Advanced Synthesis of Chiral Bisoxazoline Nickel Complexes for Asymmetric Catalysis Applications
Introduction to Next-Generation Chiral Catalysts
The landscape of asymmetric synthesis is constantly evolving, driven by the pharmaceutical industry's relentless demand for high-purity chiral intermediates. A pivotal advancement in this field is documented in patent CN103613559A, which discloses a robust and efficient synthesis method for a novel chiral bisoxazoline nickel complex. This organometallic species represents a significant leap forward in ligand design, offering a stable, well-defined coordination environment that is crucial for inducing high enantioselectivity in downstream catalytic transformations. Unlike traditional methods that often suffer from low stability or difficult purification, this patented approach utilizes a direct condensation strategy between salicylonitrile, D-phenylglycinol, and nickel salts. For R&D directors and process chemists, understanding the nuances of this synthesis is key to unlocking new pathways for creating complex chiral drugs and agrochemicals with superior optical purity.
The strategic value of this technology extends beyond mere academic interest; it addresses critical bottlenecks in the supply chain of fine chemical intermediates. By employing readily available starting materials such as o-hydroxybenzonitrile and chiral amino alcohols, the process mitigates the reliance on exotic or prohibitively expensive precursors. Furthermore, the resulting nickel complex exhibits remarkable thermal stability and structural integrity, as evidenced by detailed crystallographic data. This report delves deep into the mechanistic underpinnings and commercial implications of this synthesis, providing a comprehensive roadmap for procurement managers and supply chain heads looking to optimize their sourcing strategies for high-performance catalytic systems.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the preparation of chiral transition metal complexes for asymmetric catalysis has been fraught with inefficiencies that hinder commercial viability. Traditional routes often involve a multi-step sequence where the chiral ligand is first synthesized in isolation, purified to remove diastereomeric impurities, and subsequently metallated with the transition metal salt. This stepwise approach not only increases the overall processing time but also introduces multiple points of potential yield loss and contamination. In many legacy processes, the metallation step itself can be sluggish, requiring harsh conditions or inert atmospheres that drive up operational expenditures. Additionally, the purification of the final metal-ligand complex often necessitates column chromatography or repeated recrystallizations, which are difficult to scale and generate substantial solvent waste. These factors collectively contribute to a high cost of goods sold (COGS) and extended lead times, making it challenging for manufacturers to respond agilely to market demands for chiral active pharmaceutical ingredients (APIs).
The Novel Approach
In stark contrast, the methodology outlined in patent CN103613559A presents a streamlined, one-pot synthetic strategy that dramatically simplifies the production workflow. By combining the nitrile precursor, the chiral amino alcohol, and the nickel source directly in a chlorobenzene solvent system, the reaction facilitates both the formation of the oxazoline rings and the coordination of the nickel center in a single thermal operation. This telescoped process eliminates the need for isolating intermediate ligands, thereby reducing unit operations and minimizing material handling risks. The use of nickel chloride hexahydrate or nickel acetate tetrahydrate as the metal source is particularly advantageous, as these salts are inexpensive and commercially abundant compared to precious metals like palladium or rhodium. 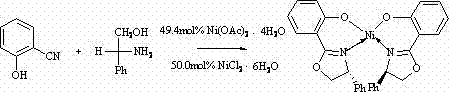 The reaction proceeds under reflux conditions for 72 hours, yielding the target complex with impressive efficiency. Post-reaction workup is equally straightforward, involving simple solvent removal, aqueous washing, and extraction, followed by a final crystallization step that delivers the product in high purity. This operational simplicity translates directly into enhanced process robustness and lower manufacturing costs.
The reaction proceeds under reflux conditions for 72 hours, yielding the target complex with impressive efficiency. Post-reaction workup is equally straightforward, involving simple solvent removal, aqueous washing, and extraction, followed by a final crystallization step that delivers the product in high purity. This operational simplicity translates directly into enhanced process robustness and lower manufacturing costs.
Mechanistic Insights into Ni-Catalyzed Oxazoline Formation
To fully appreciate the utility of this complex for R&D applications, one must examine the intricate coordination chemistry that defines its structure and reactivity. The synthesis relies on the templating effect of the nickel ion, which likely coordinates with the nitrogen and oxygen donors of the reacting species to guide the cyclization of the oxazoline rings. The resulting architecture is a rigid, chiral pocket that effectively differentiates between enantiotopic faces of substrates in catalytic reactions. Crystallographic analysis reveals a distorted square planar or octahedral geometry around the nickel center, depending on the specific solvation state, with bond lengths that indicate strong covalent character. Specifically, the Ni-O bond distances are observed to be approximately 1.83 Å, while the Ni-N bonds are slightly longer at roughly 1.89 Å. These precise metric parameters are critical for maintaining the steric bulk necessary for high enantioselectivity, ensuring that the chiral information from the phenylglycinol moiety is effectively transmitted to the catalytic site.
