Advanced Synthesis of 3-Alkoxymethyl Cephalosporin Intermediates for Commercial Scale Production
Advanced Synthesis of 3-Alkoxymethyl Cephalosporin Intermediates for Commercial Scale Production
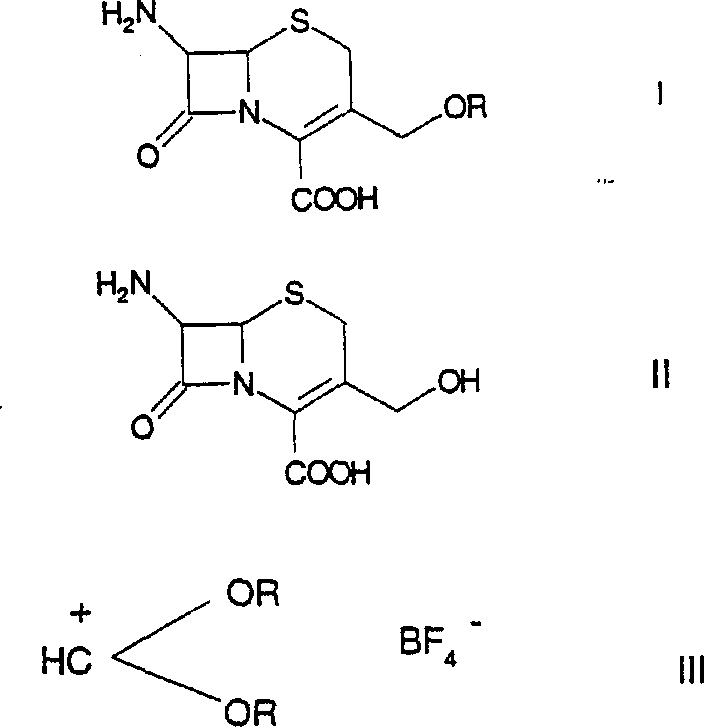
The pharmaceutical industry continuously seeks robust and scalable synthetic routes for critical antibiotic intermediates, particularly those required for third-generation cephalosporins. A pivotal advancement in this domain is documented in patent CN1050842C, which details a superior method for etherifying the 3-hydroxymethyl group of cephalosporins. This specific chemical transformation is essential for producing orally active cephalosporin prodrugs, such as cefpodoxime proxetil, where the lipophilicity of the molecule must be carefully tuned. The disclosed technology utilizes dioxycarbenium tetrafluoroborate reagents to achieve this modification, offering a distinct departure from the harsh and inefficient conditions characteristic of legacy manufacturing processes. By leveraging this innovative approach, manufacturers can overcome historical bottlenecks related to yield loss and impurity profiles, thereby securing a more reliable supply chain for high-value beta-lactam antibiotics.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the etherification of the 3-hydroxymethyl position in 7-aminocephalosporanic acid (7-ACA) derivatives has been plagued by significant technical inefficiencies and environmental concerns. Prior art methods, such as those described in EP0262744, relied heavily on the use of strong Lewis acids like antimony pentachloride (SbCl5), bismuth trichloride (BiCl3), or zinc chloride (ZnCl2) in large excesses. These traditional protocols not only suffered from disappointingly low yields but also generated substantial quantities of hazardous waste, creating a heavy burden on ecological compliance and waste treatment infrastructure. Furthermore, alternative approaches involving boron trifluoride (BF3) in sulfolane, as seen in AT384222, necessitated high reaction temperatures and massive excesses of reagents, often resulting in poor quality 7-amino-3-methoxymethyl-3-cephem-4-carboxylic acid (7-AMCA). Perhaps most critically, these acidic conditions frequently promoted the formation of stable lactone by-products, which are notoriously difficult to separate and significantly degrade the purity of the final intermediate, forcing manufacturers to implement costly and time-consuming purification steps.
