Advanced Synthesis of Polyfluorinated Liquid Crystal Intermediates for High-Performance Display Applications
Advanced Synthesis of Polyfluorinated Liquid Crystal Intermediates for High-Performance Display Applications
The rapid evolution of Thin-Film Transistor Liquid Crystal Display (TFT-LCD) technology demands materials with increasingly sophisticated electro-optical properties. Patent CN102617303B introduces a groundbreaking class of polyfluorinated bis(phenylethynyl)benzene derivatives designed to address critical limitations in current display formulations. These compounds are engineered to provide large optical anisotropy (Δn) and suitable dielectric anisotropy (Δε), essential parameters for next-generation In-Plane Switching (IPS) and Vertical Alignment (VA) modes. By strategically incorporating fluorine atoms into the lateral and terminal positions of the phenylacetylene backbone, this invention achieves a superior balance of low rotational viscosity and high voltage holding ratio. For procurement and R&D teams seeking a reliable display material intermediate supplier, understanding the synthetic accessibility and performance metrics of these derivatives is paramount for securing long-term supply chain stability.
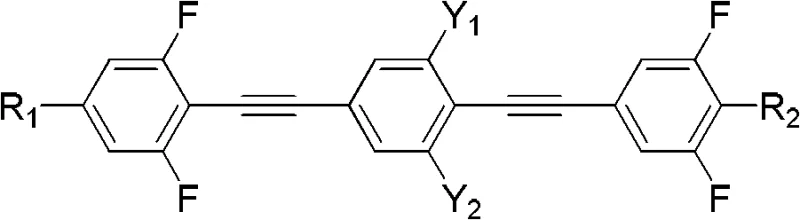
The core innovation lies in the molecular architecture, where the rigid bis(phenylethynyl)benzene core ensures high thermal stability and a wide nematic phase range. Unlike traditional cyano-based liquid crystals which suffer from ionic impurities and lower voltage retention, these fluorinated analogues offer enhanced resistivity and chemical inertness. The patent details a versatile general formula where R1 and R2 groups can be tuned from C1-C15 alkyl or alkoxy chains, allowing precise modulation of the clearing point and solubility characteristics. This tunability is crucial for formulators aiming to optimize the commercial scale-up of complex electronic chemicals without compromising on the purity standards required for high-end display panels.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the liquid crystal industry has relied heavily on cyano-substituted biphenyls and phenylcyclohexanes to achieve high dielectric anisotropy. However, these legacy materials present significant challenges for modern high-resolution displays. The presence of the cyano group often leads to the introduction of ionic impurities during synthesis or degradation over time, resulting in poor voltage holding ratios and image sticking issues. Furthermore, ester-based liquid crystals, while offering wide phase ranges, typically exhibit high rotational viscosity, which slows down the response time of the display pixels. These drawbacks necessitate the development of alternative structures that can maintain high performance while eliminating the inherent instability of polar cyano groups. The search for cost reduction in electronic chemical manufacturing has often been hindered by the complex purification steps required to remove these ionic contaminants from conventional materials.
The Novel Approach
The synthesis route disclosed in CN102617303B offers a robust alternative through a modular cross-coupling strategy. By utilizing palladium-catalyzed Sonogashira coupling reactions, the process constructs the conjugated carbon backbone with high efficiency and selectivity. This method avoids the harsh conditions often associated with older synthetic pathways, operating at moderate temperatures between 50°C and 110°C. The use of trimethylsilyl protecting groups allows for controlled stepwise assembly of the molecule, ensuring that the final product possesses the exact structural configuration required for optimal liquid crystalline behavior. This streamlined approach not only simplifies the production workflow but also enhances the overall yield and purity profile, making it an attractive option for reducing lead time for high-purity display intermediates.
Mechanistic Insights into Pd-Catalyzed Cross-Coupling
The heart of this synthesis is the iterative use of palladium-catalyzed cross-coupling, specifically the Sonogashira reaction, which links aryl halides with terminal alkynes. The mechanism involves the oxidative addition of the aryl halide (iodide or bromide) to the Pd(0) catalyst, followed by transmetallation with the copper-acetylide species generated in situ. This step is critical for forming the carbon-carbon triple bonds that define the rigid rod-like shape of the liquid crystal molecule. The patent specifies the use of tetrakis(triphenylphosphine)palladium as the catalyst, which provides excellent activity under the described conditions. The subsequent reductive elimination releases the coupled product and regenerates the active catalyst species. This catalytic cycle is highly efficient, requiring only 0.1% to 5% molar loading of the palladium complex, which is a significant factor in managing the cost of goods sold for large-scale production runs.
