Revolutionizing Voriconazole Intermediate Production via Safe Halogen Exchange Technology
Revolutionizing Voriconazole Intermediate Production via Safe Halogen Exchange Technology
The global demand for second-generation triazole antifungal agents, particularly Voriconazole, has necessitated a rigorous re-evaluation of supply chain strategies for key intermediates. Patent CN103664584A introduces a transformative methodology for the preparation of alpha-fluoro-propionyl acetate, a critical building block in the synthesis of 6-ethyl-5-fluoro-4-hydroxy pyrimidine. This technical breakthrough addresses the longstanding industry pain points associated with hazardous reagents and complex multi-step sequences. By shifting from dangerous elemental fluorine or hydrofluoric acid protocols to a benign halogen exchange mechanism utilizing inorganic fluorides, this innovation offers a robust pathway for reliable pharmaceutical intermediate supplier networks seeking to enhance process safety without compromising yield.
The strategic value of this patent lies in its dual-step approach: initial chlorination followed by nucleophilic fluorination mediated by polyethylene glycol. This sequence effectively bypasses the equipment corrosion and toxicity issues that have historically plagued the manufacturing of fluorinated esters. For R&D directors and procurement managers alike, the transition to this methodology represents a significant opportunity to stabilize supply chains and reduce the total cost of ownership for antifungal API production. The following analysis dissects the mechanistic advantages and commercial implications of adopting this superior synthetic route.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historical approaches to synthesizing alpha-fluoro-propionyl acetate have been fraught with significant technical and logistical hurdles that impede efficient commercial scale-up of complex fluorinated compounds. One prominent prior art method, disclosed in Chinese patent application CN200310108345, relies on 2,3,3,3-tetrafluoro methyl propionate as a starting material. This route is inherently flawed due to the scarcity and high cost of the tetrafluoro precursor, coupled with a lengthy sequence involving alcoholysis, saponification, and condensation steps that cumulatively erode overall yield. Furthermore, the acylation reactions employed in this legacy process utilize propionyl chloride, which is known to cause severe equipment damage through corrosion, thereby increasing capital expenditure and maintenance downtime.
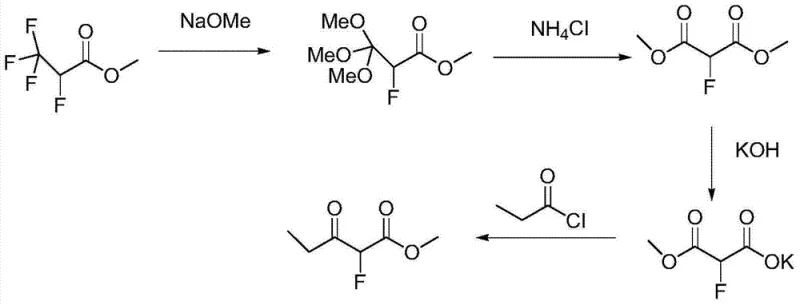
Another conventional pathway, described in CN200910224742, attempts to simplify the route via direct acylation of fluoro ethyl acetate with propionyl chloride. While conceptually shorter, this method suffers from abysmal reaction yields, reported to be as low as 30%, rendering it economically unviable for large-scale manufacturing. Additionally, the decomposition of propionyl chloride during the process generates corrosive gases, posing environmental and safety challenges. A third approach involves the direct fluorination of propionyl acetic ester using elemental fluorine or hydrogen fluoride, as seen in CN201010118357. This method is particularly dangerous, requiring specialized containment for hypertoxic and corrosive reagents, and often results in difficult-to-separate poly-fluorinated byproducts that compromise product purity.
