Scalable Synthesis of 2-Amino-6-Bromopyridine via Novel 1,3-Dichloropropanol Route for Commercial API Production
Scalable Synthesis of 2-Amino-6-Bromopyridine via Novel 1,3-Dichloropropanol Route for Commercial API Production
The landscape of pharmaceutical intermediate manufacturing is constantly evolving, driven by the need for safer, more efficient, and cost-effective synthetic pathways. A pivotal advancement in this domain is detailed in patent CN115784978B, which discloses a robust method for synthesizing 2-amino-6-bromopyridine. This compound serves as a critical building block for a wide array of bioactive molecules, including natural products, therapeutic drugs, and luminescent materials. The patented technology shifts the paradigm from traditional, hazardous multi-step sequences to a streamlined two-step process originating from 1,3-dichloropropanol. By leveraging nucleophilic substitution followed by a controlled cyclization, this innovation addresses long-standing pain points in yield optimization and operational safety, positioning it as a superior choice for a reliable pharmaceutical intermediate supplier aiming to secure the global supply chain.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of 2-amino-6-bromopyridine has been plagued by significant technical hurdles that impede efficient commercial production. One of the most common legacy routes involves the use of epichlorohydrin as a starting material. While conceptually straightforward, the three-membered epoxy ring is highly reactive and prone to uncontrolled cracking under acidic or basic catalysis. This inherent instability leads to a plethora of side reactions, drastically lowering the conversion rate and overall yield. Moreover, the cyclization step in this traditional method typically relies on a glacial acetic acid solution of hydrogen bromide. This reagent combination is notoriously difficult to handle on an industrial scale; it generates dense, rolling smoke upon exposure to air, posing severe safety risks and environmental compliance challenges. The low acidity of acetic acid further necessitates a total dependence on hydrogen bromide for proton supply, resulting in sluggish reaction kinetics and inefficient resource utilization.
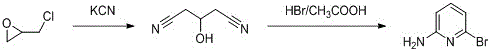
Alternative pathways, such as the ammonification of 2,6-dibromopyridine, present their own set of economic and technical barriers. Although this route appears shorter in terms of step count, the starting material, 2,6-dibromopyridine, is prohibitively expensive, driving up the cost of goods sold (COGS) significantly. Furthermore, the reaction conditions are severe, and the selectivity is poor, often leading to the formation of unwanted byproducts like 2,6-diaminopyridine. Other methods involving the hydrolysis of 6-bromo-2-pyridinecarboxylic acid or the oxidation of 2-amino-6-methylpyridine involve excessive reaction steps. These elongated synthetic sequences not only accumulate yield losses at each stage but also complicate production operations, making them less attractive for cost reduction in API manufacturing.
The Novel Approach
In stark contrast to these cumbersome legacy methods, the technology disclosed in CN115784978B introduces a highly efficient strategy utilizing 1,3-dichloropropanol, a readily available and cost-effective commodity chemical. This novel approach condenses the synthesis into just two main reaction stages, dramatically simplifying the workflow. The first stage involves a cyanation reaction where 1,3-dichloropropanol reacts with sodium cyanide in the presence of potassium iodide to form 3-hydroxyglutaronitrile. This step is characterized by mild heating and high conversion, avoiding the violent ring-opening issues of epichlorohydrin. The second stage employs a clever cyclization strategy using hydrogen bromide gas and trifluoroacetic acid (TFA). Unlike the problematic glacial acetic acid mixtures of the past, the use of TFA enhances the reaction efficiency and control. This method not only improves the overall yield but also significantly enhances operational safety, making it an ideal candidate for the commercial scale-up of complex heterocycles.
Mechanistic Insights into Cyanation and Acid-Catalyzed Cyclization
The success of this synthetic route lies in the precise control of nucleophilic substitution and subsequent cyclization mechanisms. In the first step, potassium iodide acts as a crucial catalyst, facilitating the displacement of chloride ions by cyanide groups. The reaction proceeds in ethanol at a moderate temperature of 70°C, ensuring that the 1,3-dichloropropanol is converted into 3-hydroxyglutaronitrile with high fidelity. The presence of the hydroxyl group in the intermediate is pivotal, as it remains intact during this stage, setting the stage for the subsequent ring closure. The use of ethanol as a solvent provides an optimal medium for the solubility of both the organic substrate and the inorganic cyanide salt, promoting homogeneous reaction conditions that minimize side product formation.
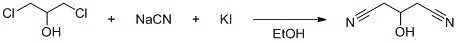
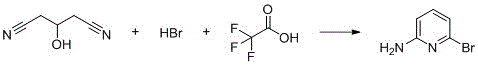
The second mechanistic phase involves the transformation of the linear nitrile intermediate into the aromatic pyridine ring. This is achieved through the introduction of hydrogen bromide gas in the presence of trifluoroacetic acid at room temperature. The strong acidity of TFA, combined with the nucleophilic attack of the nitrogen species generated in situ, drives the cyclization forward. The bromine atom is introduced simultaneously or subsequently via the HBr, securing the 6-position substitution. This one-pot cyclization and bromination strategy is highly elegant, as it avoids the need for separate halogenation steps that often require hazardous reagents like elemental bromine. The reaction mixture is then carefully neutralized to a pH of 9.5-10.5, precipitating the target high-purity 2-amino-6-bromopyridine while leaving impurities in the aqueous phase.