Impurity control is meticulously addressed through the choice of reagents and reaction conditions. The use of inert atmospheres, specifically high-purity argon, prevents oxidative degradation of the sensitive alkyne intermediates and the palladium catalyst. Furthermore, the deprotection steps, such as the removal of the trimethylsilyl group using potassium carbonate or the elimination of acetone from the propargylic alcohol precursor using sodium hydroxide, are designed to proceed cleanly without affecting the fluorine substituents. Fluorine atoms are known for their strong electron-withdrawing effects, which can sometimes deactivate aromatic rings towards coupling; however, the optimized conditions ensure complete conversion. This rigorous control over side reactions ensures that the final high-purity OLED material or liquid crystal monomer meets the stringent specifications required by panel manufacturers, minimizing the risk of display defects caused by trace impurities.
How to Synthesize Polyfluorinated Bis(phenylethynyl)benzene Efficiently
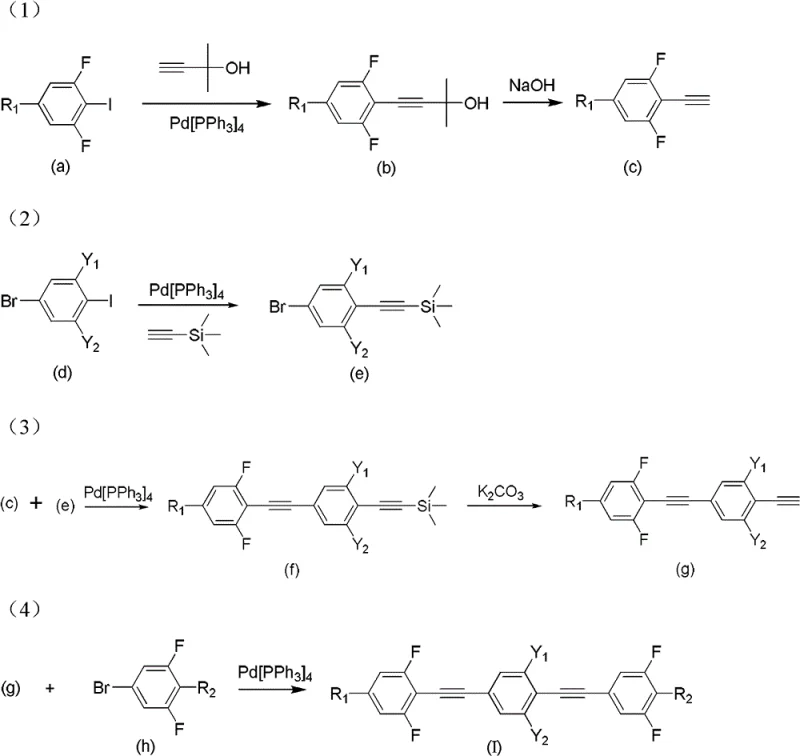
The preparation of these advanced derivatives follows a logical four-step sequence that maximizes atom economy and operational simplicity. The process begins with the functionalization of a fluorinated iodobenzene, followed by the construction of the central diarylacetylene unit, and concludes with the attachment of the terminal fluorinated ring. Each step utilizes standard organic synthesis techniques that are well-understood in the fine chemical industry, facilitating easy technology transfer from the laboratory to the pilot plant. The detailed reaction conditions, including specific molar ratios and solvent systems like tetrahydrofuran and triethylamine, are optimized to ensure reproducibility. For technical teams looking to implement this chemistry, the following guide outlines the standardized synthetic steps derived directly from the patent examples.
- Couple fluorine-substituted iodobenzene with 3-methyl-1-butyn-3-ol using Pd(PPh3)4, followed by NaOH-mediated deprotection to yield a terminal alkyne.
- Perform a Sonogashira coupling between a bromo-iodobenzene derivative and trimethylsilylacetylene to generate a protected alkyne intermediate.
- Cross-couple the terminal alkyne from step 1 with the protected alkyne from step 2, followed by K2CO3 desilylation to extend the conjugated system.
