Advanced Synthesis of Cephem Onium Salts for Commercial Cefpirazole Production
Advanced Synthesis of Cephem Onium Salts for Commercial Cefpirazole Production
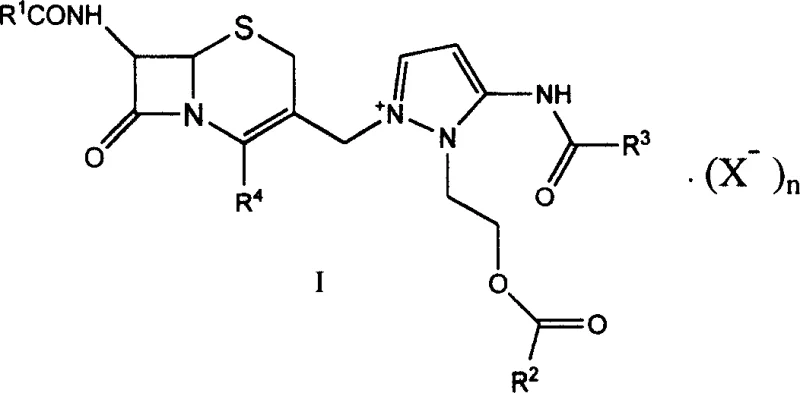
The pharmaceutical industry constantly seeks robust and scalable pathways for producing third-generation cephalosporins, and patent CN1613860A presents a significant breakthrough in this domain. This intellectual property discloses a novel class of cephem-type onium salt compounds, specifically 7β-alkanoylamino-3-[3-alkanoylamino-2-(2-alkanoyloxyethyl)-1-pyrazolium]methyl-3-cephem-4-carboxylates, which serve as critical intermediates for the synthesis of Cefpirazole Sulfate. Unlike traditional methods that rely on hazardous and expensive reagents, this innovation introduces a streamlined approach that enhances both safety and economic viability. For R&D directors and procurement specialists, understanding the structural nuances and synthetic advantages of this intermediate is paramount for optimizing supply chains and reducing manufacturing costs. The following analysis delves into the technical specifics of this patent, highlighting its potential to redefine the production standards for high-purity antibiotic intermediates in the global market.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of cefpirazole and related cephalosporins has been plagued by the use of aggressive chemical reagents that pose significant safety and environmental challenges. Prior art, such as the methods described in Japanese Patent JP 4-173792 and Chinese Patent 88106644.3, often necessitates the utilization of phosphorus pentachloride, phosgene, or sodium iodide to facilitate key transformation steps. These reagents are not only costly but also highly corrosive and toxic, requiring specialized handling equipment and rigorous waste treatment protocols that drive up operational expenditures. Furthermore, the harsh reaction conditions associated with these traditional halogenating agents can lead to the formation of unwanted by-products and degradation of the sensitive beta-lactam ring, thereby compromising the overall yield and purity of the final active pharmaceutical ingredient. For supply chain managers, reliance on such volatile chemistries introduces risks related to regulatory compliance and the availability of specialized raw materials, making the search for alternative routes a strategic imperative.
The Novel Approach
In stark contrast to the legacy methodologies, the process outlined in patent CN1613860A offers a remarkably温和 (mild) and efficient alternative that circumvents the need for dangerous halogenating agents. The core innovation lies in the preparation of the cephem onium salt intermediate through a direct quaternization reaction between a 7β-alkanoylamino-3-chloromethyl-3-cephem-4-carboxylic acid and a specific pyrazole derivative. This approach eliminates the requirement for expensive silyl amide reagents and iodine salts, substituting them with readily available and cost-effective bases and organic solvents. By shifting the synthetic paradigm to these milder conditions, typically ranging from -20°C to 60°C, the process significantly reduces the thermal stress on the molecular structure, preserving the integrity of the beta-lactam core. This technological shift not only simplifies the operational workflow but also aligns with modern green chemistry principles, offering a sustainable pathway for the commercial scale-up of complex antibiotic intermediates without the burden of hazardous waste management.
