Advanced Synthesis of Fluorinated Azacyclo-Alkyne Liquid Crystal Compounds for Next-Gen Displays
Advanced Synthesis of Fluorinated Azacyclo-Alkyne Liquid Crystal Compounds for Next-Gen Displays
The rapid evolution of flat-panel display technologies, particularly Thin-Film Transistor Liquid Crystal Displays (TFT-LCD) and Polymer Dispersed Liquid Crystal (PDLC) systems, demands materials with increasingly sophisticated electro-optical properties. Patent CN103524394A introduces a groundbreaking class of fluorine-containing azacyclo-alkyne liquid crystal compounds that address the critical industry need for high optical anisotropy (Δn) combined with low viscosity and high resistivity. These materials are engineered to enhance response speeds and broaden the operating temperature range of modern display panels. The core innovation lies in the strategic incorporation of an alkyne bond between fluorinated heterocyclic rings and phenyl groups, a structural motif that significantly improves refractive index without compromising the fluidity required for fast switching. As a leading reliable electronic chemical supplier, understanding the nuances of such patented synthetic routes is essential for securing a competitive edge in the supply of high-performance display materials.
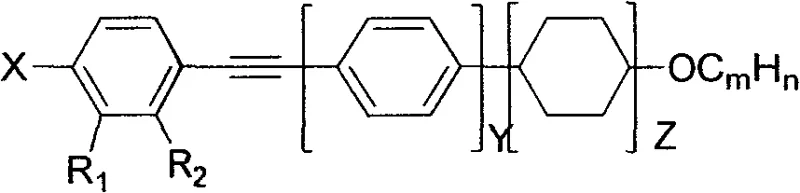
The structural versatility of these compounds allows for precise tuning of physical parameters. By modifying the terminal alkyl chain length (represented by CmHn) and the degree of fluorination on the nitrogen-containing heterocycle, manufacturers can tailor the clearing point and dielectric anisotropy to specific panel requirements. This level of customization is vital for R&D teams aiming to optimize mixture formulations for next-generation screens. The patent outlines two primary structural variants, Formula A and Formula B, which differ primarily in the linkage group connecting the core to the terminal tail, offering a broad portfolio of potential candidates for diverse application scenarios in the optoelectronics sector.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional synthesis pathways for fluorinated liquid crystal intermediates often suffer from significant bottlenecks that hinder cost reduction in display material manufacturing. Conventional routes frequently rely on multi-step halogenation and coupling sequences that require expensive palladium catalysts in large excesses, difficult-to-remove heavy metal residues, and harsh reaction conditions that degrade sensitive fluorinated moieties. Furthermore, older methods for constructing the nitrogen-containing heterocyclic core often involve low-yielding cyclization steps or require protected intermediates that add unnecessary synthetic burden. These inefficiencies result in prolonged production cycles, increased waste generation, and ultimately, higher costs per kilogram of the final active pharmaceutical ingredient (API) or electronic chemical. For procurement managers, these legacy processes translate into volatile pricing and extended lead times that disrupt supply chain stability.
The Novel Approach
The methodology described in CN103524394A represents a paradigm shift towards efficiency and scalability. By utilizing 2,2,3,3-tetrafluoro-1,4-butanediol as a foundational building block, the process bypasses many of the tedious fluorination steps typically required. The novel route employs a streamlined sequence involving triflation, nucleophilic substitution, and a highly efficient dehydrofluorination step to construct the fluorinated pyrrole or pyrrolidine ring system. This approach not only simplifies the overall synthetic tree but also enhances the overall yield by minimizing side reactions associated with unstable intermediates. The use of polyethylene glycol (PEG-600) as a phase transfer medium in the coupling stage further exemplifies the green chemistry principles embedded in this design, facilitating easier workup and purification. This technological leap enables the commercial scale-up of complex fluorinated compounds with greater economic viability and environmental compliance.
Mechanistic Insights into Sonogashira Coupling and Heterocycle Formation
The heart of this synthetic strategy lies in the robust construction of the carbon-carbon triple bond linking the aromatic cores, achieved through a modified Sonogashira coupling reaction. In this specific embodiment, the reaction utilizes a copper iodide (CuI) and triphenylphosphine (PPh3) catalytic system in the presence of potassium carbonate and PEG-600. The mechanism involves the oxidative addition of the aryl iodide to the copper species, followed by transmetallation with the terminal alkyne and subsequent reductive elimination to form the alkyne bridge. The inclusion of PEG-600 is particularly noteworthy as it acts as a non-volatile, recyclable solvent system that stabilizes the catalytic species and enhances the solubility of the inorganic base, thereby driving the reaction to completion under relatively mild thermal conditions (60-150°C). This mechanistic optimization is crucial for maintaining the integrity of the sensitive fluorinated heterocycle during the bond-forming event.
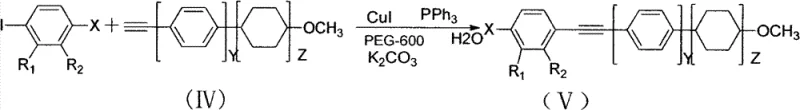
Impurity control is another critical aspect addressed by the detailed reaction conditions provided in the patent. The dehydrofluorination step, which converts the tetrafluoropyrrolidine precursor into the difluoropyrrole derivative using sodium tert-butoxide in DMSO, is highly sensitive to moisture and temperature. The patent specifies rigorous drying protocols using anhydrous magnesium sulfate and precise temperature controls (90-100°C) to prevent the formation of polymeric byproducts or incomplete elimination species. Post-reaction purification via silica gel column chromatography with specific eluent ratios (e.g., petroleum ether to ethyl acetate at 50:1) ensures that trace metal catalysts and unreacted starting materials are removed to levels acceptable for electronic grade applications. This attention to detail in the purification protocol guarantees the high-purity liquid crystal intermediates necessary for preventing image sticking or voltage holding ratio degradation in the final display device.
