Scalable Synthesis of Penicinotam via Novel Gould-Jacob and Suzuki Coupling Strategies
Scalable Synthesis of Penicinotam via Novel Gould-Jacob and Suzuki Coupling Strategies
The development of efficient synthetic routes for bioactive natural products remains a cornerstone of modern agrochemical and pharmaceutical research. Patent CN108623581B discloses a groundbreaking methodology for the preparation of Penicinotam, a potent compound exhibiting significant pesticidal activity. This technical insight report analyzes the novel synthetic pathway which diverges sharply from traditional approaches by leveraging a robust Gould-Jacob reaction sequence followed by a sophisticated Suzuki-Miyaura cross-coupling. The strategic redesign of the synthetic tree not only addresses critical safety concerns associated with legacy processes but also establishes a scalable framework suitable for industrial manufacturing. By shifting the paradigm from hazardous cyanohydrin chemistry to a more benign condensation and cyclization strategy, this innovation offers a compelling value proposition for stakeholders focused on sustainable and cost-effective production of complex heterocyclic intermediates.
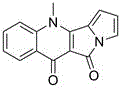
The structural complexity of Penicinotam, characterized by its fused quinoline and pyrrole moieties, presents unique challenges in regioselective functionalization. The patented method successfully navigates these challenges through a meticulously orchestrated six-step sequence that ensures high purity and yield. For R&D directors and process chemists, the elimination of toxic reagents while maintaining high atom economy represents a significant advancement in green chemistry principles applied to agrochemical intermediate synthesis. This report delves deep into the mechanistic nuances and commercial implications of this route, providing a comprehensive overview for decision-makers evaluating supply chain partners for high-value bioactive compounds.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of quinolone-based natural products like Penicinotam has been plagued by reliance on hazardous reagents and convoluted reaction sequences that hinder scalability. Prior art, including methods referenced in background literature, often necessitates the use of acetone cyanohydrin, a substance classified as highly toxic and posing severe risks to both human health and environmental safety. The handling of such dangerous materials requires specialized infrastructure, rigorous safety protocols, and expensive waste treatment systems, all of which drastically inflate the operational expenditure of manufacturing facilities. Furthermore, conventional routes frequently suffer from poor regioselectivity during the construction of the quinoline core, leading to complex mixtures of isomers that are difficult and costly to separate. These purification bottlenecks not only reduce overall process yield but also introduce variability in the quality of the final active pharmaceutical or agrochemical ingredient, creating unacceptable risks for downstream formulation.
The Novel Approach
In stark contrast, the methodology outlined in CN108623581B introduces a streamlined and safer alternative that fundamentally reimagines the construction of the quinoline scaffold. By initiating the synthesis with the condensation of aniline and ethyl ethoxymethylenemalonate, the process utilizes readily available, commodity-grade starting materials that are abundant in the global chemical market. This Gould-Jacob reaction variant efficiently constructs the quinoline parent ring under thermal conditions, bypassing the need for toxic cyanide sources entirely. The subsequent functionalization steps, including selective halogenation and palladium-catalyzed coupling, are designed to proceed with high fidelity, minimizing the formation of byproducts. This shift towards safer reagents and more predictable reaction outcomes translates directly into reduced regulatory burden and lower capital investment for safety compliance, making the process inherently more attractive for large-scale commercial adoption in the agrochemical sector.
Mechanistic Insights into Gould-Jacob Cyclization and Suzuki Coupling
The heart of this synthetic strategy lies in the precise execution of the Gould-Jacob reaction, which serves as the foundation for building the heterocyclic core. The mechanism involves the initial nucleophilic attack of the aniline nitrogen on the electrophilic carbon of the ethoxymethylenemalonate, followed by a thermal electrocyclic ring closure that aromatizes to form the 4-hydroxyquinoline derivative. This step is critical as it sets the stereochemical and electronic stage for all subsequent transformations. Following the formation of the quinoline core, the process employs a sequential halogenation strategy where the 4-hydroxy group is first converted to a chloride, activating the ring for further substitution. Subsequently, an oxidative bromination protocol is utilized to introduce a bromine atom selectively at the 2-position. This regioselective halogenation is paramount, as it creates the necessary handle for the pivotal carbon-carbon bond-forming event that follows, ensuring that the pyrrole moiety is attached at the correct position to mimic the natural product's bioactivity.

The crown jewel of this synthetic route is the Suzuki-Miyaura cross-coupling reaction, which joins the functionalized quinoline fragment with the N-protected pyrrole boronic acid. This transition metal-catalyzed transformation is renowned for its tolerance of various functional groups, yet it requires careful optimization when dealing with electron-deficient heterocycles. The patent specifies the use of palladium catalysts, such as palladium acetate or tetrakis(triphenylphosphine)palladium, in conjunction with bases like cesium carbonate to facilitate the transmetallation and reductive elimination steps. The choice of solvent, typically a mixture involving dioxane or toluene, plays a crucial role in solubilizing the inorganic base and stabilizing the catalytic cycle. Following the coupling, the removal of the tert-butoxycarbonyl (Boc) protecting group under Brønsted acidic conditions reveals the free pyrrole nitrogen, setting the stage for the final intramolecular cyclization. This final ring-closing step, mediated by a base at elevated temperatures, forms the second lactam ring, completing the intricate polycyclic architecture of Penicinotam with high structural integrity.
