Industrial Scale-Up of N,N'-Dialkoxy-N,N'-Dialkyl Oxalamides via Safe Ester Aminolysis
Industrial Scale-Up of N,N'-Dialkoxy-N,N'-Dialkyl Oxalamides via Safe Ester Aminolysis
The pharmaceutical and agrochemical industries are constantly seeking robust, scalable synthetic routes for complex intermediates that balance high purity with operational safety. A pivotal advancement in this domain is detailed in Chinese Patent CN100386310C, which discloses a novel preparation method for N,N'-dialkoxy-N,N'-dialkyl oxalamides. These compounds serve as critical building blocks, often functioning similarly to Weinreb amides or serving as precursors for heterocyclic scaffolds in drug discovery. The patent outlines a transformative shift from hazardous acid chloride chemistry to a benign ester aminolysis protocol. By leveraging readily available oxalic acid diesters and N-alkyl-O-alkylhydroxylamines in the presence of a base, this methodology achieves exceptional conversion rates while completely circumventing the generation of toxic byproducts. For R&D directors and procurement specialists alike, this represents a significant opportunity to optimize supply chains for high-purity pharmaceutical intermediates through a greener, more cost-effective manufacturing paradigm.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of N,N'-dialkoxy-N,N'-dialkyl oxalamides relied heavily on the reaction between oxalyl chloride and N,O-disubstituted hydroxylamines, typically conducted in the presence of pyridine. While chemically effective on a small laboratory scale, this conventional approach presents severe liabilities for industrial application. The primary concern is the inherent instability of oxalyl chloride, which poses a significant safety hazard due to its tendency to decompose into highly toxic phosgene gas and carbon monoxide upon exposure to moisture or heat. Furthermore, the workup and purification stages of this traditional method necessitate the use of methylene chloride (dichloromethane), a solvent classified as a probable human carcinogen. The regulatory burden associated with handling, storing, and disposing of chlorinated solvents has increased dramatically, driving up operational costs and complicating environmental compliance. Consequently, relying on this legacy chemistry creates substantial bottlenecks for the commercial scale-up of complex pharmaceutical intermediates, rendering it unsuitable for modern, sustainable manufacturing facilities.
The Novel Approach
In stark contrast, the innovative process described in Patent CN100386310C utilizes oxalic acid diesters as the electrophilic starting material, reacting them with N-alkyl-O-alkylhydroxylamines or their acid salts under basic conditions. This strategic substitution of reagents fundamentally alters the safety profile and economic viability of the synthesis. By replacing the volatile and toxic oxalyl chloride with stable oxalic acid diesters, the risk of phosgene generation is entirely eliminated. Moreover, the reaction can be performed in a wide variety of solvents, including alcohols like methanol and ethanol, which are far less toxic and easier to recover than chlorinated alternatives. The patent data indicates that this method not only simplifies the operational workflow by removing the need for complex safety containment systems but also delivers superior yields. For a reliable agrochemical intermediate supplier or pharma partner, adopting this novel approach translates directly into reduced liability, lower waste treatment costs, and a more resilient production schedule capable of meeting stringent global quality standards.
Mechanistic Insights into Base-Catalyzed Ester Aminolysis
The core of this synthetic breakthrough lies in the nucleophilic acyl substitution mechanism facilitated by the presence of a strong base. The reaction initiates with the deprotonation of the N-alkyl-O-alkylhydroxylamine salt (Formula 2) by an alkali metal alkoxide, such as sodium methoxide or sodium ethoxide. This deprotonation generates the free amine species in situ, which acts as a potent nucleophile. This nucleophile then attacks one of the carbonyl carbons of the oxalic acid diester (Formula 1). 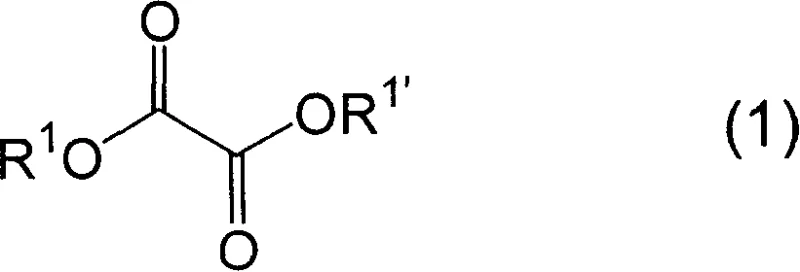 The tetrahedral intermediate formed during this attack subsequently collapses, expelling an alkoxide leaving group (RO-) and forming the first amide bond. Because the reaction is conducted with an excess of the hydroxylamine derivative (typically 1.5 to 3.0 molar equivalents relative to the diester), the process repeats at the second carbonyl center. This double addition results in the formation of the symmetric N,N'-dialkoxy-N,N'-dialkyl oxalamide structure (Formula 3).
