Revolutionizing Alpha-Hydroxy Amide Production: A Catalyst-Free, Air-Oxidized Commercial Strategy
The pharmaceutical and fine chemical industries are constantly seeking robust, scalable, and environmentally benign synthetic routes for critical intermediates. Patent CN103755584A introduces a groundbreaking methodology for the synthesis of alpha-hydroxy amide compounds, a structural motif prevalent in numerous bioactive molecules and enzyme inhibitors. Unlike traditional approaches that rely on toxic reagents or precious metal catalysts, this invention leverages a simple yet highly effective system comprising potassium hydroxide, dimethyl sulfoxide (DMSO), and atmospheric air. For R&D directors and procurement specialists, this represents a paradigm shift towards sustainable manufacturing, offering a pathway to high-purity pharmaceutical intermediates without the burden of heavy metal contamination or complex multi-step sequences. The technical elegance of using air as a stoichiometric oxidant not only simplifies the reaction engineering but also drastically reduces the environmental footprint associated with waste disposal.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the construction of the alpha-hydroxy amide scaffold has been fraught with synthetic challenges that impede efficient commercial scale-up. One of the most prevalent traditional strategies involves the activation of alpha-hydroxy carboxylic acids into acid chlorides followed by amidation, a process depicted in standard literature. This conventional route necessitates the use of hazardous chlorinating agents and often requires tedious protection and deprotection steps for the hydroxyl group to prevent side reactions. Furthermore, alternative methods utilizing palladium-catalyzed oxidation of unsaturated amides introduce significant cost volatility due to the reliance on precious metals, alongside the rigorous requirement for metal scavenging to meet stringent pharmaceutical purity standards. These legacy processes are characterized by low atom economy, the generation of substantial chemical waste, and operational complexities that make them less attractive for modern, high-volume manufacturing environments.
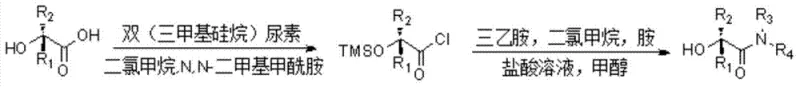
The Novel Approach
In stark contrast to these cumbersome legacy protocols, the method disclosed in CN103755584A offers a direct, one-step oxidative transformation that bypasses the need for pre-functionalized starting materials. By employing a basic system of potassium hydroxide or potassium tert-butoxide in DMSO under an air atmosphere, the reaction achieves direct hydroxylation at the alpha-position of the amide. This approach eliminates the necessity for toxic cyanide reagents or expensive transition metal catalysts, thereby streamlining the supply chain and reducing raw material costs. The operational simplicity is further enhanced by the mild reaction conditions, typically ranging from 50°C to 80°C, which allows for easy temperature control and minimizes energy consumption. This novel strategy not only improves the overall yield, reported to reach up to 98% in optimized examples, but also ensures a cleaner reaction profile that facilitates downstream purification.
Mechanistic Insights into Base-Mediated Aerobic Oxidation
The core of this technological advancement lies in the unique mechanistic pathway facilitated by the strong base and molecular oxygen. In the presence of potassium hydroxide, the alpha-proton of the amide substrate is abstracted to form a resonance-stabilized enolate intermediate. This nucleophilic species subsequently reacts with dissolved oxygen from the air, initiating a radical oxidation sequence that installs the hydroxyl functionality directly onto the carbon skeleton. The use of DMSO as a polar aprotic solvent is critical, as it stabilizes the ionic intermediates and enhances the solubility of both the organic substrate and the inorganic base. This mechanism avoids the formation of unstable acyl halides or organometallic complexes, resulting in a reaction profile that is inherently safer and more predictable for large-scale operations. The absence of transition metals means there is no risk of catalyst poisoning or the need for specialized ligands, making the process robust against variations in substrate electronic properties.
From an impurity control perspective, this metal-free oxidation strategy offers distinct advantages for the production of high-purity pharmaceutical intermediates. Traditional palladium-catalyzed routes often leave trace residues of heavy metals that require costly and time-consuming removal steps, such as treatment with activated carbon or specialized scavengers, to comply with ICH Q3D guidelines. In this aerobic oxidation system, the primary byproducts are water and inorganic salts, which are easily removed during the standard aqueous workup procedure involving saturated brine and ethyl acetate extraction. The high selectivity of the base-mediated oxidation minimizes the formation of over-oxidized byproducts like alpha-keto amides, ensuring that the crude product obtained after concentration is of sufficient quality for final purification. This inherent cleanliness of the reaction mixture translates directly into higher recovery rates during column chromatography and reduced solvent usage, aligning perfectly with green chemistry principles.
How to Synthesize Alpha-Hydroxy Amides Efficiently
The implementation of this synthesis route is designed for practicality, allowing chemists to produce target compounds with minimal equipment specialization. The process begins with the precise mixing of the amide substrate, the base, and the solvent in a sealed reaction vessel, ensuring an adequate supply of air for the oxidation step. Following the reaction period, the workup procedure is straightforward, involving cooling, washing, and extraction, which are unit operations familiar to any standard chemical plant. For detailed operational parameters and specific stoichiometric ratios tailored to different substrates, please refer to the standardized synthesis guide below.