The Novel Approach
In stark contrast to these cumbersome legacy techniques, the methodology outlined in CN1050842C introduces a highly efficient pathway using dioxycarbenium tetrafluoroborate species. This novel reagent system allows for the direct etherification of 3-hydroxymethyl cephalosporins under remarkably mild conditions, typically ranging from -40°C to +30°C. The reaction proceeds with high selectivity, effectively bypassing the formation of the problematic lactone impurities that plague acid-catalyzed routes. By employing reagents such as dimethoxycarbenium tetrafluoroborate or diethoxycarbenium tetrafluoroborate, the process achieves superior conversion rates while requiring only a slight stoichiometric excess of the boron-containing reagent. This shift represents a fundamental optimization of the synthetic route, transforming a historically low-yielding, waste-intensive step into a streamlined, high-efficiency operation suitable for modern GMP manufacturing environments.
Mechanistic Insights into Dioxycarbenium-Mediated Etherification
The core chemical innovation lies in the unique reactivity of the dioxycarbenium cation, which acts as a potent yet controlled electrophile towards the hydroxyl group at the 3-position of the cephem nucleus. Unlike traditional Lewis acids which activate the alcohol through coordination that can inadvertently trigger intramolecular cyclization to form lactones, the dioxycarbenium species facilitates a direct nucleophilic substitution mechanism. The oxygen atom of the 3-hydroxymethyl group attacks the electrophilic carbon of the dioxycarbenium ion, leading to the displacement of an alkoxy group and the formation of the desired ether linkage. This mechanism is inherently cleaner because it avoids the generation of highly acidic microenvironments within the reaction matrix that are responsible for degrading the sensitive beta-lactam ring or promoting side reactions. The stability of the tetrafluoroborate counter-ion further contributes to the reaction's controllability, allowing for precise management of the reaction kinetics even at larger scales.
Furthermore, the suppression of lactone formation is a critical mechanistic advantage that directly correlates to improved process economics. In conventional acid-catalyzed etherification, the carboxylic acid moiety at the 4-position can attack the activated 3-position, closing the ring to form a thermodynamically stable lactone. This side reaction is effectively inhibited in the patented process due to the specific nature of the dioxycarbenium intermediate and the mild reaction conditions employed. Experimental data from the patent indicates that lactone content in the final product can be maintained as low as 0.2% to 0.5%, compared to significantly higher levels in prior art methods. This high level of chemoselectivity ensures that the structural integrity of the cephem core is preserved, reducing the need for complex downstream purification and maximizing the recovery of the valuable antibiotic intermediate.
How to Synthesize 7-Amino-3-alkoxymethyl-3-cephem-4-carboxylic Acid Efficiently
The implementation of this synthesis route requires careful attention to solvent selection and temperature control to maximize the benefits of the dioxycarbenium chemistry. The process typically begins by suspending the 3-hydroxymethyl cephalosporin starting material in a solvent system comprising organic carboxylic acid esters, such as methyl formate or ethyl formate, often in combination with sulfolane to enhance solubility and reaction homogeneity. The reaction mixture is cooled to sub-ambient temperatures, typically around -20°C, before the introduction of the etherifying agent. Whether using a pre-isolated dioxycarbenium salt or generating it in situ from boron trifluoride and orthoformates, the addition must be controlled to manage thermal output. Following the reaction period, which may last several hours depending on the specific alkyl group being introduced, the product is isolated via precipitation into water or alcohol mixtures followed by pH adjustment. For a comprehensive breakdown of the standardized operating procedures and safety protocols required for this synthesis, please refer to the detailed guide below.
- Suspend the 3-hydroxymethyl cephalosporin starting material (Formula II) in a suitable solvent system such as methyl formate and sulfolane.
- Cool the suspension to a temperature range between -40°C and +30°C, preferably around -20°C, to control reaction exothermicity.
- Add the dioxycarbenium tetrafluoroborate reagent (Formula III) either as a pre-isolated solution or generated in situ, maintaining stoichiometric excess.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this patented etherification technology translates into tangible operational improvements and risk mitigation strategies. The shift away from excessive Lewis acid usage and toxic reagents like diazomethane significantly simplifies the regulatory compliance landscape, reducing the administrative burden associated with handling hazardous materials. Moreover, the drastic reduction in by-product formation means that manufacturing batches are more consistent, leading to fewer rejected lots and a more predictable inventory flow. This reliability is crucial for maintaining continuous supply lines for downstream API production, where interruptions can have cascading effects on global drug availability. The ability to operate at moderate temperatures also lowers the energy intensity of the process, contributing to long-term sustainability goals and reducing utility costs associated with extreme heating or cooling requirements.
