Advanced Catalytic Liquid Phase Oxidation for Commercial Phthalide Production
The global demand for high-purity phthalide, a critical intermediate in the synthesis of pharmaceuticals such as the anxiolytic drug doxepin and various agrochemical fungicides, necessitates robust and efficient manufacturing technologies. Patent CN112778252A introduces a groundbreaking method for preparing phthalide through catalytic liquid phase selective oxidation, marking a significant departure from traditional, hazardous synthetic routes. This innovation utilizes a sophisticated nitrogen-doped carbon-supported bimetallic catalyst to convert o-xylene directly into phthalide using air or oxygen as the oxidant. By shifting the paradigm from corrosive acid hydrolysis or high-temperature gas phase oxidation to a controlled liquid phase system, this technology offers a compelling value proposition for manufacturers seeking to optimize yield while adhering to stricter environmental and safety standards. The technical breakthrough lies not only in the catalyst composition but in the precise orchestration of reaction conditions that maximize selectivity towards the desired lactone structure.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial production of phthalide has been plagued by significant technical and economic hurdles associated with legacy synthetic pathways. One prevalent method involves the hydrolysis of o-cyanobenzyl chloride using concentrated sulfuric acid, a process that generates severe equipment corrosion due to the aggressive acidic environment and produces vast quantities of acidic wastewater that require complex and costly treatment protocols. Another common route involves the reduction of phthalic anhydride using zinc and hydrochloric acid, which incurs high raw material costs and generates stoichiometric amounts of metal waste. Furthermore, while catalytic hydrogenation of phthalic anhydride is an alternative, it suffers from poor selectivity control, often leading to over-reduction byproducts. Perhaps most critically, the dominant gas phase oxidation of o-xylene to phthalic anhydride followed by conversion requires extreme temperatures around 350°C, creating substantial safety risks and promoting over-oxidation reactions that degrade raw material efficiency and lower overall product selectivity.
The Novel Approach
In stark contrast to these cumbersome traditional methods, the novel approach detailed in the patent leverages a heterogeneous catalytic system that operates under remarkably mild conditions. By employing a nitrogen-doped carbon-supported bimetallic catalyst, the process facilitates the direct selective oxidation of o-xylene in the liquid phase. This method effectively bypasses the need for corrosive mineral acids or expensive stoichiometric reducing agents, replacing them with molecular oxygen or air, which are abundant and cost-effective oxidants. The core innovation is the ability to selectively oxidize the two methyl groups of the o-xylene molecule into a hydroxymethyl group and a carboxyl group respectively, which then spontaneously undergo intramolecular esterification to form the phthalide ring. This streamlined pathway not only simplifies the post-treatment process by eliminating salt waste but also significantly enhances the safety profile of the operation by lowering the reaction temperature to a range of 80-180°C and pressures between 0.4-2.0 MPa.
Mechanistic Insights into N-Doped Carbon-Supported Bimetallic Catalysis
The efficacy of this synthesis relies heavily on the unique electronic and structural properties of the nitrogen-doped carbon-supported bimetallic catalyst. The catalyst is typically composed of transition metals such as cobalt and manganese dispersed on a carbon matrix that has been doped with nitrogen atoms derived from multidentate nitrogen-containing compounds like benzotriazole. The nitrogen doping plays a tripartite role in enhancing catalytic performance: firstly, the nitrogen atoms chelate the metal centers, anchoring them firmly to the carbon support to prevent leaching during the reaction; secondly, due to the higher electronegativity of nitrogen compared to carbon, the adjacent carbon atoms acquire a partial positive charge, which facilitates the adsorption and activation of oxygen molecules; and thirdly, the inherent alkalinity of the nitrogen species promotes the cyclization step. This synergistic effect ensures that the oxidation proceeds selectively to the intermediate o-hydroxymethylbenzoic acid without over-oxidizing to phthalic acid or other degradation products.
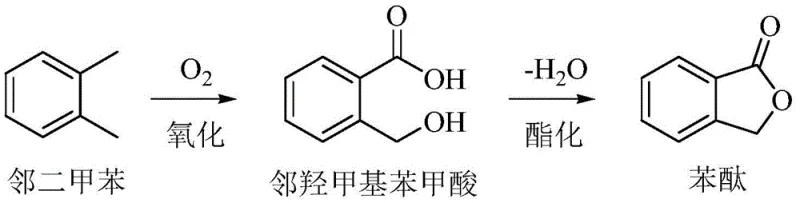
Following the initial oxidation, the reaction mechanism proceeds through a crucial intramolecular esterification step. As illustrated in the reaction pathway, the o-xylene is first oxidized to form o-hydroxymethylbenzoic acid, an unstable intermediate that contains both hydroxyl and carboxylic acid functionalities in ortho positions. Under the reaction conditions facilitated by the basic sites on the nitrogen-doped catalyst, this intermediate undergoes dehydration to close the lactone ring, yielding phthalide. The choice of bimetallic components, such as the Co-Mn combination, is critical because metals from different groups in the periodic table exhibit distinct catalytic oxidation capabilities. This difference allows for the differentiated oxidation of the two methyl groups, a feat that single-metal catalysts or metals from the same group (like Fe-Co) struggle to achieve, often resulting in lower selectivity. The optimization of metal ratios and the specific nature of the nitrogen precursor are therefore paramount in tuning the catalyst for maximum efficiency.
