Advanced 3-Step Synthesis of Venlafaxine Impurity E for Global Pharmaceutical Quality Control
The pharmaceutical industry's relentless pursuit of quality and safety has placed unprecedented scrutiny on the impurity profiles of Active Pharmaceutical Ingredients (APIs). In this context, Patent CN111675671A introduces a groundbreaking preparation method for Venlafaxine Impurity E, a critical reference standard required for the quality control of the antidepressant Venlafaxine. This patent discloses a highly efficient, three-step synthetic route that fundamentally reimagines the production of this complex spiro-cyclic compound. Unlike traditional methodologies that often suffer from excessive step counts and hazardous operating conditions, this novel approach leverages a strategic combination of condensation, Lewis acid-mediated reduction, and cyclization. For global procurement teams and R&D directors, this represents a significant opportunity to secure a reliable pharmaceutical intermediate supplier capable of delivering high-purity materials with enhanced supply chain stability. The technical breakthrough lies not just in the chemical transformation itself, but in the holistic optimization of the process parameters, ensuring that the final product meets the stringent purity specifications demanded by modern pharmacopoeias while simultaneously addressing critical environmental and safety concerns inherent in fine chemical manufacturing.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
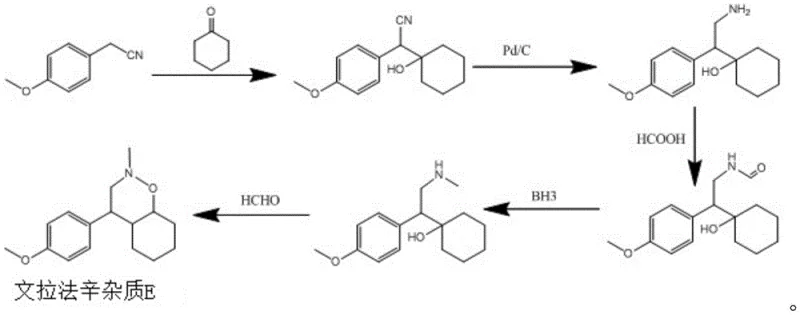
Historically, the synthesis of complex spiro-cyclic intermediates like Venlafaxine Impurity E has been plagued by inefficient multi-step sequences that introduce significant bottlenecks in production. As illustrated in the prior art, conventional routes typically involve a cumbersome five-step process that relies heavily on transition metal catalysis and hazardous reagents. Specifically, the traditional pathway necessitates the use of Palladium on Carbon (Pd/C) for hydrogenation, which requires specialized high-pressure equipment and introduces the risk of heavy metal contamination in the final product. Furthermore, these legacy methods often employ borane complexes for reduction steps, which are notoriously pyrophoric and demand rigorous safety protocols and inert atmosphere handling. The cumulative effect of these factors is a process that is not only capital intensive due to the need for specialized infrastructure but also operationally fragile. The extensive post-treatment required to remove catalyst residues and by-products generates substantial volumes of waste liquid and solid waste, creating a heavy environmental burden that conflicts with modern green chemistry initiatives. Consequently, the low overall yield and purity associated with these lengthy sequences make them economically unsustainable for large-scale commercial operations.
The Novel Approach
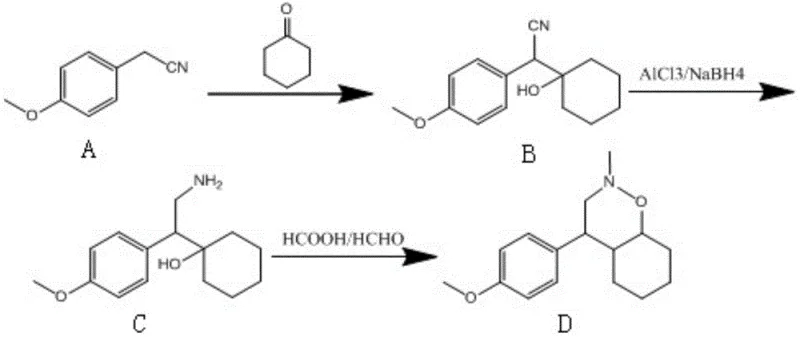
In stark contrast to the convoluted legacy pathways, the methodology described in Patent CN111675671A streamlines the synthesis into a robust three-step sequence that maximizes atom economy and operational safety. The new route initiates with a straightforward condensation reaction between 4-methoxyphenylacetonitrile and cyclohexanone, followed by a highly selective reduction using an aluminum trichloride and sodium borohydride system. This specific reagent combination is pivotal as it facilitates the reduction of the nitrile group to a primary amine under mild reflux conditions, effectively bypassing the need for high-pressure hydrogenation. The final step involves a tandem cyclization and methylation using formic acid and formaldehyde, which elegantly constructs the target oxa-aza-spiro ring system in a single pot. This consolidation of steps drastically reduces the time and resources required for isolation and purification between stages. By eliminating the reliance on precious metal catalysts and dangerous borane reagents, the novel approach not only enhances the safety profile of the manufacturing process but also simplifies the downstream processing, resulting in a cleaner product profile with significantly reduced impurity loads. This strategic redesign exemplifies how modern process chemistry can achieve cost reduction in API manufacturing through intelligent route selection rather than mere optimization of existing flawed processes.
