Advanced One-Pot Synthesis of 3-Cephem Intermediates for Scalable Antibiotic Production
The pharmaceutical industry continuously seeks robust synthetic routes for beta-lactam antibiotics, particularly 3-cephem derivatives which serve as critical scaffolds for oral cephalosporins like Cefixime and Cefdinir. Patent CN1126754C discloses a groundbreaking preparation method that addresses long-standing stability and cost issues associated with traditional 3-cephem synthesis. By utilizing a tandem reaction strategy involving phosphorus halides and phenols, this technology enables the direct conversion of protected beta-lactam precursors into stable 3-cephem compounds without isolating unstable intermediates. For global procurement teams and R&D directors, this represents a significant opportunity to optimize the supply chain for high-purity pharmaceutical intermediates. The process not only simplifies the operational workflow but also mitigates the risks associated with handling decomposition-prone species, ensuring a more reliable supply of key antibiotic building blocks for downstream formulation.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of 3-cephem derivatives represented by general formula (3) relied heavily on the substitution reactions of 7-aminocephalosporanic acid (7-ACA) or multi-step sequences involving unstable intermediates. A prevalent prior art method, such as that described by Hatfield, necessitates the isolation of a carboxylate ester intermediate (Formula 4) before deprotection. This intermediate is chemically fragile and susceptible to rapid decomposition during separation and purification processes, resulting in inconsistent yields and compromised product quality. Furthermore, conventional alcoholysis steps often mandate the use of expensive branched-chain fatty alcohols to achieve acceptable reaction kinetics, which inflates raw material costs. The subsequent deprotection phase typically requires harsh conditions, such as noble metal catalysis or strong acids like trifluoroacetic acid, which introduce additional challenges regarding catalyst poisoning by sulfur atoms and difficult solvent recovery, thereby creating bottlenecks in commercial manufacturing.
The Novel Approach
The innovative process detailed in the patent data revolutionizes this workflow by integrating alcoholysis and deprotection into a single, seamless operation within the same reaction vessel. By reacting the starting beta-lactam compound (Formula 1) with a phosphorus halide and an organic base, an imino-beta-lactam intermediate (Formula 2) is generated in situ. Crucially, instead of isolating the subsequent unstable ester, phenols are introduced directly to the reaction mixture. These phenols function dually as alcoholysis reagents and deprotecting agents, facilitating the immediate conversion to the stable 3-cephem compound (Formula 3). 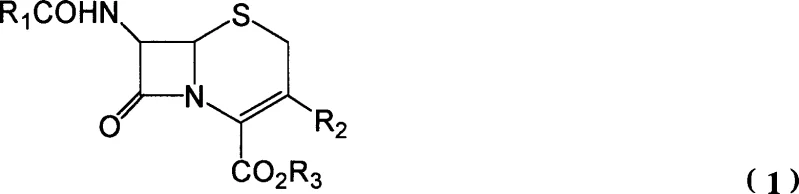 This one-pot strategy effectively bypasses the isolation of the decomposition-prone Formula 4 intermediate, significantly enhancing overall process stability. The elimination of separate deprotection steps and the replacement of costly branched alcohols with readily available phenols streamline the production line, offering a compelling solution for cost reduction in pharmaceutical intermediate manufacturing.
This one-pot strategy effectively bypasses the isolation of the decomposition-prone Formula 4 intermediate, significantly enhancing overall process stability. The elimination of separate deprotection steps and the replacement of costly branched alcohols with readily available phenols streamline the production line, offering a compelling solution for cost reduction in pharmaceutical intermediate manufacturing.
Mechanistic Insights into Phenol-Mediated Tandem Deprotection
The core mechanistic advantage of this synthesis lies in the dual functionality of the phenolic additives within the reaction matrix. Upon activation of the beta-lactam nitrogen by the phosphorus halide complex, the system becomes highly reactive towards nucleophilic attack. When phenols are introduced, they act as potent nucleophiles to effectuate the alcoholysis of the activated species. Simultaneously, the acidic nature and specific electronic properties of the phenols promote the cleavage of the carboxylate protecting group (R3), which is typically an electron-rich benzyl or diphenylmethyl ester. This concurrent mechanism ensures that the potentially unstable carboxylate ester is never allowed to accumulate in the reaction mixture. Instead, it is instantaneously converted into the free carboxylic acid form of the 3-cephem skeleton. This kinetic control is vital for maintaining high purity, as it prevents the formation of degradation byproducts that typically arise when unstable intermediates are subjected to prolonged isolation procedures or varying thermal conditions during workup.
From an impurity control perspective, this method offers superior selectivity compared to traditional acidolytic or hydrogenolytic deprotection methods. Conventional acid deprotection using trifluoroacetic acid often leads to the decomposition of the acid-labile beta-lactam ring, reducing the yield of the desired carboxylic acid. Similarly, catalytic hydrogenation is frequently hampered by catalyst poisoning due to the presence of sulfur atoms within the cephem nucleus. The phenol-mediated approach operates under milder conditions, typically between 0°C and 50°C, which preserves the integrity of the sensitive beta-lactam ring. By avoiding strong mineral acids and noble metal catalysts, the process minimizes the generation of metal residues and acid-induced degradation products. This results in a cleaner crude product profile, reducing the burden on downstream purification units and ensuring that the final active pharmaceutical ingredient meets stringent regulatory specifications for residual solvents and impurities.