Mechanistic Insights into Quaternization and Acylation
The mechanistic elegance of this synthesis is rooted in the precise control of the quaternization step, where the nucleophilic nitrogen of the pyrazole ring attacks the electrophilic chloromethyl group on the cephem nucleus. This reaction is facilitated by the presence of a suitable base, such as sodium bicarbonate or triethylamine, which neutralizes the hydrochloric acid by-product and drives the equilibrium towards the formation of the onium salt. The choice of solvent plays a critical role in this mechanism, with polar aprotic solvents like dimethylformamide or sulfolane proving particularly effective in solubilizing the reactants and stabilizing the transition state. This careful orchestration of reaction parameters ensures high conversion rates while minimizing the risk of epimerization at the chiral centers, a common pitfall in cephalosporin chemistry that can drastically reduce biological activity. For technical teams, understanding this mechanism is essential for troubleshooting and optimizing reaction kinetics during process validation.
Furthermore, the subsequent acylation of the 7-amino group and the final coupling with the AE active ester are designed to maximize regioselectivity and minimize impurity generation. The use of the AE active ester, specifically α-(2-aminothiazol-4-yl)-α-(Z)-methoxyiminoacetic acid benzothiazol-2-yl sulfhydryl ester, allows for a highly efficient coupling reaction under neutral to slightly basic conditions. This specific active ester is known for its superior reactivity compared to traditional acid chlorides, enabling the reaction to proceed rapidly at room temperature without the need for additional catalysts that might introduce metallic contaminants. The result is a final product with a cleaner impurity profile, which simplifies downstream purification processes such as crystallization and filtration. This level of control over the chemical trajectory is vital for meeting the stringent purity specifications required by global regulatory bodies for injectable antibiotics.
How to Synthesize Cephem Onium Salt Efficiently
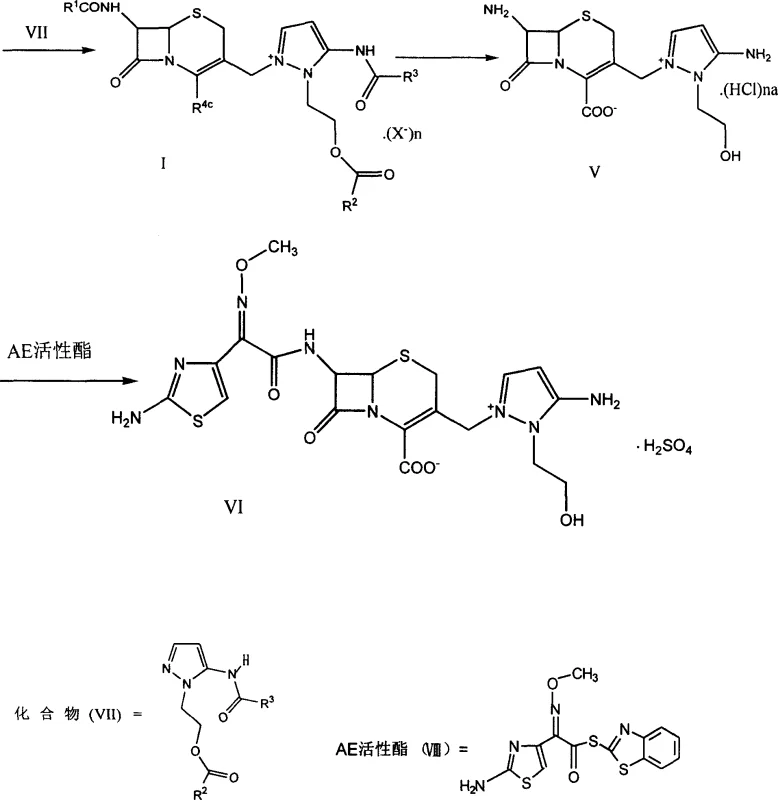
The synthesis of the target cephem onium salt involves a sequence of well-defined steps that begin with the protection of the 7-amino group followed by deprotection of the carboxyl function and final quaternization. This route is characterized by its operational simplicity, utilizing standard laboratory equipment and common chemical reagents that are easily sourced from global suppliers. The process avoids the need for cryogenic temperatures or high-pressure vessels, making it accessible for manufacturing facilities with standard infrastructure. Detailed procedural knowledge is critical for replicating the high yields reported in the patent, particularly regarding the stoichiometry of the base and the choice of precipitation solvents. For a comprehensive understanding of the standardized synthetic steps and specific reaction parameters, please refer to the guide below.
- Protect the 7β-amino group of the starting cephalosporin via acylation to form the 7β-alkanoylamino derivative.
- Remove the carboxyl protecting group to generate the free acid intermediate suitable for quaternization.