Furthermore, the stability of the complex is underpinned by the chelate effect, where the bidentate nature of the bisoxazoline ligands forms thermodynamically stable five-membered rings with the metal center. The X-ray diffraction data confirms the absolute configuration, showing a specific spatial arrangement that prevents racemization under standard reaction conditions. This structural fidelity is paramount for pharmaceutical applications where even trace amounts of the wrong enantiomer can be detrimental. The presence of the phenolic oxygen atoms in the coordination sphere also adds an electronic dimension, potentially modulating the Lewis acidity of the nickel center. For process chemists, this means the catalyst can be tuned for specific transformations, such as aldol reactions or cycloadditions, by adjusting the electronic properties of the substituents on the oxazoline rings without compromising the core structural integrity.
How to Synthesize Chiral Bisoxazoline Nickel Complex Efficiently
Implementing this synthesis in a laboratory or pilot plant setting requires strict adherence to the optimized parameters defined in the patent to ensure reproducibility and high yield. The process is designed to be operationally simple, avoiding the need for specialized high-pressure equipment or cryogenic conditions. The key to success lies in the precise stoichiometric balance of the reactants and the maintenance of a consistent reflux temperature over the extended reaction period. Detailed standard operating procedures (SOPs) regarding solvent quality and drying techniques are essential to prevent hydrolysis of the nitrile or premature degradation of the amino alcohol.
- Combine nickel chloride hexahydrate or nickel acetate tetrahdyrate with salicylonitrile and D-phenylglycinol in chlorobenzene solvent.
- Heat the mixture to reflux temperature and maintain reaction for 72 hours to ensure complete cyclization and complexation.
- Remove solvent, dissolve residue in water, extract with chloroform, and recrystallize from ethanol/trichloromethane to obtain pure single crystals.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, the adoption of this nickel-based synthesis route offers compelling advantages that resonate deeply with procurement managers and supply chain strategists focused on cost optimization and reliability. The shift from precious metal catalysts to nickel represents a fundamental change in the cost structure of chiral intermediate production. Nickel salts are orders of magnitude cheaper than palladium or platinum equivalents, and their abundance ensures a stable supply chain不受 geopolitical fluctuations affecting rare earth or precious metal markets. Moreover, the high yields reported (up to 95%) mean that less raw material is required per kilogram of finished product, directly lowering the variable cost of production. This efficiency gain is compounded by the simplified purification protocol, which reduces the consumption of high-grade solvents and silica gel, further driving down operational expenses.
- Cost Reduction in Manufacturing: The elimination of expensive precious metals and the reduction in processing steps lead to a substantial decrease in the overall cost of goods. By avoiding multi-step ligand isolation, manufacturers save significantly on labor, energy, and equipment usage. The use of commodity chemicals like chlorobenzene and ethanol as solvents ensures that material costs remain low and predictable. Additionally, the high atom economy of the reaction minimizes waste disposal costs, contributing to a leaner and more profitable manufacturing model.
- Enhanced Supply Chain Reliability: Sourcing nickel salts and basic organic building blocks like salicylonitrile is far less risky than relying on specialized chiral ligands from limited suppliers. This diversification of the supply base enhances resilience against disruptions. The robustness of the synthesis method also means that production schedules are more predictable, with fewer batches rejected due to quality issues. This reliability allows supply chain heads to maintain lower safety stocks and improve cash flow, knowing that the production of this critical catalyst intermediate can be scaled up rapidly to meet surging demand.
- Scalability and Environmental Compliance: The process is inherently scalable, moving seamlessly from gram-scale laboratory synthesis to multi-kilogram pilot runs without significant re-engineering. The use of standard reflux conditions fits well within existing reactor infrastructure, minimizing capital expenditure requirements for new facilities. Furthermore, the simplified workup reduces the volume of hazardous waste generated, aiding in compliance with increasingly stringent environmental regulations. This alignment with green chemistry principles not only mitigates regulatory risk but also enhances the corporate sustainability profile of the manufacturing entity.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production and application of this chiral nickel complex. These insights are derived directly from the experimental data and claims presented in the patent documentation, ensuring accuracy and relevance for industry professionals. Understanding these details is crucial for making informed decisions about integrating this technology into your existing production workflows.
Q: What is the typical yield for this nickel complex synthesis?
A: According to patent CN103613559A, the process achieves exceptional yields of 92% when using nickel chloride and up to 95% with nickel acetate, significantly minimizing raw material waste.
Q: Is this complex suitable for large-scale pharmaceutical manufacturing?
A: Yes, the use of common solvents like chlorobenzene and standard reflux conditions makes the process highly scalable for industrial production of chiral intermediates.
Q: How is the stereochemistry of the complex confirmed?
A: The absolute configuration is rigorously verified through single-crystal X-ray diffraction analysis, confirming the specific coordination geometry required for high enantioselectivity.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Chiral Bisoxazoline Nickel Complex Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of advanced organometallic catalysts in modern drug discovery and development. Our team of expert chemists possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from benchtop to plant floor is seamless and efficient. We are committed to delivering products that meet stringent purity specifications, supported by our rigorous QC labs equipped with state-of-the-art analytical instrumentation. Whether you require custom synthesis of novel derivatives or bulk supply of established intermediates, our infrastructure is designed to support your most demanding projects with speed and precision.
We invite you to collaborate with us to leverage this cutting-edge synthesis technology for your next generation of chiral therapeutics. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We are ready to provide specific COA data and comprehensive route feasibility assessments to demonstrate how our solutions can drive value and innovation in your supply chain. Let us be your partner in turning complex chemical challenges into commercial successes.
