Scalable Synthesis of Chiral Nebivolol Intermediates via Suzuki Cyclization for Global API Manufacturing
Scalable Synthesis of Chiral Nebivolol Intermediates via Suzuki Cyclization for Global API Manufacturing
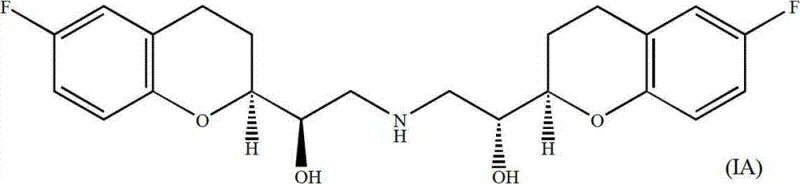
The pharmaceutical industry continuously seeks robust, scalable, and cost-effective routes for complex cardiovascular agents like Nebivolol, a third-generation beta-blocker known for its nitric oxide-mediated vasodilatory effects. Patent CN103087028A discloses a sophisticated synthetic strategy that addresses the inherent challenges of constructing the molecule's four chiral centers with high fidelity. Unlike traditional methods that often struggle with the separation of ten possible stereoisomers, this novel approach leverages a convergent synthesis starting from commercially accessible 2,2-dimethyl-1,3-dioxolane-4-carbaldehyde. By integrating vinyl Grignard addition, Mitsunobu etherification, and a pivotal intramolecular B-alkyl Suzuki coupling, the process establishes the critical chroman core with exceptional stereochemical control. This technical insight explores how this methodology transforms the manufacturing landscape for reliable pharmaceutical intermediate suppliers, offering a pathway to high-purity d-NBV and l-NBV enantiomers that meets the rigorous demands of modern regulatory standards.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of alpha,alpha'-[iminobis(methylene)]bis[6-fluoro-chroman-2-methanol] has been fraught with stereochemical complexity due to the presence of four unsymmetrical carbons, which theoretically generate up to sixteen stereoisomers. Prior art, such as European Patent EP 145067, described routes involving the reduction of chroman-2-carboxylic acid to aldehydes followed by conversion into epoxide mixtures. These conventional pathways often necessitated cumbersome column chromatography to separate racemic mixtures of (R,S) and (S,R) epoxides from (R,R) and (S,S) variants early in the synthesis. Such reliance on late-stage resolution not only depresses overall yield but also introduces significant operational costs and solvent waste, creating bottlenecks for cost reduction in pharmaceutical intermediate manufacturing. Furthermore, the handling of unstable aldehyde intermediates and the requirement for multiple protection-deprotection sequences in older routes often compromised the scalability required for commercial API production.
The Novel Approach
The methodology outlined in CN103087028A represents a paradigm shift by deferring the formation of the challenging chroman ring until after the stereochemistry of the side chain has been partially established. Instead of starting with the aromatic ring, the process begins with a chiral dioxolane aldehyde, reacting it with a vinyl Grignard reagent to install the allylic alcohol functionality with defined stereochemistry. This is followed by a Mitsunobu coupling with 2-bromo-4-fluorophenol, which efficiently links the aliphatic and aromatic domains. The true innovation lies in the subsequent hydroboration and palladium-catalyzed cyclization, which constructs the chroman ring intramolecularly. This sequence allows for the separation of diastereomers at a stage where they are chemically distinct enough for efficient purification, thereby streamlining the path to the final active enantiomers and significantly enhancing the feasibility of commercial scale-up of complex pharmaceutical intermediates.
Mechanistic Insights into B-Alkyl Suzuki Cyclization
The cornerstone of this synthetic route is the intramolecular B-alkyl Suzuki coupling, a transformation that elegantly closes the chroman ring while preserving the delicate stereochemical integrity established in previous steps. The mechanism initiates with the hydroboration of the terminal alkene in the aryl ether intermediate using reagents such as 9-BBN (9-borabicyclo[3.3.1]nonane) or disiamylborane. This step generates an organoborane species where the boron atom is positioned beta to the ether oxygen. Upon exposure to a palladium catalyst, typically Pd(0) complexes like Pd(dppf)Cl2 or Pd(PPh3)4 in the presence of a base such as potassium carbonate, oxidative addition occurs at the aryl bromide bond. The resulting organopalladium species then undergoes transmetallation with the organoborane, followed by reductive elimination to forge the carbon-carbon bond between the aromatic ring and the alkyl chain. This cyclization is highly diastereoselective, favoring the formation of the cis-fused chroman system required for biological activity.