The tetrahedral intermediate formed during this attack subsequently collapses, expelling an alkoxide leaving group (RO-) and forming the first amide bond. Because the reaction is conducted with an excess of the hydroxylamine derivative (typically 1.5 to 3.0 molar equivalents relative to the diester), the process repeats at the second carbonyl center. This double addition results in the formation of the symmetric N,N'-dialkoxy-N,N'-dialkyl oxalamide structure (Formula 3). 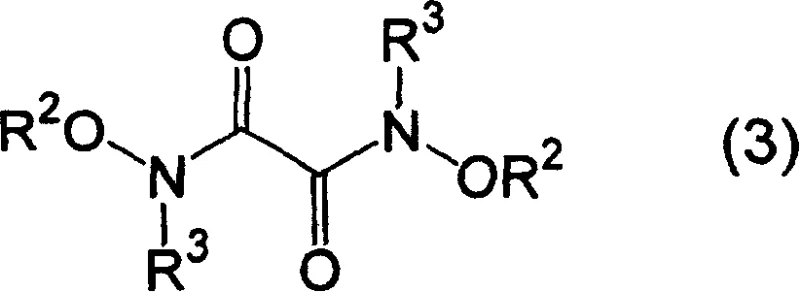 The choice of base is critical; alkali metal alkoxides are preferred because they match the solvent system (e.g., sodium methoxide in methanol), preventing transesterification side reactions that could occur with mismatched alkoxide/solvent pairs. This mechanistic precision ensures high selectivity and minimizes the formation of mono-substituted impurities, which is crucial for maintaining the purity specifications required for API intermediates.
The choice of base is critical; alkali metal alkoxides are preferred because they match the solvent system (e.g., sodium methoxide in methanol), preventing transesterification side reactions that could occur with mismatched alkoxide/solvent pairs. This mechanistic precision ensures high selectivity and minimizes the formation of mono-substituted impurities, which is crucial for maintaining the purity specifications required for API intermediates.
From an impurity control perspective, this mechanism offers distinct advantages over acid chloride routes. In traditional methods, the evolution of HCl gas requires stoichiometric amounts of base scavengers like pyridine or triethylamine, leading to difficult-to-remove ammonium salt byproducts. In the present invention, the byproduct is simply an alcohol (from the leaving group) and the salt of the acid used in the quench step (e.g., sodium acetate if acetic acid is used). These byproducts are highly water-soluble and can be easily removed during the aqueous workup phase described in the patent examples. The ability to tune the reaction temperature between -20°C and 50°C further allows process chemists to kinetically control the reaction rate, suppressing thermal degradation pathways. This level of control is essential for ensuring that the final product meets the rigorous impurity profiles demanded by regulatory bodies for cost reduction in electronic chemical manufacturing and pharmaceutical applications alike.
How to Synthesize N,N'-Dialkoxy-N,N'-Dialkyl Oxalamide Efficiently
Implementing this synthesis on a pilot or production scale requires careful attention to reagent addition rates and thermal management to maximize the reported yields of up to 96.0%. The general procedure involves charging a reactor with the oxalic acid diester, the hydroxylamine hydrochloride salt, and a solvent such as methanol or ethanol. The mixture is cooled to a range of 5°C to 10°C before the slow, dropwise addition of the base solution, typically a 28% sodium methoxide in methanol solution. Maintaining this temperature window during the exothermic addition is vital to prevent local hot spots that could degrade the sensitive hydroxylamine functionality. Following the addition, the reaction is stirred for approximately 3 hours to ensure complete conversion. The detailed standardized synthesis steps, including specific molar ratios and workup procedures derived from the patent examples, are outlined below.
- Prepare the reaction mixture by combining oxalic acid diester, N-alkyl-O-alkylhydroxylamine hydrochloride, and a suitable alcohol solvent in a reactor equipped with stirring and temperature control.
- Maintain the reaction temperature between -20°C and 50°C while slowly adding a solution of alkali metal alkoxide (e.g., sodium methoxide) to initiate the aminolysis reaction.
- Upon completion, quench the reaction with dilute acid, extract the product into an organic solvent such as ethyl acetate, and purify via crystallization or distillation.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the transition to this ester-based methodology offers profound logistical and financial benefits that extend beyond simple reagent costs. The elimination of oxalyl chloride removes a Tier-1 safety hazard from the facility, potentially lowering insurance premiums and reducing the need for specialized scrubbing systems required for phosgene containment. Furthermore, the shift away from methylene chloride to greener solvents like ethanol or isopropanol aligns with increasingly strict environmental regulations regarding volatile organic compound (VOC) emissions. This alignment ensures long-term operational continuity without the risk of sudden regulatory shutdowns. The robustness of the reaction conditions, which tolerate a wide range of temperatures and solvents, also enhances supply chain reliability by allowing for flexibility in raw material sourcing. If a specific solvent becomes scarce, the process can be adapted to use alternatives like acetonitrile or toluene without compromising the reaction outcome, thereby reducing lead time for high-purity pharmaceutical intermediates.
