Advanced Titanium-Catalyzed Asymmetric Oxidation for High-Purity Chiral Sulphoxides
Advanced Titanium-Catalyzed Asymmetric Oxidation for High-Purity Chiral Sulphoxides
The pharmaceutical industry's relentless pursuit of single-enantiomer drugs has necessitated a paradigm shift in synthetic methodology, moving away from wasteful resolution techniques toward efficient asymmetric catalysis. Patent CN102241670A discloses a groundbreaking preparation method for high-purity chiral sulphoxide compounds, addressing critical bottlenecks in the manufacturing of proton pump inhibitors (PPIs). This technology leverages a novel chiral titanium complex catalyst system to achieve direct asymmetric oxidation of thioethers, delivering exceptional enantiomeric excess and chemical purity. For R&D directors and procurement strategists, this represents a significant opportunity to optimize the production of key API intermediates like Esomeprazole and S-rabeprazole. The process is characterized by its operational simplicity, high raw material utilization, and robust scalability, making it an ideal candidate for commercial adoption in the competitive generic and specialty pharma markets.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditionally, the industrial production of chiral sulphoxides has relied heavily on chiral resolution or the use of stoichiometric chiral auxiliaries, both of which suffer from inherent inefficiencies. Chiral resolution, while established, is fundamentally limited by a maximum theoretical yield of 50%, requiring the disposal or recycling of the unwanted enantiomer, which drastically inflates production costs and environmental waste. Furthermore, methods employing chiral reagents often involve expensive, non-recyclable reagents and generate substantial amounts of byproduct waste, complicating downstream purification and increasing the burden on waste treatment facilities. These legacy processes often struggle to consistently achieve the stringent optical purity requirements demanded by modern regulatory agencies without multiple recrystallization steps, leading to extended cycle times and reduced overall throughput.
The Novel Approach
In stark contrast, the method described in CN102241670A utilizes a catalytic asymmetric oxidation strategy that fundamentally alters the economic and technical landscape of chiral sulphoxide synthesis. By employing a chiral titanium complex generated in situ from readily available alkoxytitanium compounds and amino indanol ligands, the process achieves direct stereoselective oxidation with high atom economy. This approach eliminates the 50% yield ceiling of resolution methods, allowing for theoretical yields approaching 100% relative to the starting thioether. The reaction conditions are mild and controllable, typically operating between -10°C and 50°C, which reduces energy consumption and enhances safety profiles compared to harsh oxidative conditions used in older methodologies. 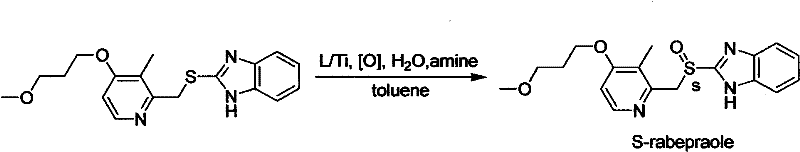
Mechanistic Insights into Titanium-Catalyzed Asymmetric Oxidation
The core of this technological breakthrough lies in the precise construction of the chiral catalyst species, which dictates the stereochemical outcome of the oxidation. The process begins with the formation of a chiral titanium complex by reacting an alkoxytitanium compound, such as titanium tetraisopropylate, with a chiral ligand, specifically a derivative of (1R, 2S)-1-amino-2-indanol. 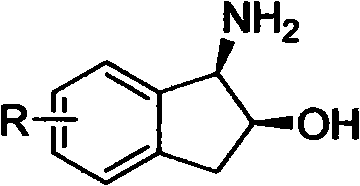 The molar ratio of the titanium source to the ligand is critically maintained between 1:1.5 and 1:3 to ensure complete coordination and the formation of the active catalytic species. The addition of a controlled amount of pure water (0.1%-1%) acts as a modifier, likely facilitating the generation of the active oxo-titanium species responsible for oxygen transfer. This catalyst system creates a highly defined chiral pocket that effectively differentiates the prochiral faces of the sulphur atom in the thioether substrate.
The molar ratio of the titanium source to the ligand is critically maintained between 1:1.5 and 1:3 to ensure complete coordination and the formation of the active catalytic species. The addition of a controlled amount of pure water (0.1%-1%) acts as a modifier, likely facilitating the generation of the active oxo-titanium species responsible for oxygen transfer. This catalyst system creates a highly defined chiral pocket that effectively differentiates the prochiral faces of the sulphur atom in the thioether substrate.
During the oxidation phase, the thioether substrate coordinates to the titanium center, and the oxidant, such as tert-butyl peroxide or cumene hydroperoxide, delivers an oxygen atom in a stereospecific manner. The presence of an amine base, such as triethylamine or N,N-diisopropylethylamine, is crucial for neutralizing acidic byproducts and maintaining the integrity of the catalyst system. This mechanistic pathway ensures that the resulting sulphoxide is formed with high enantiomeric excess, consistently achieving ee values greater than 97% as demonstrated in the patent examples for S-rabeprazole and Esomeprazole. The robustness of this mechanism allows for the accommodation of various electronic and steric environments on the pyridine and benzimidazole rings, making it a versatile platform for synthesizing a wide array of chiral sulphoxide pharmaceutical intermediates with minimal impurity formation.