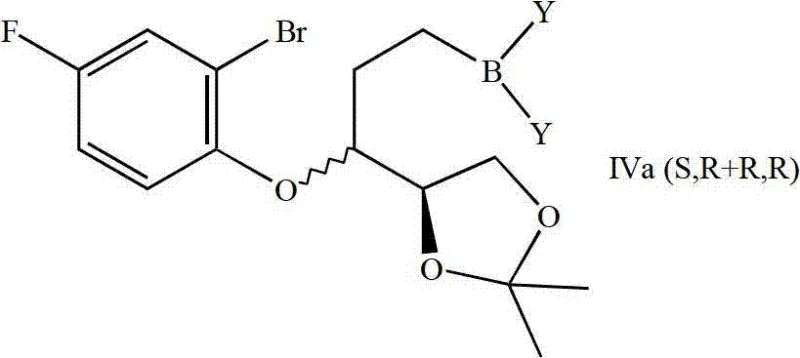
Impurity control in this mechanism is paramount, particularly regarding the suppression of protodeboronation and homocoupling side reactions. The patent specifies the use of phase-transfer catalysts like tetrabutylammonium chloride and carefully controlled temperatures ranging from 18°C to 200°C to optimize the catalytic cycle. By selecting specific ligands such as tri-o-tolylphosphine or tricyclohexylphosphine, the electronic environment of the palladium center is tuned to facilitate the difficult reductive elimination step from an sp3-hybridized carbon, which is traditionally slower than sp2-sp2 coupling. This precise tuning minimizes the formation of open-chain byproducts and ensures that the resulting chroman intermediate possesses the high optical purity necessary for downstream processing. The ability to separate the resulting diastereomeric mixture (S,R + R,R) via chromatography at this stage provides a critical checkpoint for quality assurance before proceeding to the final dimerization.
How to Synthesize Nebivolol Intermediates Efficiently
The synthesis of these high-value chiral building blocks requires a disciplined approach to reaction conditions and workup procedures to maintain stereochemical fidelity throughout the multi-step sequence. The process begins with the nucleophilic addition of vinyl magnesium bromide to the protected glyceraldehyde derivative at low temperatures (-20°C to 25°C) to prevent epimerization. Following the Mitsunobu etherification, which installs the fluoro-bromo-phenyl moiety, the crude product is subjected to hydroboration. The subsequent cyclization step is the most critical, requiring anhydrous conditions and degassed solvents to protect the palladium catalyst from oxidation. Detailed standardized operating procedures for each transformation, including specific quenching protocols and purification methods, are essential for reproducibility. For the complete step-by-step technical guide including reagent stoichiometry and safety data, please refer to the standardized protocol below.
- React 2,2-dimethyl-1,3-dioxolane-4-carbaldehyde with vinyl Grignard reagent to form the allylic alcohol intermediate.
- Perform Mitsunobu reaction with 2-bromo-4-fluorophenol to install the aryl ether linkage.
- Execute hydroboration followed by palladium-catalyzed intramolecular Suzuki coupling to cyclize the chroman ring.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this synthetic route offers tangible strategic benefits beyond mere technical elegance. By utilizing starting materials like 2,2-dimethyl-1,3-dioxolane-4-carbaldehyde and vinyl Grignard reagents, which are commodity chemicals available from multiple global vendors, the process mitigates the risk of single-source supply chain disruptions. The avoidance of exotic chiral catalysts in the early stages reduces raw material costs and simplifies the sourcing logistics. Furthermore, the convergence of the synthesis allows for the parallel production of different stereoisomeric intermediates, providing flexibility in manufacturing planning. This modularity ensures that production schedules can be adjusted dynamically to meet fluctuating market demands for either d-NBV or l-NBV without requiring a complete overhaul of the production line, thereby enhancing overall supply chain resilience.
