Advanced Manufacturing of High-Purity 3-Alkenylcephem Intermediates for Antibiotic Synthesis
Advanced Manufacturing of High-Purity 3-Alkenylcephem Intermediates for Antibiotic Synthesis
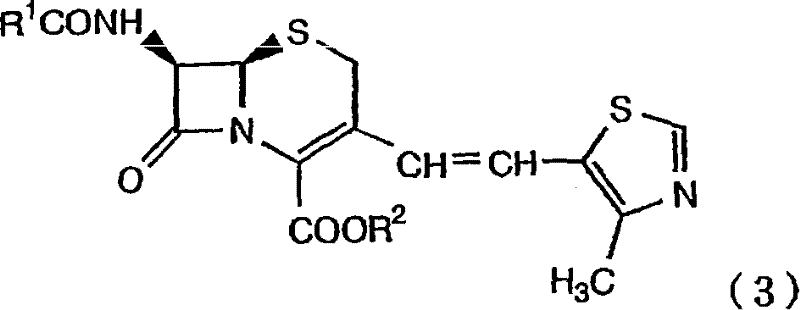
In the competitive landscape of pharmaceutical intermediate manufacturing, the ability to produce high-purity cephalosporin precursors with precise stereochemical control is paramount. The patent CN1964981B introduces a groundbreaking process for the production of 7-amino-3-[(E/Z)-2-(4-methylthiazol-5-yl)vinyl]-3-cephem-4-carboxylic acid and its alkali metal salts. This technology specifically addresses the critical challenge of maximizing the content of the biologically active Z-isomer, which is essential for the efficacy of oral cephem preparations like Cefditoren Pivoxil. By leveraging a novel purification strategy involving highly porous polymers and activated carbon, this method offers a robust alternative to traditional chromatographic separation, promising significant improvements in yield and operational efficiency for global supply chains.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the separation of geometric isomers in cephem antibiotics has relied heavily on ion exchange resin chromatography or activated carbon adsorption columns. While effective to a degree, these conventional methods present substantial logistical and economic hurdles for large-scale manufacturing. The process typically involves converting the Z/E mixture into amine or hydrochloride salts, followed by passage through packed columns. This approach suffers from significant drawbacks, including the heavy burden of eluate recovery and the complex regeneration of column packing agents. Furthermore, conventional methods often struggle to reduce the E-isomer content below critical thresholds, with some prior art reporting residual E-body content as high as 14% or 2%, which fails to meet the stringent purity requirements necessary for modern high-efficacy antibacterial agents.
The Novel Approach
The innovative methodology disclosed in the patent fundamentally shifts the purification paradigm by eliminating the need for complex column chromatography. Instead, it employs a direct treatment strategy where a highly porous polymer and/or activated carbon is added directly to the aqueous solution of the alkali metal salt of the cephem compound. This simple addition allows for the selective adsorption of the unwanted E-isomer impurities within a short processing time, typically ranging from several minutes to two hours. The result is a dramatic enhancement in the Z-isomer content, often achieving levels greater than 99% with E-isomer residues reduced to less than 0.1%. This streamlined approach not only simplifies the equipment requirements but also drastically reduces the solvent usage and waste generation associated with traditional column operations.
Mechanistic Insights into Adsorptive Purification and Enzymatic Deacylation
The core of this manufacturing process lies in a sophisticated two-stage reaction sequence followed by a targeted adsorption step. Initially, the 7-substituted acylamino-3-[(E/Z)-2-(4-methylthiazol-5-yl)vinyl]-3-cephem-4-carboxylic acid compound undergoes a deprotection reaction at the 4-position carboxylic acid group. This is achieved using alkali metal hydroxides, bicarbonates, or carbonates in an aqueous environment, often facilitated by phenolic solvents which act as both reagents and solvents to ensure stability. Following this, the 7-position substituted acyl group is removed via an enzymatic reaction using Penicillin G Acylase (PGA). This enzymatic step is crucial as it proceeds under mild aqueous conditions, preserving the delicate beta-lactam ring structure while quantitatively yielding the desired 7-amino intermediate as an alkali metal salt.
Once the enzymatic conversion is complete, the reaction mixture contains the target Z-isomer along with trace amounts of the E-isomer. The patent reveals that treating this specific aqueous alkaline solution with highly porous polymers, such as styrene-divinylbenzene copolymers with specific surface areas exceeding 400 m²/g, creates a selective environment. The porous structure and surface chemistry of these polymers preferentially interact with the E-isomer, effectively sequestering it from the solution. This mechanism allows the desired Z-isomer to remain in the supernatant with exceptional purity. Subsequent acidification of the purified solution to a pH range of 3.0 to 4.3 induces the crystallization of the high-purity Z-isomer compound, which can then be easily isolated via filtration or centrifugation.
