Scaling Chiral Oxazoline Nickel Complex Production for Advanced Asymmetric Catalysis Applications
The landscape of asymmetric catalysis is continually evolving, driven by the demand for highly efficient chiral ligands that can facilitate complex pharmaceutical transformations with precision. Patent CN103613559A introduces a robust and economically viable methodology for the synthesis of a specific chiral bisoxazoline nickel complex, a critical component in modern organometallic chemistry. This innovation addresses the persistent challenges in ligand design by offering a streamlined, one-step condensation and complexation protocol that achieves exceptional yields ranging from 92% to 95%. For research and development teams focused on optimizing catalytic cycles, this patent provides a foundational route to access high-purity nickel-based catalysts that are essential for enantioselective reactions. The technical significance lies not only in the structural integrity of the resulting complex but also in the operational simplicity that allows for seamless integration into existing manufacturing workflows.
Furthermore, the strategic selection of nickel as the central metal atom represents a pivotal shift away from the reliance on more expensive precious metals like palladium or platinum, which have traditionally dominated the field of asymmetric catalysis. By validating the efficacy of nickel complexes through rigorous crystallographic analysis, including detailed bond length and angle measurements, this technology opens new avenues for cost-effective process development. The ability to produce these complexes with such high fidelity and yield suggests a mature pathway that minimizes waste and maximizes resource efficiency. As the pharmaceutical industry seeks to reduce the cost of goods sold (COGS) for active pharmaceutical ingredients (APIs), the adoption of such nickel-based systems offers a compelling value proposition for both procurement specialists and process chemists looking to enhance their supply chain resilience.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditionally, the synthesis of chiral metal-organic complexes has often been plagued by multi-step procedures that require the prior isolation of the free ligand before metalation can occur. This conventional approach frequently involves harsh reaction conditions, sensitive intermediates that are prone to racemization, and the use of expensive transition metal precursors that drive up the overall production cost. Moreover, many existing protocols suffer from moderate yields and require extensive purification steps, such as column chromatography, which are difficult to scale and generate significant amounts of hazardous solvent waste. The reliance on precious metals also introduces supply chain vulnerabilities, as the availability and price volatility of metals like palladium can disrupt manufacturing schedules. Additionally, removing trace metal residues from the final product to meet stringent pharmaceutical purity standards often necessitates additional downstream processing, further complicating the workflow and increasing the lead time for high-purity pharmaceutical intermediates.
The Novel Approach
In stark contrast, the methodology disclosed in patent CN103613559A revolutionizes this process by combining ligand formation and metal coordination into a single, efficient reflux operation. By utilizing readily available starting materials such as salicylonitrile and D-phenylglycinol alongside inexpensive nickel salts, this novel approach eliminates the need for pre-synthesized ligands and drastically simplifies the reaction setup. The use of chlorobenzene as a solvent facilitates a high-temperature environment that drives the equilibrium towards the desired product, resulting in impressive isolated yields of up to 95%. This one-pot strategy not only reduces the number of unit operations but also minimizes solvent consumption and waste generation, aligning perfectly with green chemistry principles. The straightforward workup procedure, involving simple solvent removal and recrystallization, ensures that the final complex is obtained with high structural purity, ready for immediate application in catalytic processes without the need for complex purification techniques.
Mechanistic Insights into Ni-Catalyzed Bisoxazoline Formation
The formation of the chiral bisoxazoline nickel complex proceeds through a concerted mechanism where the nickel ion acts as both a template and a Lewis acid catalyst. Initially, the nickel salt, whether nickel chloride hexahydrate or nickel acetate tetrahydrate, dissociates in the chlorobenzene medium to provide coordinatively unsaturated nickel species. These species then interact with the nitrile group of salicylonitrile and the amino group of D-phenylglycinol. The phenolic hydroxyl group of the salicylonitrile also participates in the coordination sphere, facilitating the nucleophilic attack of the amine on the nitrile carbon. This cyclization event leads to the formation of the oxazoline ring, which immediately coordinates to the nickel center, stabilizing the transition state and preventing side reactions. The use of approximately 50 mol% of the nickel salt suggests a stoichiometric or near-stoichiometric involvement in the initial complexation, ensuring that the metal center is fully integrated into the ligand framework as it forms.
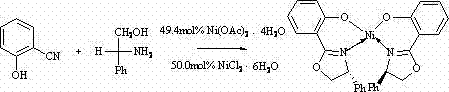
Crystallographic data provided in the patent offers profound insights into the geometric stability of the resulting complex. The analysis reveals a monoclinic crystal system with specific bond lengths, such as Ni-O bonds averaging around 1.83 Angstroms and Ni-N bonds around 1.88 Angstroms, indicating strong covalent character and stable coordination geometry. The bond angles, particularly the O-Ni-O and N-Ni-N angles approaching 180 degrees in certain configurations, suggest a distorted square planar or octahedral geometry depending on the solvation state, which is crucial for the complex's catalytic activity. This rigid structural framework is essential for inducing high enantioselectivity in subsequent catalytic applications, as it creates a well-defined chiral pocket around the metal center. Understanding these mechanistic and structural nuances allows R&D directors to predict the behavior of the catalyst in various asymmetric transformations, ensuring that the selected route delivers the required stereochemical outcomes for complex drug synthesis.