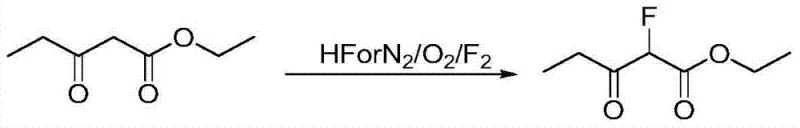
The Novel Approach
In stark contrast to these hazardous and inefficient legacy methods, the novel approach detailed in CN103664584A utilizes a pragmatic two-step sequence starting from readily available propionyl acetic ester. The first step involves a controlled chlorination using sulfuryl chloride to generate alpha-chloro-propionyl acetic ester, a stable and manageable intermediate. The second, and most critical step, employs a nucleophilic substitution where the chlorine atom is displaced by a fluoride ion derived from inexpensive inorganic salts like potassium fluoride or sodium fluoride. This reaction is facilitated by a polar aprotic solvent and, crucially, polyethylene glycol, which acts as a phase transfer catalyst to drive the reaction to completion under mild thermal conditions.
This innovative strategy eliminates the need for exotic starting materials or dangerous gaseous reagents, fundamentally altering the risk profile of the manufacturing process. By leveraging the high nucleophilicity of fluoride ions in the presence of polyethylene glycol, the method achieves high conversion rates while minimizing the formation of poly-fluorinated impurities. The result is a streamlined process that is not only safer for operators but also significantly more amenable to continuous improvement and optimization in an industrial setting, providing a clear competitive advantage for manufacturers adopting this technology.
Mechanistic Insights into Polyethylene Glycol-Mediated Halogen Exchange
The core of this technological advancement lies in the sophisticated interplay between the inorganic fluoride source and the polyethylene glycol additive within a polar aprotic medium. In traditional nucleophilic substitutions involving fluoride, the strong lattice energy of salts like KF often renders the fluoride ion insufficiently reactive in organic solvents. However, the addition of polyethylene glycol, specifically optimized as PEG-800 in this patent, creates a coordination complex with the potassium cation. This complexation effectively 'naked' the fluoride anion, drastically increasing its solubility and nucleophilic strength within solvents such as DMF or N,N-dimethylacetamide. This mechanism allows the substitution reaction to proceed efficiently at temperatures between 90°C and 160°C, avoiding the extreme conditions required by other fluorination techniques.
From an impurity control perspective, this mechanism offers superior selectivity compared to electrophilic fluorination methods. The use of stoichiometric inorganic fluoride limits the potential for over-fluorination, a common issue when using elemental fluorine gas which can lead to difluoro- or trifluoro- byproducts that are chemically similar and difficult to separate via distillation. Furthermore, the mild nature of the reaction conditions preserves the integrity of the ester functionality, preventing hydrolysis or decomposition that might occur under the harsh acidic conditions of HF-mediated processes. This high level of chemoselectivity ensures that the final crude product requires less intensive purification, directly translating to higher recovery rates and reduced solvent consumption during downstream processing.
How to Synthesize Alpha-Fluoro-Propionyl Acetate Efficiently
The implementation of this synthesis route requires precise control over reaction parameters to maximize the benefits of the halogen exchange mechanism. The process begins with the preparation of the alpha-chloro intermediate, followed by the critical fluorination step where the choice of solvent and phase transfer catalyst loading determines the final yield. Operators must ensure strict moisture control during the fluorination stage, as water can compete with the fluoride nucleophile or hydrolyze the ester. The following guide outlines the standardized operational framework derived from the patent examples, ensuring reproducibility and safety.
- React propionyl acetate with sulfuryl chloride in a low-polarity solvent to generate alpha-chloro-propionyl acetate.
- Mix the chloro-intermediate with potassium fluoride or sodium fluoride in a polar aprotic solvent like DMF.
- Add polyethylene glycol (preferably PEG-800) as a phase transfer mediator and heat to 90-160°C to complete the substitution.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this patented methodology offers profound strategic benefits that extend beyond simple chemical yield. The shift away from specialized, hazardous reagents like elemental fluorine or tetrafluoro precursors towards commodity chemicals such as sulfuryl chloride and potassium fluoride dramatically simplifies the sourcing landscape. This transition mitigates the risk of supply disruptions caused by the limited availability of niche fluorinating agents, ensuring a more resilient and continuous supply of high-purity pharmaceutical intermediates. Furthermore, the elimination of highly corrosive reagents reduces the burden on facility maintenance and extends the lifespan of reactor vessels, contributing to long-term capital preservation.
