Revolutionizing Pyrazoline Production: Mechanochemical Synthesis for Commercial Scale-Up
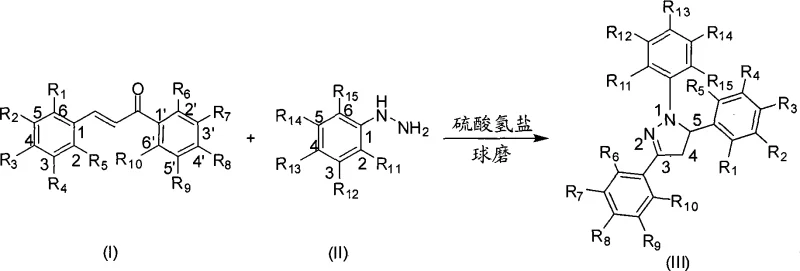
The landscape of fine chemical manufacturing is undergoing a paradigm shift towards greener, more efficient methodologies, as exemplified by the groundbreaking technology disclosed in patent CN101440065A. This patent introduces a novel mechanochemical preparation method for 1,3,5-triaryl-2-pyrazoline compounds, a class of heterocycles renowned for their extensive biological activities including anti-inflammatory, analgesic, and antibacterial properties. Unlike traditional synthetic routes that rely heavily on volatile organic solvents and harsh thermal conditions, this innovation leverages high-energy mechanical forces to drive the cyclocondensation reaction between chalcone derivatives and phenylhydrazine compounds. By utilizing bisulfate salts as catalysts and silica gel as a grinding aid within a closed ball milling system, the process achieves remarkable reaction rates and yields while completely eliminating solvent pollution. For R&D directors and procurement strategists alike, this represents a critical advancement in the reliable supply of high-purity pharmaceutical intermediates, offering a pathway to significantly reduce both environmental footprint and production costs without compromising on chemical quality or structural integrity.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of 1,3,5-triaryl-2-pyrazolines has been dominated by solution-phase chemistry that necessitates the use of strong acids like acetic acid or hydrochloric acid, or strong bases such as sodium hydroxide, often under reflux conditions. These conventional methods suffer from inherent inefficiencies, including prolonged reaction times that can extend over several hours, leading to increased energy consumption and lower throughput in a manufacturing setting. Furthermore, the reliance on large volumes of organic solvents creates substantial downstream burdens related to solvent recovery, waste disposal, and environmental compliance, which directly inflate the operational expenditure for chemical producers. The use of corrosive reagents also poses significant safety risks to equipment and personnel, while the thermal stress imposed on sensitive functional groups can sometimes lead to unwanted side reactions and a broader impurity profile that complicates purification. Consequently, the industry has long sought a more sustainable alternative that could overcome these thermodynamic and kinetic barriers without sacrificing the versatility needed to accommodate diverse substrate substitutions.
The Novel Approach
The mechanochemical strategy outlined in the patent data fundamentally reimagines the reaction environment by replacing bulk solvent media with solid-state grinding, thereby creating a microenvironment of extremely high local concentration at the collision points of the grinding media. This approach utilizes mild bisulfate catalysts, such as potassium bisulfate or sodium bisulfate, which are sufficiently acidic to promote the cyclization but benign enough to avoid the severe corrosion issues associated with liquid mineral acids. The addition of silica gel acts not merely as an inert filler but as a crucial grinding aid that facilitates the transfer of mechanical energy to the reactant molecules, effectively lowering the activation energy barrier for the formation of the pyrazoline ring. By operating at room temperature or with minimal thermal input generated solely by friction, this method drastically shortens the reaction window to mere minutes, typically between 2 to 20 minutes, while maintaining excellent yields across a wide variety of substituted chalcones and hydrazines. This transition from thermal to mechanical activation marks a significant leap forward in process intensification, making it an ideal candidate for cost reduction in fine chemical manufacturing.