- React the chloromethyl cephem with a pyrazole derivative in the presence of a base to form the target onium salt.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, the adoption of this synthesis route offers substantial strategic benefits for procurement managers and supply chain heads looking to optimize their antibiotic portfolios. The primary advantage lies in the drastic simplification of the raw material list, replacing exotic and expensive reagents with commodity chemicals that are subject to less price volatility. This shift not only lowers the direct cost of goods sold but also mitigates the risk of supply disruptions caused by the limited availability of specialized catalysts or halogenating agents. Additionally, the mild reaction conditions translate to lower energy consumption and reduced wear and tear on processing equipment, contributing to long-term operational savings. For organizations focused on cost reduction in cephalosporin manufacturing, this technology represents a viable pathway to enhance margin stability and competitive pricing in the generic drug market.
- Cost Reduction in Manufacturing: The elimination of expensive reagents such as sodium iodide and silyl amides directly impacts the bottom line by reducing the variable costs associated with each batch production. Furthermore, the avoidance of corrosive agents like phosgene reduces the capital expenditure required for specialized corrosion-resistant reactors and containment systems. The simplified workup procedure, which often involves straightforward precipitation and filtration, minimizes solvent usage and recovery costs, leading to a more lean and efficient manufacturing process. These cumulative savings allow for a more aggressive pricing strategy while maintaining healthy profit margins, a critical factor in the highly competitive landscape of generic antibiotics.
- Enhanced Supply Chain Reliability: By relying on widely available starting materials and solvents, the supply chain becomes more resilient to geopolitical shocks and logistical bottlenecks. The robustness of the synthetic route ensures consistent output quality, reducing the frequency of batch failures and the associated costs of reprocessing or disposal. This reliability is crucial for maintaining uninterrupted supply to downstream formulation partners and meeting contractual delivery obligations. Moreover, the reduced hazard profile of the process simplifies regulatory filings and inspections, accelerating the time-to-market for new generic versions of cefpirazole and ensuring continuous availability for patients.
- Scalability and Environmental Compliance: The process is inherently scalable, having been designed with commercial production in mind, allowing for seamless transition from pilot plant to multi-ton annual capacity. The mild conditions and absence of heavy metal catalysts simplify waste treatment, making it easier to comply with increasingly stringent environmental regulations regarding effluent discharge. This environmental compatibility not only reduces the cost of waste management but also enhances the corporate sustainability profile, which is becoming a key differentiator in B2B procurement decisions. The ability to scale up complex antibiotic intermediates without compromising safety or quality is a significant asset for any contract development and manufacturing organization.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the synthesis and application of the cephem onium salt intermediate described in patent CN1613860A. These answers are derived directly from the patent specifications and are intended to provide clarity on the feasibility and advantages of this manufacturing route. Understanding these details is essential for stakeholders evaluating the potential integration of this technology into their existing production lines. The responses cover aspects ranging from reaction safety to product purity, offering a holistic view of the process capabilities.
Q: What are the advantages of this new cephem onium salt synthesis method?
A: The method avoids hazardous reagents like phosgene and phosphorus pentachloride, utilizing mild reaction conditions and inexpensive raw materials for safer and more cost-effective production.
Q: How does this process improve impurity control in cefpirazole manufacturing?
A: By operating at lower temperatures and avoiding harsh halogenating agents, the process minimizes side reactions and degradation, resulting in a higher purity profile for the final antibiotic.
Q: Is this synthesis route scalable for industrial production?
A: Yes, the use of common organic solvents and simple workup procedures, such as precipitation and filtration, makes the process highly adaptable for large-scale commercial manufacturing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Cefpirazole Intermediate Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of high-quality intermediates in the production of life-saving antibiotics, and we are uniquely positioned to support your manufacturing needs with our advanced technical capabilities. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that we can meet your volume requirements with consistency and precision. We adhere to stringent purity specifications and operate rigorous QC labs to guarantee that every batch of cephem onium salt meets the highest international standards. Our commitment to excellence extends beyond mere compliance; we actively collaborate with our partners to optimize processes for maximum efficiency and cost-effectiveness, leveraging our deep understanding of cephalosporin chemistry to deliver superior results.
We invite you to engage with our technical procurement team to discuss how our capabilities can align with your strategic goals for cefpirazole production. By requesting a Customized Cost-Saving Analysis, you can gain valuable insights into how our optimized synthesis route can reduce your overall manufacturing expenses. We encourage you to contact us today to obtain specific COA data and route feasibility assessments tailored to your project requirements. Let us be your trusted partner in navigating the complexities of antibiotic synthesis, ensuring a reliable supply of high-purity intermediates for your global operations.