- Cost Reduction in Manufacturing: The economic impact of this process is driven primarily by the substitution of expensive and dangerous fluorinating agents with low-cost inorganic salts. By utilizing potassium fluoride or sodium fluoride, manufacturers can achieve substantial cost savings on raw material procurement. Additionally, the inclusion of polyethylene glycol optimizes the efficiency of the fluoride usage, meaning less reagent is wasted in side reactions or remains unreacted in the waste stream. This material efficiency, combined with the simplified purification requirements due to fewer byproducts, leads to a significantly lower cost of goods sold (COGS) for the final intermediate.
- Enhanced Supply Chain Reliability: The reliance on widely available commodity chemicals ensures that production schedules are not held hostage by the lead times of specialized reagents. Propionyl acetate and sulfuryl chloride are produced at massive scales globally, providing a buffer against market volatility. This stability allows for more accurate forecasting and inventory planning, reducing the need for safety stock and freeing up working capital. The robustness of the process also means that production can be easily transferred between different manufacturing sites without requiring specialized infrastructure, further de-risking the supply chain.
- Scalability and Environmental Compliance: From an environmental and regulatory standpoint, this process offers a cleaner alternative to traditional fluorination. The absence of toxic gas emissions and the use of less hazardous waste streams simplify compliance with increasingly stringent environmental regulations. The mild reaction conditions also reduce energy consumption compared to high-pressure or cryogenic alternatives. These factors collectively facilitate easier permitting for new production lines and support corporate sustainability goals, making the technology attractive for long-term investment and commercial scale-up of complex fluorinated compounds.
Frequently Asked Questions (FAQ)
The following questions address common technical and operational inquiries regarding the implementation of this synthesis method. These insights are derived directly from the experimental data and comparative analysis provided in the patent documentation, offering clarity on safety, scalability, and quality control measures.
Q: Why is the new fluorination method safer than traditional HF-based routes?
A: Traditional methods often utilize hazardous hydrogen fluoride (HF) or elemental fluorine gas, which require specialized corrosion-resistant equipment and pose severe safety risks. The patented method replaces these with solid inorganic fluorides like potassium fluoride, eliminating the need for handling toxic gases and reducing equipment maintenance costs.
Q: What role does polyethylene glycol play in this reaction?
A: Polyethylene glycol acts as a phase transfer catalyst that complexes with the metal cation of the fluoride salt. This enhances the solubility and nucleophilicity of the fluoride ion in the organic polar aprotic solvent, significantly improving reaction yield and allowing for milder reaction conditions.
Q: Can this process be scaled for industrial manufacturing?
A: Yes, the process is designed for industrial suitability. It uses readily available commodity chemicals like propionyl acetate and sulfuryl chloride, avoids extreme pressures or cryogenic temperatures, and utilizes standard stainless steel reactors rather than specialized Hastelloy equipment required for HF processing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Alpha-Fluoro-Propionyl Acetate Supplier
At NINGBO INNO PHARMCHEM, we recognize that the successful commercialization of advanced antifungal therapies depends on the consistent availability of high-quality intermediates. Our technical team has extensively analyzed the pathway described in CN103664584A and possesses the expertise to implement this safe, high-yield protocol effectively. We bring extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your supply needs are met with precision. Our facilities are equipped with rigorous QC labs capable of verifying stringent purity specifications, guaranteeing that every batch of alpha-fluoro-propionyl acetate meets the exacting standards required for API synthesis.
We invite you to collaborate with us to leverage this superior technology for your Voriconazole supply chain. By partnering with our technical procurement team, you can access a Customized Cost-Saving Analysis tailored to your specific volume requirements. We encourage you to contact us today to request specific COA data and route feasibility assessments, allowing us to demonstrate how our optimized manufacturing capabilities can drive value and security for your organization.