Mechanistic Insights into Mechanochemical Cyclocondensation
The underlying mechanism of this solvent-free transformation relies on the unique physicochemical phenomena induced by high-speed ball milling, where kinetic energy is directly converted into chemical potential through repeated impact and shear forces. In the absence of a solvent cage, the reactant molecules—chalcone and phenylhydrazine—are forced into intimate contact with the solid acid catalyst, creating transient hot spots of high pressure and temperature that accelerate the nucleophilic attack of the hydrazine nitrogen on the enone system of the chalcone. The bisulfate anion likely serves to protonate the carbonyl oxygen or the beta-carbon of the chalcone, increasing its electrophilicity and facilitating the subsequent intramolecular cyclization to form the five-membered pyrazoline ring. This mechanical activation bypasses the diffusion limitations typical of solution chemistry, allowing the reaction to proceed with exceptional speed and selectivity even for sterically hindered or electronically deactivated substrates. The result is a highly efficient catalytic cycle that minimizes the formation of by-products, thereby simplifying the isolation of the target molecule and ensuring a cleaner crude reaction mixture.
Impurity control in this mechanochemical system is inherently superior due to the absence of solvent-mediated side reactions and the precise control over reaction time afforded by the milling process. In traditional reflux methods, prolonged exposure to heat and acidic media can lead to decomposition of sensitive functional groups or polymerization of the starting materials, whereas the rapid nature of ball milling limits the residence time of the product in the reactive environment. Furthermore, the solid-state nature of the reaction prevents the solvation of intermediate species that might otherwise lead to alternative reaction pathways, thus channeling the conversion almost exclusively towards the desired 1,3,5-triaryl-2-pyrazoline structure. The use of silica gel also aids in adsorbing any minor liquid by-products or moisture, keeping the reaction matrix dry and further enhancing the selectivity. For quality control teams, this means that the resulting material exhibits a consistent and predictable impurity profile, reducing the need for extensive chromatographic purification and enabling direct crystallization from simple alcohol washes.
How to Synthesize 1,3,5-Triaryl-2-Pyrazolines Efficiently
To implement this cutting-edge synthesis in a laboratory or pilot plant setting, operators must adhere to a streamlined protocol that maximizes the efficiency of the mechanical energy transfer while ensuring safety and reproducibility. The process begins with the precise weighing of the chalcone derivative and the corresponding phenylhydrazine compound, which are then combined with a catalytic amount of a bisulfate salt and a substantial excess of silica gel to serve as the grinding matrix.
- Load chalcone derivatives, phenylhydrazine compounds, and a bisulfate catalyst (such as KHSO4 or NaHSO4) into a closed ball milling tank with silica gel as a grinding aid.
- Operate the ball mill at high speeds ranging from 1290 to 2220 rpm for a short duration of 2 to 20 minutes to induce the cyclocondensation reaction without external heating.
- Wash the reaction mixture with hot ethanol, filter to remove silica gel, and concentrate the filtrate to crystallize the pure 1,3,5-triaryl-2-pyrazoline product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain leaders, the adoption of this mechanochemical technology offers profound strategic benefits that extend far beyond simple reaction kinetics, fundamentally altering the cost structure and risk profile of producing complex heterocyclic intermediates. By eliminating the need for organic solvents, manufacturers can bypass the volatile pricing markets associated with petrochemical-derived liquids and avoid the capital-intensive infrastructure required for solvent storage, recycling, and waste treatment. This solvent-free paradigm also drastically reduces the volumetric footprint of the production process, allowing for higher batch densities and more efficient use of reactor space, which translates directly into improved asset utilization and lower fixed costs per kilogram of product. Moreover, the simplified workup procedure, which involves basic filtration and crystallization rather than complex extraction and distillation sequences, significantly lowers labor requirements and utility consumption, contributing to a leaner and more agile manufacturing operation.
- Cost Reduction in Manufacturing: The economic impact of removing solvents and harsh reagents from the process cannot be overstated, as it eradicates the expenses linked to solvent purchase, recovery, and hazardous waste disposal. Traditional methods often incur hidden costs related to the degradation of equipment by strong acids and the energy required to maintain reflux temperatures over long periods, whereas this mechanochemical route operates at ambient temperatures with minimal energy input. The use of inexpensive and readily available catalysts like potassium bisulfate further drives down the raw material bill, while the high yields observed across diverse substrates ensure that precious starting materials are not wasted on side products. Consequently, the overall cost of goods sold (COGS) for these pyrazoline derivatives can be substantially optimized, providing a competitive edge in price-sensitive markets without compromising on quality standards.
