride, the amidation step involves the nucleophilic attack of the primary amine from the omega-aminocaprylic acid sodium on the carbonyl carbon of the acyl chloride. This step is exothermic and requires careful temperature control, typically maintained between 0°C and 10°C, to prevent runaway reactions and ensure the selectivity of the amide bond formation over potential esterification or other side reactions. The inclusion of anhydrous aluminum trichloride as a Lewis acid catalyst further enhances the electrophilicity of the carbonyl carbon, facilitating the attack by the amine nucleophile and resulting in improved yields of up to 72.4%. This mechanistic understanding allows process chemists to fine-tune reaction parameters such as addition rates and stirring speeds to maximize throughput while minimizing the generation of impurities that could compromise the safety profile of the final drug product.
How to Synthesize N-Salicyloyl-8-Aminocaprylic Acid Sodium Efficiently
The execution of this synthesis requires strict adherence to anhydrous conditions and precise stoichiometric control to achieve the reported high yields and purity levels. The process begins with the activation of salicylic acid in a dry solvent like 1,4-dioxane, where it is treated with a slight excess of thionyl chloride to ensure complete conversion to the acid chloride. After the evolution of gases ceases, the reaction mixture is purged with nitrogen to remove residual acidic vapors before the introduction of the amine component. The subsequent amidation is performed by adding a solution of omega-aminocaprylic acid sodium dropwise to the cooled acyl chloride solution, maintaining the temperature below 10°C to control the exotherm. Detailed standardized operating procedures for scaling this reaction from laboratory to pilot plant are critical for maintaining consistency, and the specific step-by-step guidelines are outlined below for technical reference.
- React salicylic acid with a chlorinating agent like thionyl chloride at 30-40°C to form salicyloyl chloride.
- Purge excess reagents and gases with nitrogen to ensure a clean reaction environment for the next step.
- Perform amidation by reacting the salicyloyl chloride intermediate with omega-aminocaprylic acid sodium at 0-10°C.
Commercial Advantages for Procurement and Supply Chain Teams
From a supply chain perspective, the adoption of this synthetic route offers profound benefits related to raw material availability and process safety. Salicylic acid is a commodity chemical produced on a massive global scale, ensuring a stable and cost-effective supply base that is less susceptible to market volatility compared to specialized protected amino acid derivatives. The elimination of high-temperature processing steps reduces the energy footprint of the manufacturing facility, aligning with modern sustainability goals and potentially lowering utility costs associated with heating and cooling systems. Furthermore, the simplified workup procedure, which involves straightforward pH adjustment and recrystallization, minimizes the consumption of organic solvents and reduces the volume of hazardous waste requiring disposal, thereby streamlining environmental compliance protocols.
- Cost Reduction in Manufacturing: The shift from a multi-step synthesis involving protection and deprotection to a direct acylation strategy drastically reduces the number of unit operations, leading to significant savings in labor, equipment time, and solvent usage. By avoiding the need for expensive protecting groups and the reagents required to install and remove them, the overall material cost per kilogram of the final product is substantially lowered. Additionally, the higher reaction yield means that less starting material is wasted, improving the overall mass balance and reducing the cost of goods sold for the manufacturer.
- Enhanced Supply Chain Reliability: Utilizing widely available commodity chemicals like salicylic acid and thionyl chloride mitigates the risk of supply disruptions that often plague specialty chemical markets. The robustness of the reaction conditions, which do not require exotic catalysts or extreme pressures, allows for production in a broader range of facilities, diversifying the potential supplier base and enhancing supply security. This reliability is crucial for pharmaceutical companies that require consistent quality and uninterrupted delivery of key excipients to maintain their own production schedules for finished dosage forms.
- Scalability and Environmental Compliance: The mild reaction temperatures and the absence of heavy metal catalysts simplify the scale-up process, as heat transfer and mixing requirements are less demanding than those for high-temperature polymerizations. The gaseous byproducts generated during the acyl chloride formation can be easily scrubbed and neutralized using standard abatement systems, ensuring that the process meets stringent environmental regulations. This ease of scale-up facilitates the rapid transition from clinical trial material to commercial production volumes, accelerating the time to market for new drug formulations utilizing this absorption enhancer.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the synthesis and application of N-salicyloyl-8-aminocaprylic acid sodium. These answers are derived directly from the experimental data and comparative analysis presented in the patent documentation, providing a factual basis for decision-making. Understanding these nuances helps stakeholders evaluate the feasibility of integrating this intermediate into their specific development pipelines.
Q: What are the advantages of the acyl chloride method over the polysalicylic acid system?
A: The acyl chloride method operates under significantly milder conditions (0-80°C) compared to the harsh temperatures (nearly 200°C) required for the polysalicylic acid system, resulting in higher yields and better product quality.
Q: How does the addition of aluminum trichloride affect the synthesis yield?
A: Adding a catalytic amount of anhydrous aluminum trichloride during the acyl chloride reaction step can increase the target product yield from approximately 68% to over 72% by facilitating the activation of the carboxyl group.
Q: Is this synthesis route suitable for large-scale commercial production?
A: Yes, the route utilizes readily available raw materials like salicylic acid and thionyl chloride, and avoids extreme temperatures, making it highly scalable and safer for industrial manufacturing environments.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable N-Salicyloyl-8-Aminocaprylic Acid Sodium Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-quality absorption enhancers play in the advancement of oral biologic therapies. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that we can meet the rigorous demands of global pharmaceutical clients. We are committed to delivering N-salicyloyl-8-aminocaprylic acid sodium with stringent purity specifications, supported by our rigorous QC labs that employ state-of-the-art analytical techniques to verify identity and assay. Our capability to replicate and optimize the patented acyl chloride route allows us to offer a product that balances performance with cost-efficiency, making us an ideal partner for your long-term supply needs.
We invite you to engage with our technical procurement team to discuss how our manufacturing capabilities can support your specific project requirements. By requesting a Customized Cost-Saving Analysis, you can gain deeper insights into how our optimized process can reduce your overall material costs. We encourage potential partners to contact us directly to obtain specific COA data and route feasibility assessments, ensuring that our solutions are perfectly aligned with your development timelines and quality standards.