- Mix the specific amide substrate with a strong base (potassium hydroxide or potassium tert-butoxide) in dimethyl sulfoxide (DMSO) solvent within a sealed vessel under an air atmosphere.
- Heat the reaction mixture to a moderate temperature range of 50°C to 80°C and maintain magnetic stirring for a duration of 12 to 36 hours to ensure complete conversion.
- Upon completion, cool the mixture, perform an aqueous workup with saturated brine, extract with ethyl acetate, dry over anhydrous sodium sulfate, and purify the crude product via silica gel column chromatography.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this technology addresses several critical pain points associated with the sourcing of complex organic intermediates. The elimination of expensive catalysts and toxic reagents fundamentally alters the cost structure of the manufacturing process, leading to substantial savings in raw material expenditure. Moreover, the reliance on commodity chemicals like potassium hydroxide and DMSO ensures a stable and resilient supply chain, unaffected by the geopolitical fluctuations that often impact the availability of precious metals or specialized reagents. The simplified process flow also reduces the lead time for production batches, enabling faster response to market demands and more agile inventory management strategies for downstream API manufacturers.
- Cost Reduction in Manufacturing: The most significant economic driver of this technology is the complete removal of transition metal catalysts, such as palladium, which are subject to high market volatility and add considerable expense to the bill of materials. By replacing these with inexpensive inorganic bases and utilizing free atmospheric oxygen as the oxidant, the variable cost per kilogram of the product is drastically reduced. Additionally, the high yield and selectivity of the reaction minimize the loss of valuable starting materials, further enhancing the overall process economics and providing a competitive pricing advantage in the global market for pharmaceutical intermediates.
- Enhanced Supply Chain Reliability: Sourcing strategies are greatly simplified as the key reagents—potassium hydroxide, potassium tert-butoxide, and dimethyl sulfoxide—are bulk commodity chemicals available from multiple global suppliers with consistent quality. This diversification of the supply base mitigates the risk of single-source dependency that plagues processes relying on proprietary ligands or rare earth catalysts. The robustness of the reaction conditions also means that production is less susceptible to delays caused by the need for specialized handling or storage of sensitive reagents, ensuring a continuous and reliable flow of materials to support uninterrupted API synthesis schedules.
- Scalability and Environmental Compliance: The transition from laboratory to commercial scale is facilitated by the mild thermal requirements and the absence of exothermic hazards associated with strong oxidizing agents like chromates or permanganates. The use of air as an oxidant eliminates the need for high-pressure oxygen cylinders, reducing safety risks and capital investment in pressure-rated equipment. Furthermore, the generation of minimal hazardous waste aligns with increasingly stringent environmental regulations, lowering the costs associated with waste treatment and disposal while enhancing the corporate sustainability profile of the manufacturing facility.
Frequently Asked Questions (FAQ)
The following questions address common technical inquiries regarding the implementation and benefits of this novel synthesis method. These insights are derived directly from the experimental data and comparative analysis presented in the patent documentation, providing clarity on how this technology outperforms existing state-of-the-art methods in terms of efficiency and safety.
Q: Does this synthesis method require expensive transition metal catalysts?
A: No, the patented method (CN103755584A) specifically avoids the use of complex or expensive transition metal catalysts like palladium, utilizing a simple potassium hydroxide and DMSO system instead.
Q: What is the primary oxidant used in this green chemistry approach?
A: The process utilizes atmospheric air as the green oxidant, which directly converts the substrate to the target alpha-hydroxy amide in a single step, significantly reducing chemical waste.
Q: How does the yield of this method compare to traditional acid chloride routes?
A: While traditional methods often suffer from low yields due to side reactions and protection steps, this novel aerobic oxidation method achieves yields as high as 98%, demonstrating superior efficiency for industrial production.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Alpha-Hydroxy Amides Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of the catalyst-free aerobic oxidation technology described in CN103755584A for the production of high-value pharmaceutical intermediates. As a dedicated CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the theoretical benefits of this green chemistry approach are fully realized in a GMP-compliant environment. Our rigorous QC labs and stringent purity specifications guarantee that every batch of alpha-hydroxy amides meets the exacting standards required for drug substance manufacturing, free from the heavy metal contaminants often associated with traditional catalytic routes.
We invite forward-thinking pharmaceutical companies to collaborate with us to leverage this cost-effective and sustainable synthesis platform. By partnering with our technical procurement team, you can obtain a Customized Cost-Saving Analysis that quantifies the economic benefits of switching to this metal-free process for your specific pipeline. We encourage you to contact us today to request specific COA data and route feasibility assessments, allowing us to demonstrate how our advanced manufacturing capabilities can accelerate your development timelines and optimize your supply chain costs.