- Enhanced Supply Chain Reliability: From a supply chain perspective, the robustness of this synthetic route ensures greater continuity of supply by reducing dependency on specialized reagents and complex logistical chains for solvent management. The raw materials required, specifically chalcones and phenylhydrazines, are commodity chemicals with well-established global supply networks, minimizing the risk of bottlenecks or shortages that could disrupt production schedules. Additionally, the stability of the solid-state reaction mixture allows for greater flexibility in scheduling and batching, as there is no immediate urgency to process solvent-containing solutions that might degrade over time. This resilience makes the supply chain more adaptable to fluctuating demand patterns, ensuring that customers receive their orders of high-purity pharmaceutical intermediates with consistent lead times and reliability.
- Scalability and Environmental Compliance: Scaling this process from gram to ton quantities is inherently safer and more straightforward because it avoids the thermal runaway risks and explosion hazards associated with heating large volumes of flammable solvents. The mechanochemical approach aligns perfectly with increasingly stringent environmental regulations regarding volatile organic compound (VOC) emissions and hazardous waste generation, positioning manufacturers as leaders in sustainable chemistry. As regulatory bodies worldwide tighten restrictions on industrial effluents, adopting a near-zero waste technology future-proofs the production facility against compliance penalties and reputational damage. This environmental stewardship not only satisfies corporate social responsibility goals but also appeals to downstream partners who are increasingly auditing their suppliers for green credentials, thereby strengthening long-term business relationships.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this mechanochemical synthesis, drawing directly from the experimental data and beneficial effects reported in the patent literature. Understanding these details is crucial for technical teams evaluating the feasibility of integrating this method into existing production lines or for procurement specialists assessing the quality assurances associated with the final product.
Q: What are the primary advantages of using mechanochemistry for pyrazoline synthesis compared to traditional solvent-based methods?
A: The mechanochemical approach described in patent CN101440065A eliminates the need for volatile organic solvents and strong corrosive acids or bases typically required in reflux conditions. This results in a safer production environment, drastically reduced reaction times from hours to minutes, and simplified post-treatment processes that avoid complex solvent recovery systems.
Q: How does the use of bisulfate salts and silica gel impact the reaction yield and purity?
A: Bisulfate salts act as mild yet effective solid acid catalysts that facilitate the cyclization under mechanical force, while silica gel serves as a grinding aid to ensure uniform mixing and local high concentration. This combination allows for consistently high yields (often exceeding 90%) across a broad range of substrates, including those with electron-withdrawing or electron-donating groups, ensuring a robust impurity profile.
Q: Is this mechanochemical process suitable for large-scale industrial manufacturing?
A: Yes, the process is highly scalable because it operates under solvent-free conditions, which removes the safety hazards and engineering constraints associated with handling large volumes of flammable solvents at high temperatures. The simplicity of the workup procedure further enhances its viability for commercial scale-up, aligning with modern green chemistry principles and environmental regulations.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 1,3,5-Triaryl-2-Pyrazoline Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of the mechanochemical synthesis described in patent CN101440065A and have integrated similar green chemistry principles into our own manufacturing capabilities to serve the global pharmaceutical and agrochemical industries. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory innovation to industrial reality is seamless and efficient. We maintain stringent purity specifications and operate rigorous QC labs equipped with advanced analytical instrumentation to guarantee that every batch of 1,3,5-triaryl-2-pyrazoline meets the highest international standards for identity, assay, and impurity profiles. By leveraging our expertise in process optimization and solid-state chemistry, we deliver intermediates that empower our clients to accelerate their drug development pipelines with confidence and reliability.
We invite you to engage with our technical procurement team to discuss how our advanced manufacturing solutions can support your specific project requirements and cost targets. Whether you need a Customized Cost-Saving Analysis for your current supply chain or require detailed technical documentation to validate our processes, we are ready to provide comprehensive support. Please contact us today to request specific COA data and route feasibility assessments, and let us demonstrate how our commitment to innovation and quality can become a cornerstone of your supply chain strategy.