How to Synthesize 3-Cephem Compounds Efficiently
Implementing this synthesis route requires precise control over reaction parameters to maximize the benefits of the tandem mechanism. The process begins with the activation of the starting material using a phosphorus halide, such as phosphorus pentachloride or phosphorus oxychloride, in the presence of an organic base like pyridine or triethylamine. This step must be conducted at controlled low temperatures, generally ranging from -30°C to 30°C, to ensure the selective formation of the imino-intermediate without inducing side reactions. Following activation, the phenolic reagent is added to the same reactor, and the temperature is adjusted to facilitate the coupled alcoholysis and deprotection. Detailed standard operating procedures regarding stoichiometry, solvent selection, and quenching protocols are essential for reproducibility. The detailed standardized synthesis steps are outlined in the guide below.
- React the starting beta-lactam compound (Formula 1) with a phosphorus halide and organic base at low temperatures (-30 to 30°C) to form the imino-beta-lactam intermediate.
- Without isolating the intermediate, add phenol directly to the reaction system to perform simultaneous alcoholysis and carboxylate deprotection.
- Quench the reaction with water, adjust pH to precipitate the stable 3-cephem product (Formula 3), and purify via crystallization.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this patented methodology translates into tangible operational efficiencies and risk mitigation. The primary economic driver is the substantial simplification of the process flow, which directly correlates to reduced manufacturing costs. By eliminating the isolation of the unstable intermediate Formula 4, the process removes an entire unit operation, including filtration, drying, and re-dissolution steps. This reduction in unit operations lowers energy consumption, decreases labor requirements, and minimizes the physical footprint required for production. Furthermore, the substitution of expensive branched-chain fatty alcohols with cost-effective phenols significantly reduces raw material expenditure. The avoidance of noble metal catalysts not only cuts material costs but also removes the complex and costly steps associated with metal scavenging and validation, which are critical in GMP environments.
- Cost Reduction in Manufacturing: The economic benefits of this process are driven by the consolidation of multiple reaction steps into a single reactor setup. Traditional methods often require distinct vessels for activation, alcoholysis, isolation, and deprotection, each incurring capital and operational expenses. By combining alcoholysis and deprotection, the new method drastically reduces solvent usage and waste generation. The elimination of noble metal catalysts removes the need for expensive palladium or platinum inputs and the associated recovery infrastructure. Additionally, the use of phenols, which are commodity chemicals, replaces specialized, high-cost alcohols, leading to a more favorable bill of materials. These factors collectively contribute to a leaner manufacturing model that enhances margin potential for high-volume antibiotic production.
- Enhanced Supply Chain Reliability: Supply chain resilience is significantly bolstered by the robustness of this synthetic route. Conventional methods that rely on unstable intermediates are prone to batch failures and yield fluctuations, which can disrupt supply schedules. The one-pot nature of this process minimizes handling of sensitive materials, thereby increasing batch success rates and consistency. Moreover, the reagents involved, such as phosphorus halides and common phenols, are widely available in the global chemical market, reducing the risk of raw material shortages. The simplified workflow also shortens the overall cycle time from raw material intake to finished intermediate, allowing for faster response to market demand fluctuations and improved inventory turnover rates for critical pharmaceutical ingredients.
- Scalability and Environmental Compliance: Scaling this process to commercial volumes is facilitated by its straightforward engineering requirements. Operating in a single reactor reduces the complexity of plant design and the potential for cross-contamination between steps. From an environmental standpoint, the process generates less waste solvent and avoids the disposal issues associated with heavy metal catalysts. The reduced use of hazardous acids like trifluoroacetic acid further aligns with green chemistry principles and stricter environmental regulations. This compliance ease lowers the regulatory burden and waste treatment costs, making the process highly attractive for large-scale facilities aiming to meet sustainability goals while maintaining high production throughput for complex pharmaceutical intermediates.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this 3-cephem synthesis technology. These insights are derived directly from the experimental data and comparative analysis provided in the patent documentation. Understanding these nuances is crucial for technical teams evaluating the feasibility of technology transfer and for procurement specialists assessing the long-term viability of the supply source. The answers highlight the specific advantages in terms of yield stability, impurity profiles, and operational simplicity that distinguish this method from legacy processes.
Q: Why is the conventional separation of intermediate Formula 4 problematic?
A: Conventional methods require isolating the carboxylate ester of Formula 4, which is inherently unstable and prone to decomposition during purification, leading to significant yield loss and purity issues.
Q: How does the phenol-mediated method improve cost efficiency?
A: This method eliminates the need for expensive branched-chain fatty alcohols and noble metal catalysts, while combining alcoholysis and deprotection into a single step, drastically reducing solvent usage and processing time.
Q: Is this process suitable for large-scale antibiotic manufacturing?
A: Yes, the process is designed for single-reactor operation, avoiding complex isolation steps of unstable intermediates, which enhances operational safety and scalability for commercial production of cephalosporins.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 3-Cephem Compounds Supplier
As the global demand for high-quality beta-lactam antibiotics continues to rise, securing a supply partner with deep technical expertise in advanced synthesis is paramount. NINGBO INNO PHARMCHEM stands at the forefront of this sector, leveraging cutting-edge processes like the phenol-mediated deprotection technique to deliver superior pharmaceutical intermediates. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the theoretical benefits of this patent are fully realized in a GMP-compliant environment. We maintain stringent purity specifications and operate rigorous QC labs to guarantee that every batch of 3-cephem compound meets the exacting standards required for downstream antibiotic synthesis, providing our partners with unwavering confidence in product quality.
We invite international pharmaceutical companies and contract manufacturers to collaborate with us to optimize their supply chains. By adopting this efficient synthesis route, we can offer a Customized Cost-Saving Analysis tailored to your specific volume requirements and quality targets. We encourage you to contact our technical procurement team to request specific COA data and route feasibility assessments. Together, we can drive down the cost of goods for essential antibiotics while ensuring a stable, continuous supply of high-purity intermediates, positioning your organization for success in the competitive global healthcare market.