How to Synthesize Fluorinated Azacyclo-Alkyne Liquid Crystal Compounds Efficiently
Implementing this synthesis requires strict adherence to the stoichiometric ratios and environmental controls outlined in the patent examples. The process begins with the activation of the diol backbone followed by ring closure, setting the stage for the final coupling. Operators must ensure that all glassware is flame-dried and that inert atmosphere techniques are employed during the copper-catalyzed steps to prevent oxidation of the phosphine ligands. The following guide summarizes the critical operational phases derived from the experimental data, providing a roadmap for laboratory validation and pilot plant trials. Detailed standardized synthesis steps are provided in the guide below to ensure reproducibility and safety during handling of reactive fluorinated species.
- React 2,2,3,3-tetrafluoro-1,4-butanediol with trifluoromethanesulfonic anhydride to form the bis-triflate intermediate.
- Perform nucleophilic substitution with iodoaniline followed by dehydrofluorination using sodium tert-butoxide to form the fluorinated pyrrole or pyrrolidine core.
- Execute a copper-catalyzed Sonogashira coupling reaction with terminal alkynes in PEG-600/water system to finalize the liquid crystal structure.
Commercial Advantages for Procurement and Supply Chain Teams
For stakeholders focused on the bottom line and operational continuity, the adoption of this synthetic route offers tangible benefits beyond mere technical performance. The shift towards a more convergent synthesis strategy significantly reduces the number of unit operations required to reach the final target molecule. By consolidating steps and utilizing robust catalytic systems, the overall process mass intensity (PMI) is improved, leading to substantial cost savings in raw material consumption and waste disposal. This efficiency directly impacts the total cost of ownership for display manufacturers seeking to optimize their bill of materials without sacrificing quality. Furthermore, the reliance on commodity chemicals like 2,2,3,3-tetrafluoro-1,4-butanediol mitigates the risk of supply shortages associated with exotic or proprietary reagents.
- Cost Reduction in Manufacturing: The elimination of expensive noble metal catalysts in favor of more economical copper-based systems, combined with the high yields reported in the patent examples (often exceeding 90% in coupling steps), drives down the variable cost per unit. The simplified purification workflow reduces solvent usage and energy consumption associated with distillation and recrystallization, contributing to a leaner manufacturing footprint. These factors collectively enable a more competitive pricing structure for the final liquid crystal mixture, allowing panel makers to maintain margins in a price-sensitive market.
- Enhanced Supply Chain Reliability: The use of stable, commercially available starting materials ensures that production schedules are not held hostage by the lead times of custom-synthesized intermediates. The robustness of the reaction conditions, which tolerate a range of temperatures and concentrations without significant yield loss, provides a buffer against minor process variations that might otherwise cause batch failures. This resilience is key to reducing lead time for high-purity liquid crystal monomers, ensuring that Just-In-Time delivery models can be supported effectively even during periods of high demand.
- Scalability and Environmental Compliance: The process avoids the use of chlorinated solvents in the final coupling step, opting instead for greener alternatives like PEG-water mixtures, which aligns with increasingly stringent global environmental regulations. The scalability of the Sonogashira coupling under these conditions has been demonstrated in similar industrial contexts, suggesting a smooth path from gram-scale laboratory synthesis to ton-scale commercial production. This ease of scale-up minimizes the technical risk for contract development and manufacturing organizations (CDMOs) looking to bring new display materials to market rapidly.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production and application of these advanced liquid crystal materials. The answers are derived directly from the experimental data and claims within the patent documentation, providing clarity on the feasibility and performance characteristics of the technology. Understanding these details is crucial for technical procurement teams evaluating new suppliers or formulation partners.
Q: What are the key performance advantages of fluorinated azacyclo-alkyne liquid crystals?
A: These compounds exhibit large optical anisotropy (Δn), moderate dielectric anisotropy (Δε), low viscosity, and high clearing points, making them ideal for fast-response TFT-LCD and PDLC applications.
Q: How does the patented synthesis method improve production efficiency?
A: The method utilizes readily available 2,2,3,3-tetrafluoro-1,4-butanediol and employs a streamlined sequence of triflation, cyclization, and coupling reactions that avoid harsh conditions and simplify purification via standard column chromatography.
Q: Can the physical properties of these liquid crystals be tuned?
A: Yes, the mesomorphic phase transition temperature, viscosity, and dielectric constant can be precisely regulated by adjusting the length of the terminal alkyl chain and the fluorine content within the heterocyclic ring.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Fluorinated Azacyclo-Alkyne Liquid Crystal Compound Supplier
At NINGBO INNO PHARMCHEM, we recognize the pivotal role that high-performance intermediates play in the advancement of display technologies. Our team of expert chemists possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from patent to product is seamless and efficient. We are committed to delivering materials that meet stringent purity specifications, utilizing our rigorous QC labs to verify every batch against the highest industry standards. Our capability to handle complex fluorinated chemistries safely and effectively makes us an ideal partner for your liquid crystal supply needs.
We invite you to engage with our technical procurement team to discuss how we can support your specific formulation requirements. Whether you need a Customized Cost-Saving Analysis for your current supply chain or require specific COA data and route feasibility assessments for new targets, we are ready to provide the data-driven insights you need. Contact us today to explore how our manufacturing expertise can accelerate your product development timeline and enhance your competitive position in the global display market.