How to Synthesize Chiral Oxazoline Nickel Complex Efficiently
To replicate the high-yield synthesis described in the patent, precise control over reaction parameters and reagent quality is paramount. The process begins with the careful weighing of nickel salts and organic precursors to maintain the specific molar ratios that drive the reaction to completion. The choice between nickel chloride and nickel acetate offers flexibility, with both delivering excellent results, allowing manufacturers to select based on local availability and cost considerations. The reflux condition in chlorobenzene must be maintained consistently for the full 72-hour duration to ensure complete conversion, as the formation of the oxazoline ring is kinetically controlled and requires sustained thermal energy. Following the reaction, the workup procedure is designed to maximize recovery while ensuring purity, utilizing a biphasic extraction system that effectively separates the organic complex from inorganic byproducts. Detailed standardized operating procedures for each stage of this synthesis are outlined below to guide technical teams in achieving reproducible results.
- Combine salicylonitrile and D-phenylglycinol with 50 mol% nickel chloride hexahydrate or nickel acetate tetrahydrate in chlorobenzene solvent.
- Heat the mixture to reflux conditions and maintain the reaction for 72 hours to ensure complete complexation and cyclization.
- Remove the solvent, perform aqueous workup with chloroform extraction, and recrystallize from an ethanol-chloroform mixture to obtain pure single crystals.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, the adoption of this nickel-based synthesis route offers substantial strategic benefits for procurement managers and supply chain heads looking to optimize their operational expenditures. The primary advantage lies in the drastic reduction of raw material costs achieved by substituting expensive precious metal catalysts with abundant and affordable nickel salts. This shift not only lowers the direct material cost per kilogram of the produced intermediate but also mitigates the financial risks associated with the volatile pricing of noble metals. Furthermore, the high reaction yield of over 90% significantly improves the overall mass balance of the process, meaning less raw material is wasted and fewer batches are needed to meet production targets. This efficiency translates directly into improved margin profiles for the final pharmaceutical products that utilize these intermediates, providing a competitive edge in price-sensitive markets.
- Cost Reduction in Manufacturing: The elimination of multi-step ligand synthesis and the use of low-cost nickel precursors result in a significantly simplified cost structure. By avoiding the need for specialized precious metal recovery systems and reducing the number of purification steps, manufacturers can achieve substantial cost savings in utility consumption and labor. The high atom economy of the one-pot reaction ensures that the majority of the input mass is converted into valuable product, minimizing the disposal costs associated with chemical waste. Additionally, the robustness of the reaction conditions reduces the likelihood of batch failures, further protecting the bottom line from the financial impact of reprocessing or scrapped materials.
- Enhanced Supply Chain Reliability: The reliance on commodity chemicals such as salicylonitrile and phenylglycinol ensures a stable and diversified supply base, reducing the risk of shortages that often plague specialty reagents. Since the synthesis does not depend on a single source of exotic catalysts, procurement teams can easily qualify multiple suppliers for the starting materials, thereby enhancing supply continuity. The scalability of the reflux process means that production volumes can be ramped up quickly to meet surging demand without the need for significant capital investment in new reactor types. This flexibility allows supply chain managers to respond agilely to market fluctuations and maintain consistent inventory levels for their downstream customers.
- Scalability and Environmental Compliance: The process utilizes standard industrial solvents and equipment, making the transition from laboratory scale to commercial production seamless and predictable. The simplified workup involving extraction and crystallization is easily adaptable to large-scale separation units, ensuring that throughput can be increased without compromising product quality. Moreover, the reduced use of hazardous reagents and the potential for solvent recycling align with increasingly stringent environmental regulations, lowering the compliance burden on manufacturing sites. This environmentally friendly profile not only reduces the risk of regulatory penalties but also enhances the corporate sustainability image, which is becoming a critical factor in supplier selection for major multinational corporations.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the synthesis and application of this chiral nickel complex. These answers are derived directly from the experimental data and structural analysis provided in the patent documentation, ensuring accuracy and relevance for technical decision-makers. Understanding these specifics helps in evaluating the feasibility of integrating this technology into existing production lines and assessing its potential impact on product quality and cost efficiency.
Q: What are the key advantages of using nickel salts over palladium for this ligand synthesis?
A: Using nickel salts such as nickel chloride hexahydrate significantly reduces raw material costs compared to precious metals like palladium, while the patent demonstrates comparable high yields of 92-95%.
Q: How is the stereochemical purity ensured in this synthesis route?
A: The chirality is intrinsically derived from the use of enantiomerically pure D-phenylglycinol as the starting amine, which transfers its stereochemical information directly to the oxazoline ring during the cyclization process.
Q: Is this process suitable for large-scale manufacturing?
A: Yes, the process utilizes standard reflux conditions in chlorobenzene and simple extraction workups, making it highly adaptable for commercial scale-up without requiring specialized high-pressure equipment.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Chiral Oxazoline Nickel Complex Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-performance chiral ligands play in the development of next-generation pharmaceuticals and fine chemicals. Our team of expert chemists possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from benchtop discovery to industrial manufacturing is smooth and efficient. We are committed to delivering products that meet stringent purity specifications, supported by our rigorous QC labs that employ advanced analytical techniques to verify every batch. Whether you require custom synthesis of specific nickel complexes or large-scale supply of established intermediates, our infrastructure is designed to support your most demanding projects with reliability and precision.
We invite you to collaborate with us to explore how this innovative nickel-based technology can optimize your specific catalytic processes. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your current manufacturing setup, highlighting potential areas for efficiency gains. Please contact us today to request specific COA data and route feasibility assessments, and let us demonstrate how our expertise can drive value and innovation in your supply chain.