- Finalize the synthesis by coupling the extended alkyne with a fluorinated bromobenzene derivative under inert atmosphere to obtain the target bis(phenylethynyl)benzene.
Commercial Advantages for Procurement and Supply Chain Teams
From a supply chain perspective, the adoption of this synthesis route offers compelling advantages over traditional methods for producing high-performance liquid crystals. The primary benefit stems from the accessibility of the starting materials. The patent highlights that raw materials such as fluorinated iodobenzenes and bromobenzenes are commercially available and easy to obtain, reducing the risk of supply bottlenecks. This availability is crucial for maintaining continuous production schedules in a volatile global market. Additionally, the reaction conditions are relatively mild, avoiding the need for extreme cryogenic temperatures or high-pressure equipment, which translates to lower capital expenditure and operational costs for manufacturing facilities. These factors collectively contribute to a more resilient and cost-effective supply chain for critical display components.
- Cost Reduction in Manufacturing: The synthetic pathway eliminates the need for expensive and difficult-to-remove cyano groups, thereby simplifying the purification process. By using efficient palladium catalysis with low catalyst loading, the consumption of precious metals is minimized, directly impacting the variable cost of production. The high yields reported in the examples, often exceeding 70-80% for individual steps, mean less waste and lower raw material consumption per kilogram of final product. This efficiency drives significant cost savings without the need for complex process intensification technologies, making the economic case for switching to these fluorinated derivatives very strong for volume manufacturers.
- Enhanced Supply Chain Reliability: The modularity of the synthesis allows for the stocking of key intermediates, such as the protected alkyne derivatives, which can be rapidly converted to the final product based on demand fluctuations. This flexibility reduces the lead time required to fulfill large orders, a critical metric for procurement managers dealing with just-in-time manufacturing models. Furthermore, the robustness of the reaction against moisture and oxygen, when properly managed with inert gas, ensures consistent batch-to-batch quality. This reliability minimizes the risk of production delays caused by failed batches or off-spec material, ensuring a steady flow of materials to downstream formulators.
- Scalability and Environmental Compliance: The process is inherently scalable, as demonstrated by the use of common organic solvents like toluene and methanol which are easily recovered and recycled in an industrial setting. The absence of highly toxic reagents or hazardous byproducts simplifies waste treatment and aligns with increasingly strict environmental regulations. The ability to scale from gram-scale laboratory synthesis to multi-ton annual production is supported by the straightforward workup procedures, such as filtration and recrystallization, which do not require specialized chromatography on a large scale. This scalability ensures that the technology can meet the growing global demand for high-resolution displays sustainably.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production and application of these polyfluorinated derivatives. The answers are derived from the specific experimental data and beneficial effects described in the patent documentation. Understanding these details helps stakeholders make informed decisions about integrating these materials into their product portfolios. The focus is on practical implementation, performance expectations, and the strategic value of this chemical platform.
Q: What are the key physical properties of these polyfluorinated derivatives?
A: These compounds exhibit large optical anisotropy (Δn), suitable dielectric anisotropy (Δε), high clearing points, and low rotational viscosity, making them ideal for TFT-LCD applications.
Q: How does the fluorine substitution impact the manufacturing process?
A: Fluorine substitution lowers the melting point and viscosity of the liquid crystal mixture while enhancing chemical stability, and the synthesis utilizes readily available raw materials via efficient cross-coupling.
Q: Is this synthesis route scalable for industrial production?
A: Yes, the patent explicitly states the route is simple, uses accessible raw materials, and operates under moderate temperatures (50-110°C), making it highly suitable for large-scale industrial manufacturing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Polyfluorinated Bis(phenylethynyl)benzene Derivative Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-performance intermediates play in the advancement of display technology. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your supply needs are met with precision and consistency. We are committed to delivering products that adhere to stringent purity specifications, utilizing our rigorous QC labs to verify every batch against the highest industry standards. Our expertise in palladium-catalyzed couplings and fluorine chemistry positions us as an ideal partner for bringing these advanced liquid crystal materials from patent to market.
We invite you to collaborate with us to explore the full potential of this technology for your specific applications. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your volume requirements. We are ready to provide specific COA data and route feasibility assessments to demonstrate how our manufacturing capabilities can support your growth in the competitive electronic materials sector. Let us help you secure a stable supply of next-generation liquid crystal intermediates.