How to Synthesize 7-Amino-3-alkenylcephem Compounds Efficiently
The synthesis of these critical antibiotic intermediates requires precise control over reaction conditions to maintain stereochemical integrity. The process begins with the preparation of the alkali metal salt solution through deprotection and enzymatic cleavage, ensuring that the reaction environment remains within optimal temperature and pH ranges to prevent degradation. Once the crude aqueous solution containing the E/Z mixture is obtained, the purification phase commences immediately. The detailed standardized synthesis steps, including specific reagent ratios, temperature controls, and filtration protocols required to replicate this high-yield process, are outlined in the guide below.
- Perform deprotection of the 4-position carboxylic acid protecting group using phenols or acidic conditions to form the alkali metal salt.
- Conduct an enzymatic reaction using Penicillin G Acylase (PGA) in an aqueous system to remove the 7-position acyl group.
- Treat the resulting aqueous solution with highly porous polymer or activated carbon to selectively adsorb E-isomers, followed by acidification to precipitate the high-purity Z-isomer.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this purification technology translates into tangible operational efficiencies and risk mitigation. By replacing complex chromatographic columns with a simple batch treatment using porous polymers or activated carbon, manufacturers can significantly reduce the capital expenditure associated with specialized separation equipment. The elimination of column packing and regeneration cycles means that production lines can operate with higher throughput and less downtime, directly addressing the industry-wide challenge of reducing lead time for high-purity pharmaceutical intermediates. Furthermore, the simplicity of the filtration-based separation enhances the overall reliability of the supply chain, minimizing the risk of batch failures due to column channeling or resin degradation.
- Cost Reduction in Manufacturing: The shift away from ion exchange chromatography eliminates the recurring costs associated with purchasing, regenerating, and disposing of expensive resin materials. Additionally, the reduction in solvent volumes required for elution and column washing leads to substantial savings in raw material costs and waste disposal fees. The process operates efficiently in aqueous systems, reducing the reliance on costly organic solvents and lowering the overall environmental compliance burden, which collectively drives down the cost of goods sold for these complex intermediates.
- Enhanced Supply Chain Reliability: The robustness of the adsorptive purification method ensures consistent product quality across different production batches, a critical factor for maintaining long-term contracts with pharmaceutical clients. Since the process does not rely on the variable performance of packed columns, which can degrade over time, the manufacturing timeline becomes more predictable. This stability allows suppliers to offer more reliable delivery schedules, ensuring that downstream API manufacturers receive their critical raw materials without unexpected delays caused by purification bottlenecks.
- Scalability and Environmental Compliance: Scaling this process from pilot plant to commercial production is straightforward because it utilizes standard mixing and filtration equipment rather than specialized chromatography skids. The use of water as the primary solvent medium aligns with green chemistry principles, significantly reducing the emission of volatile organic compounds (VOCs). This environmental advantage simplifies regulatory approvals and permits, facilitating faster market entry for new generic antibiotics that rely on these high-purity cephem intermediates.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this advanced purification technology. These insights are derived directly from the patent specifications and are designed to clarify the operational benefits for potential partners and technical stakeholders evaluating this manufacturing route for their supply chains.
Q: What is the primary advantage of using highly porous polymers over ion exchange resins?
A: Highly porous polymers allow for direct treatment in aqueous solutions without the need for complex column packing or regeneration cycles, significantly simplifying the workflow and reducing operational burdens compared to traditional chromatography.
Q: How does this process ensure high Z-isomer purity?
A: The process utilizes the selective adsorption properties of specific porous polymers or activated carbon which preferentially bind the E-isomer impurities, allowing the desired Z-isomer to remain in solution with purity levels exceeding 99%.
Q: Is this method suitable for large-scale industrial production?
A: Yes, the method avoids expensive chromatography columns and uses standard filtration techniques for solid removal, making it highly scalable and economically advantageous for commercial manufacturing of antibiotic intermediates.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 7-Amino-3-alkenylcephem Supplier
At NINGBO INNO PHARMCHEM, we recognize the strategic importance of high-purity intermediates in the global antibiotic market. Our technical team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the sophisticated purification techniques described in patent CN1964981B can be seamlessly integrated into our manufacturing operations. We are committed to delivering products that meet stringent purity specifications, utilizing our rigorous QC labs to verify Z-isomer content and minimize E-isomer impurities to levels that exceed industry standards, thereby supporting the efficacy and safety of the final pharmaceutical products.
We invite pharmaceutical companies and contract manufacturers to collaborate with us to leverage this cost-effective and scalable technology. By partnering with our technical procurement team, you can request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We encourage you to contact us today to obtain specific COA data and route feasibility assessments, ensuring that your supply chain is optimized for both quality and economic efficiency in the production of next-generation cephalosporin antibiotics.