Mechanistic Insights into AlCl3/NaBH4 Mediated Reduction and Cyclization
The core innovation of this synthetic strategy resides in the second step, where the nitrile functionality is reduced to a primary amine using a generated alane species in situ. When aluminum trichloride (AlCl3) is mixed with sodium borohydride (NaBH4) in tetrahydrofuran (THF), it generates a potent reducing environment capable of converting nitriles to amines with high chemoselectivity. Unlike catalytic hydrogenation which can sometimes lead to over-reduction or hydrogenolysis of sensitive functional groups, this hydride transfer mechanism is controlled by the stoichiometry of the reagents and the temperature of the reflux. The Lewis acidic nature of the aluminum species likely activates the nitrile nitrogen, facilitating the nucleophilic attack by the hydride ion. This mechanistic pathway is particularly advantageous for substrates containing other reducible groups, as it offers a level of tunability that gaseous hydrogen cannot match. Furthermore, the reaction conditions (reflux in THF) are easily scalable in standard glass-lined reactors, removing the barrier of entry for manufacturers who lack high-pressure autoclaves. The subsequent quenching with acid not only neutralizes the reaction mixture but also helps in the hydrolysis of any aluminum complexes, ensuring that the free amine is released efficiently for the next stage. This precise control over the reduction step is fundamental to achieving the high purity levels observed in the final product, as it minimizes the formation of side products that often arise from non-selective reduction methods.
Following the reduction, the cyclization step employs a classic Eschweiler-Clarke type mechanism adapted for ring closure. The interaction between the newly formed primary amine, formaldehyde, and formic acid leads to the formation of an iminium intermediate, which is subsequently reduced by the formic acid to the tertiary amine. In this specific molecular architecture, the proximity of the hydroxyl group on the cyclohexane ring allows for an intramolecular nucleophilic attack, closing the ring to form the stable six-membered oxazine moiety fused to the spiro-center. The use of formic acid serves a dual purpose: it acts as both the source of the methyl group (via formaldehyde activation) and the reducing agent for the iminium ion. This one-pot transformation is thermodynamically driven and proceeds smoothly at temperatures between 50°C and 80°C. The robustness of this cyclization is evident in the consistent liquid phase purity of over 99% reported in the patent examples. By understanding these mechanistic nuances, R&D teams can better appreciate the reliability of this high-purity pharmaceutical intermediate production method, ensuring that the material supplied for analytical method validation or toxicological studies is of the highest possible quality.
How to Synthesize Venlafaxine Impurity E Efficiently
The practical implementation of this synthesis requires careful attention to reagent ratios and temperature control to maximize yield and purity. The process begins with the condensation of 4-methoxyphenylacetonitrile and cyclohexanone in an ethanol solvent system, where the pH is carefully managed using a strong base like sodium hydroxide to initiate the nucleophilic attack. Following the isolation of the cyano-intermediate, the critical reduction step is performed in THF, requiring a precise mass ratio of aluminum trichloride to the substrate to ensure complete conversion without excessive reagent waste. The final cyclization is a straightforward heating process with formic acid and formaldehyde, but the workup involving ethyl acetate extraction and vacuum distillation is crucial for removing residual acids and solvents. While the general workflow is outlined here, the specific operational parameters such as stirring rates, addition times, and exact quenching protocols are vital for reproducibility. For detailed standardized operating procedures and specific batch records, please refer to the comprehensive guide below.
- Condensation Reaction: Dissolve 4-methoxyphenylacetonitrile in ethanol, adjust to alkaline with strong base, add cyclohexanone, react at 15-30°C for 4-5 hours, then quench with acid to obtain the cyano-intermediate.
- Reduction Reaction: In THF solvent, mix aluminum trichloride and sodium borohydride, add the cyano-intermediate, reflux for 3-4 hours, quench with acid, and extract to obtain the amino-alcohol intermediate.
- Cyclization Reaction: React the amino-alcohol intermediate with formic acid and formaldehyde at 50-80°C for 2-3 hours, followed by extraction and distillation to isolate the final spiro-cyclic product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this novel synthetic route offers tangible benefits that extend far beyond simple chemical curiosity. The primary advantage lies in the drastic simplification of the supply chain for raw materials. By eliminating the need for high-pressure hydrogen gas and specialized catalysts like Palladium on Carbon, manufacturers can source reagents from a much broader and more stable vendor base. Aluminum trichloride and sodium borohydride are commodity chemicals with established global supply chains, reducing the risk of production stoppages due to raw material shortages. Furthermore, the removal of the hydrogenation step eliminates the need for costly maintenance and certification of high-pressure vessels, leading to significant capital expenditure savings and lower overhead costs per kilogram of product. This operational efficiency translates directly into a more competitive pricing structure for the final intermediate, allowing downstream API manufacturers to optimize their cost of goods sold (COGS). Additionally, the simplified post-treatment process reduces the consumption of solvents and the generation of hazardous waste, aligning with increasingly strict environmental regulations and reducing disposal costs.