How to Synthesize Phthalide Efficiently
The synthesis of phthalide via this advanced oxidative protocol requires precise control over catalyst preparation and reaction parameters to ensure reproducibility and high yield. The process begins with the meticulous preparation of the heterogeneous catalyst, involving the impregnation of metal salts onto a nitrogen-rich organic framework followed by high-temperature pyrolysis. Once the catalyst is activated, it is introduced into a pressurized reactor containing the o-xylene substrate. The system is then heated and pressurized with oxygen to initiate the selective oxidation. While the general procedure is straightforward, the specific nuances of temperature ramping, pressure maintenance, and catalyst loading are critical for preventing side reactions. For a comprehensive understanding of the operational parameters and safety protocols required for implementation, please refer to the standardized synthesis steps outlined below.
- Preparation of the Nitrogen-Doped Carbon-Supported Bimetallic Catalyst by reacting metal salts (e.g., Cobalt and Manganese) with a multidentate nitrogen-containing compound followed by roasting at 600-800°C.
- Loading the reactor with o-xylene and the prepared catalyst, then heating the mixture to a reaction temperature between 80°C and 180°C.
- Introducing oxygen or air to maintain a pressure of 0.4-2.0 MPa for 2-10 hours to achieve selective oxidation and subsequent intramolecular esterification.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this catalytic liquid phase oxidation technology translates into tangible strategic benefits regarding cost structure and operational reliability. The shift away from corrosive reagents like sulfuric acid and stoichiometric reducing agents like zinc powder fundamentally alters the cost equation by eliminating the expenses associated with handling hazardous materials and treating toxic waste streams. Furthermore, the use of air or oxygen as the primary oxidant represents a drastic reduction in raw material costs compared to specialized chemical oxidants. The mild reaction conditions also imply that existing stainless steel reactors can often be utilized without the need for exotic alloy upgrades required for high-temperature gas phase processes, thereby reducing capital expenditure barriers for scale-up. These factors collectively contribute to a more resilient and cost-efficient supply chain for phthalide derivatives.
- Cost Reduction in Manufacturing: The elimination of expensive and corrosive reagents such as concentrated sulfuric acid and zinc powder leads to substantial savings in raw material procurement and waste disposal costs. By utilizing a heterogeneous catalyst that can be recovered and reused multiple times without significant loss of activity, the process minimizes the recurring cost of catalyst replenishment. Additionally, the lower energy requirements associated with operating at 150°C rather than 350°C result in significantly reduced utility costs for heating and cooling, further enhancing the overall economic viability of the manufacturing process.
- Enhanced Supply Chain Reliability: The reliance on commodity chemicals like o-xylene and air/oxygen ensures a stable and secure supply of raw materials, mitigating the risks associated with sourcing specialized or regulated reagents. The robustness of the nitrogen-doped carbon catalyst, which demonstrates stability over multiple cycles, ensures consistent production throughput and reduces the frequency of unplanned shutdowns for catalyst changeovers. This reliability is crucial for maintaining continuous supply to downstream pharmaceutical and agrochemical customers who demand strict adherence to delivery schedules.
- Scalability and Environmental Compliance: The liquid phase nature of the reaction facilitates easier heat and mass transfer management compared to gas phase fixed-bed reactors, making the scale-up from pilot to commercial production more predictable and controllable. The process generates minimal acidic wastewater and no heavy metal salt waste, aligning perfectly with increasingly stringent global environmental regulations. This compliance advantage reduces the regulatory burden and potential liability for manufacturers, ensuring long-term operational continuity in regions with strict environmental oversight.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this phthalide synthesis technology. These answers are derived directly from the experimental data and technical disclosures found in the patent literature, providing a factual basis for decision-making. Understanding these details is essential for R&D teams evaluating the feasibility of technology transfer and for procurement teams assessing the long-term value of the supply partnership.
Q: What are the primary advantages of the liquid phase oxidation method over traditional gas phase oxidation for phthalide?
A: The liquid phase method described in patent CN112778252A operates at significantly milder temperatures (80-180°C) compared to the harsh conditions (~350°C) required for gas phase oxidation. This reduces energy consumption, minimizes over-oxidation risks, and greatly improves process safety while maintaining high product selectivity.
Q: Why is the nitrogen-doped carbon support critical for the catalyst's performance?
A: The nitrogen doping serves multiple functions: it chelates the metal components to prevent leaching, increases the positive charge density on adjacent carbon atoms to favor oxygen adsorption, and provides alkalinity that promotes the intramolecular esterification of the intermediate o-hydroxymethylbenzoic acid into phthalide.
Q: Can the bimetallic catalyst be recycled for industrial applications?
A: Yes, the patent data demonstrates that the nitrogen-doped carbon-supported bimetallic catalyst exhibits excellent stability and can be recovered and reused. Experimental examples show that after recycling, the catalyst maintains high conversion rates of o-xylene and phthalide selectivity, ensuring long-term economic viability.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Phthalide Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of advanced catalytic oxidation technologies in modernizing the production of key fine chemical intermediates like phthalide. As a premier CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that innovative laboratory methods are successfully translated into robust industrial processes. Our commitment to quality is underpinned by stringent purity specifications and rigorous QC labs that verify every batch meets the exacting standards required by the global pharmaceutical and agrochemical industries. We are uniquely positioned to leverage this patented technology to deliver high-purity phthalide with superior consistency and reliability.
We invite forward-thinking partners to collaborate with us to unlock the full commercial potential of this efficient synthesis route. By engaging with our technical procurement team, you can request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We encourage you to reach out today to obtain specific COA data and route feasibility assessments that demonstrate how our optimized manufacturing capabilities can enhance your supply chain efficiency and reduce your total cost of ownership.