- Cost Reduction in Manufacturing: The economic argument for this process is compelling when analyzing the total cost of ownership. By utilizing oxalic acid diesters, which are commodity chemicals produced on a massive scale, the raw material input costs are significantly stabilized compared to the volatile pricing of specialty acid chlorides. Additionally, the simplified workup procedure—often involving a simple acid quench followed by extraction and crystallization—reduces the number of unit operations required. Fewer processing steps mean lower energy consumption, reduced labor hours, and decreased solvent usage per kilogram of product. The high yields reported, reaching up to 96.0% in optimized examples, directly translate to improved atom economy and less waste generation. This efficiency drives down the cost per kilogram of the final active ingredient, providing a competitive edge in cost reduction in pharmaceutical intermediates manufacturing.
- Enhanced Supply Chain Reliability: Supply chain resilience is bolstered by the use of widely available starting materials. Oxalic acid diesters and N-alkyl-O-alkylhydroxylamine salts are produced by numerous global chemical manufacturers, reducing the risk of single-source dependency. The stability of these reagents allows for bulk purchasing and long-term storage without significant degradation, enabling strategic stockpiling to buffer against market fluctuations. Moreover, the mild reaction conditions (-20°C to 50°C) do not require extreme cryogenic cooling or high-pressure equipment, meaning the process can be executed in standard glass-lined or stainless steel reactors found in most multipurpose chemical plants. This compatibility ensures that production slots can be secured easily, facilitating the commercial scale-up of complex polymer additives or fine chemicals without requiring capital-intensive plant modifications.
- Scalability and Environmental Compliance: Scaling this process from grams to tons is straightforward due to the homogeneous nature of the reaction mixture and the manageable exotherm. The ability to control the reaction rate through dosing allows for safe operation in large vessels, mitigating the risk of thermal runaway. From an environmental standpoint, the absence of chlorinated solvents and toxic gases simplifies waste stream management. The aqueous waste generated contains primarily inorganic salts and alcohols, which are easier to treat in standard wastewater treatment facilities compared to halogenated organic waste. This 'green' profile not only reduces disposal fees but also enhances the corporate sustainability metrics of the manufacturing entity, a factor that is increasingly weighted in vendor selection processes by major multinational corporations seeking eco-friendly partners.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this patented technology. Understanding these nuances is critical for R&D teams evaluating route feasibility and procurement officers assessing vendor capabilities. The answers are derived directly from the experimental data and technical disclosures within Patent CN100386310C, ensuring accuracy and relevance to real-world production scenarios. We encourage stakeholders to review these points to gain a clearer picture of how this methodology can be integrated into existing manufacturing portfolios.
Q: Why is the oxalate ester route preferred over oxalyl chloride for industrial synthesis?
A: The oxalate ester route eliminates the use of oxalyl chloride, which decomposes into toxic phosgene and carbon monoxide. Additionally, it avoids carcinogenic solvents like methylene chloride, significantly improving operator safety and waste disposal compliance.
Q: What yields can be expected from this base-catalyzed aminolysis process?
A: Experimental data from Patent CN100386310C demonstrates isolation yields ranging from 61.6% to as high as 96.0%, depending on the specific ester substrate and reaction conditions employed.
Q: Can this process be scaled for commercial API intermediate production?
A: Yes, the process utilizes mild temperatures (-20°C to 50°C) and commercially available reagents like sodium methoxide and dimethyl oxalate, making it highly suitable for multi-ton scale-up without requiring specialized cryogenic equipment.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable N,N'-Dialkoxy-N,N'-Dialkyl Oxalamide Supplier
At NINGBO INNO PHARMCHEM, we recognize that the successful translation of laboratory patents into commercial reality requires more than just chemical knowledge; it demands engineering excellence and unwavering commitment to quality. As a premier CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production. Our facilities are equipped with the necessary infrastructure to handle the base-catalyzed aminolysis process safely and efficiently, adhering to stringent purity specifications and operating within rigorous QC labs. We understand that for your projects, consistency is key, and our process validation protocols ensure that every batch of N,N'-dialkoxy-N,N'-dialkyl oxalamide meets the exacting standards required for downstream API synthesis.
We invite you to collaborate with us to leverage this advanced synthetic route for your next project. Our technical team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements and purity needs. We encourage you to contact our technical procurement team today to request specific COA data and route feasibility assessments. By partnering with us, you secure not just a supplier, but a strategic ally dedicated to optimizing your supply chain and accelerating your time to market with high-quality, cost-effective chemical solutions.