How to Synthesize Chiral Sulphoxide Compounds Efficiently
The synthesis protocol outlined in the patent provides a clear, three-stage workflow designed for reproducibility and scale-up. The process initiates with the preparation of the chiral catalyst solution, followed by the addition of the thioether substrate and the controlled introduction of the oxidant. Temperature control is paramount during the oxidation step, where the reaction is initiated at low temperatures (-10°C to 10°C) to manage exothermicity and maximize stereoselectivity, before warming to 30°C to drive the reaction to completion.
- Prepare the chiral titanium catalyst complex by reacting an alkoxytitanium compound with a chiral amino indanol ligand in a nonpolar solvent with controlled water addition.
- Perform asymmetric oxidation by adding the corresponding thioether substrate and amine base to the catalyst solution, followed by the dropwise addition of an oxidant at controlled low temperatures.
- Purify the crude chiral sulphoxide product through neutralization, extraction, and distillation to achieve high chemical and optical purity.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this catalytic technology translates into tangible strategic advantages regarding cost structure and supply reliability. The shift from resolution to asymmetric catalysis inherently doubles the potential output from the same amount of starting thioether material, effectively reducing the raw material cost per kilogram of the final API intermediate. Furthermore, the use of common, commodity-grade solvents like toluene and ethyl acetate, along with commercially available oxidants and titanium sources, mitigates supply chain risks associated with specialized or scarce reagents. The simplified workup procedure, involving standard neutralization and extraction, reduces the complexity of manufacturing operations and lowers the barrier for technology transfer to large-scale production facilities.
- Cost Reduction in Manufacturing: The elimination of chiral resolution steps removes the need for expensive resolving agents and the associated loss of 50% of the material, leading to substantial cost savings in raw material procurement. Additionally, the catalytic nature of the titanium complex means that expensive chiral ligands are used in sub-stoichiometric amounts relative to the product, further driving down the cost of goods sold. The simplified purification process reduces solvent consumption and energy usage during distillation and crystallization, contributing to a leaner and more cost-effective manufacturing profile.
- Enhanced Supply Chain Reliability: By relying on robust, non-proprietary starting materials such as titanium alkoxides and common amines, the supply chain becomes less vulnerable to disruptions caused by single-source suppliers of exotic reagents. The high yield and purity achieved in a single pass reduce the need for reprocessing or recycling loops, ensuring faster turnaround times and more predictable delivery schedules for downstream API manufacturers. This reliability is critical for maintaining continuous production lines for high-demand medications like proton pump inhibitors.
- Scalability and Environmental Compliance: The process operates under mild conditions and utilizes solvents that are well-understood in terms of safety and environmental handling, facilitating easier regulatory approval for commercial scale-up. The high atom economy of the catalytic oxidation minimizes waste generation, aligning with green chemistry principles and reducing the costs associated with waste disposal and environmental compliance. This makes the technology not only economically attractive but also sustainable for long-term commercial operation.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this chiral sulphoxide synthesis technology. These insights are derived directly from the experimental data and technical specifications provided in the patent documentation, offering a transparent view of the process capabilities.
Q: What are the primary advantages of this asymmetric catalytic method over traditional chiral resolution?
A: This method offers superior atom economy and avoids the theoretical 50% yield limit inherent in chiral resolution processes. By utilizing a chiral titanium complex catalyst, the process directly synthesizes the desired enantiomer with high optical purity (ee value >97%), significantly reducing raw material waste and downstream separation costs.
Q: Which specific proton pump inhibitors can be synthesized using this technology?
A: The patented process is versatile and specifically demonstrated for the synthesis of key proton pump inhibitor intermediates, including S-rabeprazole, S-pantoprazole, and Esomeprazole. The method accommodates various substituents on the pyridine and benzimidazole rings, making it applicable to a broad range of chiral sulphoxide-based pharmaceuticals.
Q: How does the catalyst system ensure high enantiomeric excess?
A: The system utilizes a specific chiral ligand, (1R, 2S)-1-amino-2-indanol derivative, coordinated with an alkoxytitanium compound. This creates a rigid chiral environment around the titanium center that sterically directs the oxidation of the sulphur atom, ensuring the formation of the desired S-enantiomer with ee values consistently exceeding 97%.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Chiral Sulphoxide Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of advanced catalytic technologies like the one described in CN102241670A for the production of high-value pharmaceutical intermediates. As a premier CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that laboratory innovations are seamlessly translated into industrial reality. Our state-of-the-art facilities are equipped with rigorous QC labs capable of verifying stringent purity specifications, including chiral HPLC analysis to guarantee ee values exceeding 98% for critical intermediates like Esomeprazole and S-rabeprazole.
We invite global pharmaceutical partners to collaborate with us to leverage this efficient synthesis route for your supply chain needs. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We are prepared to provide specific COA data and comprehensive route feasibility assessments to demonstrate how our manufacturing capabilities can enhance your product quality while optimizing your overall production costs.