- Cost Reduction in Manufacturing: The elimination of precious metal catalysts such as Palladium on Carbon removes a major cost driver from the bill of materials. Precious metals are subject to volatile market prices and require expensive recovery or disposal processes to meet regulatory limits on heavy metals in pharmaceuticals. By switching to a base metal-mediated reduction using aluminum and boron, the process inherently lowers the material cost. Moreover, the reduction in step count from five to three significantly decreases the cumulative loss of material at each isolation stage, thereby improving the overall yield and reducing the amount of starting material required to produce a fixed quantity of the final product. This efficiency gain is compounded by the reduced energy consumption, as the process operates at atmospheric pressure and moderate temperatures, avoiding the high energy inputs associated with high-pressure hydrogenation reactors.
- Enhanced Supply Chain Reliability: Dependence on specialized equipment and hazardous reagents often creates single points of failure in a supply chain. The new method's reliance on standard glass-lined or stainless steel reactors and common organic solvents like ethanol and ethyl acetate means that production can be easily transferred between different manufacturing sites without significant retrofitting. This flexibility is crucial for maintaining supply continuity in the face of geopolitical disruptions or local regulatory changes. The use of non-pyrophoric reagents also simplifies logistics and storage, as the materials do not require the same level of hazardous material handling and transportation permits as borane complexes or compressed hydrogen. This ease of handling ensures that production schedules can be maintained with greater predictability, reducing lead times for customers awaiting critical quality control standards.
- Scalability and Environmental Compliance: Scaling chemical processes from the laboratory to industrial production often reveals hidden complexities, particularly regarding heat transfer and mixing in exothermic reactions. The proposed method utilizes reactions that are well-suited for scale-up, with the condensation and cyclization steps being relatively benign in terms of thermal runaway risk. The reduction step, while exothermic, is managed through controlled addition in a refluxing solvent, a technique that is easily replicated at the 100 MT scale. From an environmental perspective, the reduction in waste liquid and the absence of heavy metal contaminants simplify the wastewater treatment process. This not only lowers the cost of environmental compliance but also positions the manufacturer as a sustainable partner, a factor that is increasingly weighted in the vendor selection criteria of major multinational pharmaceutical companies. The ability to produce commercial scale-up of complex pharmaceutical intermediates with a smaller environmental footprint is a distinct competitive advantage in the current market landscape.
Frequently Asked Questions (FAQ)
Understanding the technical and commercial implications of this new synthesis method is essential for stakeholders involved in the procurement and quality assurance of pharmaceutical intermediates. The following questions address common inquiries regarding the safety, purity, and scalability of the process described in Patent CN111675671A. These answers are derived directly from the technical disclosures and experimental data provided in the patent documentation, ensuring accuracy and relevance for decision-makers. Whether you are evaluating this material for method validation or considering it for long-term supply contracts, these insights provide a clear picture of the technology's capabilities.
Q: How does the new AlCl3/NaBH4 reduction method improve safety over traditional hydrogenation?
A: The novel protocol eliminates the need for high-pressure hydrogenation equipment and hazardous borane reagents. By utilizing aluminum trichloride and sodium borohydride in tetrahydrofuran, the reaction proceeds under atmospheric reflux conditions, significantly reducing operational risks and infrastructure costs associated with high-pressure vessels.
Q: What purity levels can be achieved with this 3-step synthetic route?
A: Experimental data from the patent indicates that liquid phase purity consistently exceeds 99.0% across various optimization parameters. The simplified post-treatment process, which avoids complex chromatographic separations often required in longer synthetic routes, contributes to this high level of chemical purity suitable for reference standard production.
Q: Is this process scalable for industrial production of Venlafaxine intermediates?
A: Yes, the process is explicitly designed for industrial scalability. It utilizes common solvents like ethanol and ethyl acetate, avoids expensive transition metal catalysts like Palladium on Carbon which require rigorous removal steps, and generates less waste liquid, making it economically and environmentally viable for large-scale manufacturing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Venlafaxine Impurity E Supplier
At NINGBO INNO PHARMCHEM, we recognize that the integrity of your analytical data depends entirely on the quality of your reference standards. Our technical team has thoroughly analyzed the synthetic route disclosed in CN111675671A and confirmed its viability for robust, large-scale production. We possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that we can meet your volume requirements without compromising on quality. Our facilities are equipped with rigorous QC labs capable of verifying the stringent purity specifications required for Venlafaxine Impurity E, utilizing advanced chromatographic techniques to guarantee that every batch exceeds the 99% purity threshold. We understand the critical nature of impurity profiling in regulatory submissions and are committed to providing materials that stand up to the most demanding audits.
We invite you to collaborate with us to optimize your supply chain for Venlafaxine intermediates. Our team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume needs, demonstrating how our streamlined process can reduce your overall procurement costs. Please contact our technical procurement team today to request specific COA data and discuss route feasibility assessments for your upcoming projects. Let us be your partner in achieving excellence in pharmaceutical quality control.