- Cost Reduction in Manufacturing: The elimination of massive excesses of Lewis acids and the reduction in purification steps drive significant cost efficiencies. By minimizing the formation of lactone impurities, the process avoids the need for expensive chromatographic separations or multiple recrystallizations that are often required to meet pharmacopeial standards. The higher overall yield directly increases the throughput of existing reactor capacity, allowing manufacturers to produce more kilograms of intermediate per batch without capital expenditure on new equipment. Additionally, the reduced consumption of reagents lowers the raw material cost per kilogram of finished product, enhancing the overall margin profile for this critical intermediate.
- Enhanced Supply Chain Reliability: The use of more stable and manageable reagents improves the robustness of the supply chain. Traditional methods relying on unstable or highly toxic reagents often face sourcing bottlenecks or strict transportation regulations that can delay production schedules. The novel method utilizes reagents that are easier to handle and store, reducing the risk of supply disruptions due to regulatory changes or vendor limitations. Furthermore, the simplified work-up procedure, which involves straightforward precipitation and filtration, shortens the cycle time per batch, enabling faster turnaround times and more responsive fulfillment of customer orders.
- Scalability and Environmental Compliance: The process is inherently designed for scale-up, with reaction conditions that are easily transferable from pilot plant to commercial production volumes. The reduction in hazardous waste generation aligns with increasingly stringent environmental regulations, minimizing the costs associated with waste disposal and treatment. By avoiding the use of heavy metal catalysts and toxic methylating agents, the facility reduces its environmental footprint and mitigates the risk of regulatory penalties. This eco-friendly profile not only satisfies internal sustainability mandates but also appeals to global pharmaceutical partners who prioritize green chemistry principles in their supplier selection criteria.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this advanced cephalosporin synthesis technology. These insights are derived directly from the experimental data and claims presented in the patent literature, providing a factual basis for evaluating the process viability. Understanding these nuances is essential for technical teams assessing the feasibility of technology transfer and for commercial teams negotiating supply agreements based on quality and cost metrics.
Q: How does the dioxycarbenium tetrafluoroborate method improve upon traditional Lewis acid catalysis?
A: Traditional methods using Lewis acids like SbCl5 or excess BF3 often result in low yields and significant formation of unwanted lactone by-products. The novel method utilizing dioxycarbenium tetrafluoroborate operates under milder conditions and specifically suppresses lactonization, leading to significantly higher purity and yield of the desired 3-alkoxymethyl cephalosporin.
Q: What are the typical reaction conditions for this etherification process?
A: The reaction is typically conducted at low to moderate temperatures, ranging from -40°C to +30°C. Preferred solvents include esters of organic carboxylic acids like methyl formate, often mixed with sulfolane. The reagent is used in slight excess (1.2 to 5 equivalents) to ensure complete conversion without requiring the massive excesses seen in older protocols.
Q: Why is minimizing lactone impurity critical in cephalosporin intermediate production?
A: Lactone formation is a major side reaction that degrades product quality and complicates downstream purification. High levels of lactone impurities can reduce the overall yield of the final API and require expensive chromatographic separation steps. The patented method reduces lactone content to as low as 0.2%, streamlining the manufacturing process.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 7-Amino-3-alkoxymethyl-3-cephem-4-carboxylic Acid Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-quality intermediates play in the global antibiotic supply chain. Our technical team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory innovation to industrial reality is seamless and efficient. We are committed to delivering products that meet stringent purity specifications, leveraging our rigorous QC labs to verify that every batch conforms to the highest standards of chemical integrity. Our facility is equipped to handle complex heterocyclic chemistry with the precision required for beta-lactam manufacturing, guaranteeing consistency and reliability for our long-term partners.
We invite you to collaborate with us to optimize your cephalosporin supply chain through the adoption of these advanced synthetic methodologies. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements and quality targets. Please contact us today to request specific COA data and route feasibility assessments, and let us demonstrate how our expertise can drive value and efficiency in your manufacturing operations.