- Cost Reduction in Manufacturing: The economic viability of this process is driven by the elimination of expensive resolution steps early in the synthesis. Traditional routes often discard up to half of the material during initial racemic separations, whereas this method builds chirality from the start or separates diastereomers at a higher molecular weight stage where yield loss is less impactful. Additionally, the use of robust catalytic systems like the Suzuki coupling reduces the reliance on stoichiometric amounts of heavy metals, lowering both reagent costs and the expense associated with metal scavenging and waste disposal. The streamlined sequence also reduces the total number of unit operations, leading to lower labor and utility consumption per kilogram of finished intermediate.
- Enhanced Supply Chain Reliability: The reliance on well-established reaction types such as Grignard additions and Mitsunobu reactions ensures that the process can be transferred easily between different manufacturing sites without significant re-validation burdens. These reactions are widely understood in the fine chemical industry, meaning that contract manufacturing organizations (CMOs) can readily adopt the technology. The stability of the key intermediates, such as the protected chroman alcohols, allows for safer storage and transportation, reducing the need for just-in-time delivery of unstable precursors. This stability buffer provides procurement teams with greater negotiating power and the ability to build strategic inventory levels to hedge against market volatility.
- Scalability and Environmental Compliance: From an environmental and scaling perspective, the process is designed to minimize solvent usage and hazardous waste generation. The intramolecular nature of the cyclization step improves atom economy compared to intermolecular couplings that might produce oligomeric byproducts. The ability to perform the cyclization in mixed solvent systems, including water-miscible organic solvents, facilitates easier workup and solvent recovery. As regulatory pressure on pharmaceutical manufacturing emissions increases, adopting a route that inherently generates less waste and utilizes less toxic reagents positions the supply chain for long-term compliance with green chemistry initiatives and environmental protection agency standards.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this patented synthesis for Nebivolol intermediates. These answers are derived directly from the experimental data and claims within CN103087028A, providing a factual basis for decision-making. Understanding the nuances of stereochemical control and the specific advantages of the Suzuki cyclization step is crucial for R&D teams evaluating this technology for transfer. The responses below clarify the operational parameters and the strategic value proposition of this manufacturing route.
Q: What are the key advantages of the Suzuki cyclization route for Nebivolol?
A: This route utilizes commercially available starting materials and avoids early-stage resolution of racemic mixtures, allowing for better stereocontrol during the ring-closing step.
Q: How is stereochemistry controlled in this synthesis?
A: Stereochemistry is managed through the use of chiral starting aldehydes and diastereoselective hydroboration, followed by chromatographic separation of diastereomers prior to dimerization.
Q: Is this process suitable for large-scale commercial production?
A: Yes, the reliance on robust reactions like Grignard addition and Suzuki coupling, which are well-understood in process chemistry, makes this route highly amenable to scale-up from kilogram to metric ton quantities.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Nebivolol Intermediate Supplier
At NINGBO INNO PHARMCHEM, we recognize that the transition from patent literature to commercial reality requires more than just chemical knowledge; it demands deep process engineering expertise. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the intricate stereochemical controls described in CN103087028A are maintained at an industrial level. We operate stringent purity specifications and utilize rigorous QC labs equipped with chiral HPLC and NMR capabilities to verify the enantiomeric excess of every batch. Our commitment to quality ensures that the Nebivolol intermediates we supply meet the exacting standards required for final API synthesis, minimizing the risk of downstream failures.
We invite global pharmaceutical partners to collaborate with us to optimize this synthesis for your specific supply chain needs. By leveraging our process development capabilities, we can provide a Customized Cost-Saving Analysis tailored to your volume requirements, identifying further opportunities for efficiency gains. We encourage you to contact our technical procurement team to request specific COA data and route feasibility assessments. Together, we can secure a stable, high-quality supply of these critical cardiovascular intermediates, driving value and reliability in your drug manufacturing operations.