How to Synthesize 2-Amino-6-Bromopyridine Efficiently
Implementing this patented process requires adherence to specific operational parameters to maximize yield and purity. The procedure begins with the preparation of the nitrile intermediate, followed by a workup to isolate the oily substance, which is then subjected to the cyclization conditions. The simplicity of the workup—involving standard extraction and drying techniques—makes this route particularly attractive for plant-level execution. For R&D teams looking to replicate or license this technology, the following guide outlines the critical operational flow derived directly from the patent examples.
- React 1,3-dichloropropanol with sodium cyanide and potassium iodide in ethanol at 70°C to form 3-hydroxyglutaronitrile.
- Concentrate the reaction mixture, extract with ethyl acetate, and dry to obtain the oily intermediate.
- React the intermediate with hydrogen bromide gas and trifluoroacetic acid at room temperature, then neutralize and crystallize to obtain 2-amino-6-bromopyridine.
Commercial Advantages for Procurement and Supply Chain Teams
From a procurement and supply chain perspective, the adoption of this novel synthesis method offers transformative benefits that directly impact the bottom line and operational resilience. Traditional methods often rely on specialized, high-cost starting materials or generate significant hazardous waste, both of which strain the supply chain. By shifting to a route based on 1,3-dichloropropanol, manufacturers can leverage a stable, commodity-grade feedstock that is less susceptible to market volatility. This transition fundamentally alters the cost structure of the final intermediate, enabling significant savings without compromising on quality. Furthermore, the reduction in reaction steps from four or five down to just two drastically reduces the processing time and equipment occupancy, enhancing overall plant throughput.
- Cost Reduction in Manufacturing: The elimination of expensive starting materials like 2,6-dibromopyridine results in a substantial decrease in raw material costs. Additionally, the removal of transition metal catalysts or complex purification steps associated with older routes means that downstream processing is simplified. The high yield per step (consistently above 85% in examples) ensures that less raw material is wasted, directly translating to a lower cost per kilogram of the final API intermediate. This efficiency allows for more competitive pricing strategies in the global market.
- Enhanced Supply Chain Reliability: Relying on commodity chemicals like 1,3-dichloropropanol and sodium cyanide ensures a robust supply chain that is not dependent on niche suppliers. The mild reaction conditions (70°C and room temperature) reduce the risk of batch failures due to thermal runaway or equipment stress, ensuring consistent delivery schedules. This reliability is crucial for reducing lead time for high-purity intermediates, allowing downstream drug manufacturers to maintain their own production schedules without interruption.
- Scalability and Environmental Compliance: The process avoids the use of glacial acetic acid/HBr mixtures that generate dense, corrosive smoke, thereby simplifying废气 (waste gas) treatment requirements. The use of standard solvents like ethanol and ethyl acetate facilitates easier solvent recovery and recycling. This alignment with green chemistry principles not only reduces environmental compliance costs but also future-proofs the manufacturing process against tightening regulatory standards, ensuring long-term viability.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the synthesis of 2-amino-6-bromopyridine via this patented route. These insights are derived from the specific experimental data and beneficial effects outlined in the patent documentation, providing clarity for technical decision-makers evaluating this technology for integration into their supply chains.
Q: What are the primary advantages of the 1,3-dichloropropanol route over the epichlorohydrin method?
A: The 1,3-dichloropropanol route avoids the severe side reactions and low conversion rates associated with the three-membered ring opening of epichlorohydrin. Furthermore, it eliminates the need for hazardous glacial acetic acid/HBr mixtures that produce dense smoke and are difficult to scale, resulting in a safer and more operable process.
Q: How does the new method improve yield compared to traditional synthesis?
A: Traditional methods often suffer from multi-step losses or poor selectivity (e.g., forming 2,6-diaminopyridine impurities). The patented two-step process achieves high conversion rates with yields exceeding 85% per step, significantly boosting overall throughput and reducing waste.
Q: Is this synthesis method suitable for large-scale commercial production?
A: Yes, the process utilizes mild reaction conditions (70°C for step 1, room temperature for step 2) and common solvents like ethanol and ethyl acetate. The avoidance of extreme conditions and the use of stable intermediates make it highly amenable to commercial scale-up in standard reactor setups.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 2-Amino-6-Bromopyridine Supplier
The technological breakthroughs presented in CN115784978B underscore the immense potential for optimizing the production of key pharmaceutical intermediates. At NINGBO INNO PHARMCHEM, we specialize in translating such innovative laboratory protocols into robust, industrial-scale realities. As a premier CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production. Our facilities are equipped with rigorous QC labs capable of meeting stringent purity specifications required by top-tier global pharmaceutical companies, ensuring that every batch of 2-amino-6-bromopyridine delivered meets the highest standards of quality and consistency.
We invite procurement leaders and R&D directors to collaborate with us to leverage this advanced synthesis route for their projects. By partnering with our technical procurement team, you can access a Customized Cost-Saving Analysis tailored to your specific volume requirements. We encourage you to reach out today to request specific COA data and comprehensive route feasibility assessments, ensuring that your supply chain is built on the foundation of the most efficient and reliable chemistry available.