How to Synthesize Penicinotam Efficiently
Executing this synthesis requires strict adherence to the reaction parameters defined in the patent to ensure optimal yield and purity profiles suitable for commercial applications. The process begins with the high-temperature condensation of aniline derivatives, followed by a series of workup procedures involving extraction and chromatography to isolate key intermediates like the 4-chloroquinoline-3-carboxylic acid ethyl ester. Each step, from the oxidative bromination to the final cyclization, demands precise control over temperature and stoichiometry to prevent side reactions such as over-halogenation or hydrolysis of the ester groups. The detailed standard operating procedures for each transformation, including specific solvent choices and reaction times, are critical for reproducibility. For a comprehensive breakdown of the exact experimental conditions and stoichiometric ratios required to replicate this high-yielding pathway, please refer to the standardized synthesis guide below.
- Condense aniline with ethyl ethoxymethylenemalonate followed by Gould-Jacob cyclization to form the quinoline core.
- Perform chlorination at the 4-position followed by selective oxidative bromination at the 2-position of the quinoline ring.
- Execute Suzuki-Miyaura coupling with N-Boc-pyrrole boronic acid, followed by deprotection and base-mediated cyclization to yield Penicinotam.
Commercial Advantages for Procurement and Supply Chain Teams
From a procurement and supply chain perspective, the adoption of this novel synthetic route offers transformative benefits that extend far beyond simple chemical efficiency. The primary advantage lies in the drastic simplification of the raw material portfolio, shifting away from specialized, high-risk chemicals to bulk commodities that are universally accessible. This transition mitigates the risk of supply disruptions caused by regulatory crackdowns on toxic substances, ensuring a more resilient and continuous flow of materials into the production facility. Furthermore, the elimination of hazardous waste streams associated with cyanide chemistry significantly reduces the environmental compliance costs and liability exposure for the manufacturer. These factors combine to create a supply chain that is not only more robust against external shocks but also aligned with the increasingly stringent sustainability mandates imposed by global regulatory bodies and end-users in the agrochemical industry.
- Cost Reduction in Manufacturing: The economic implications of switching to this aniline-based route are profound, primarily driven by the removal of expensive safety infrastructure and waste disposal costs. By eliminating the need for acetone cyanohydrin, manufacturers avoid the substantial expenses related to specialized storage, handling equipment, and the neutralization of toxic effluents. Additionally, the use of commodity starting materials like aniline and diethyl ethoxymethylenemalonate ensures that raw material costs remain stable and predictable, shielding the production budget from the volatility often seen in niche reagent markets. The improved selectivity of the Gould-Jacob and Suzuki steps also means higher crude yields, which reduces the load on downstream purification units and lowers the consumption of solvents and silica gel, further driving down the overall cost of goods sold.
- Enhanced Supply Chain Reliability: Supply chain continuity is significantly bolstered by the reliance on widely produced chemical feedstocks that are available from multiple global vendors. Unlike proprietary or highly regulated reagents that may have single-source dependencies, aniline and malonate esters are produced at massive scales for various industries, guaranteeing availability even during market fluctuations. This diversification of the supplier base reduces the leverage of any single vendor and allows procurement teams to negotiate more favorable terms. Moreover, the simplified logistics of transporting non-hazardous materials streamline the inbound supply chain, reducing lead times and minimizing the administrative burden associated with shipping dangerous goods, thereby enhancing the overall agility of the manufacturing operation.
- Scalability and Environmental Compliance: The process is inherently designed for scale-up, with reaction conditions that are easily transferable from laboratory glassware to industrial reactors without requiring exotic equipment. The absence of highly toxic intermediates simplifies the environmental permitting process, allowing for faster expansion of production capacity to meet growing market demand. Waste generation is minimized through high-yielding steps and the avoidance of heavy metal contaminants often associated with less optimized coupling reactions. This alignment with green chemistry principles not only future-proofs the manufacturing site against tightening environmental regulations but also enhances the brand reputation of the final agrochemical product among environmentally conscious consumers and distributors.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this Penicinotam synthesis route. These answers are derived directly from the technical specifications and beneficial effects described in the patent documentation, aiming to clarify the operational advantages for potential partners. Understanding these nuances is essential for R&D and procurement teams evaluating the feasibility of integrating this intermediate into their broader value chain. The responses highlight the balance between chemical innovation and practical manufacturability that defines this new approach.
Q: Why is the new Penicinotam synthesis route safer than previous methods?
A: The novel route eliminates the use of acetone cyanohydrin, a highly toxic reagent found in conventional methods, replacing it with safer, commodity-grade starting materials like aniline and malonate esters.
Q: What represents the key challenge in the Suzuki coupling step for this molecule?
A: The key challenge lies in coupling the sterically hindered 2-bromo-4-chloroquinoline intermediate with the pyrrole boronic acid without causing deboronation or homocoupling, which is managed by optimizing palladium catalyst loading and base selection.
Q: How does this process impact supply chain stability for agrochemical manufacturers?
A: By utilizing widely available bulk chemicals such as aniline and avoiding specialized, hazardous reagents, the process ensures a robust and continuous supply chain with reduced regulatory hurdles for raw material procurement.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Penicinotam Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of adopting advanced synthetic methodologies to maintain competitiveness in the global agrochemical market. Our team of expert process chemists possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory discovery to industrial reality is seamless and efficient. We are committed to delivering high-purity Penicinotam intermediates that meet stringent purity specifications, supported by our rigorous QC labs equipped with state-of-the-art analytical instrumentation. Our capability to implement the novel Gould-Jacob and Suzuki coupling strategies described in CN108623581B allows us to offer a superior product profile with a significantly reduced environmental footprint, aligning perfectly with the sustainability goals of modern agrochemical enterprises.
We invite you to collaborate with us to leverage these technological advancements for your specific application needs. Our technical sales team is prepared to provide a Customized Cost-Saving Analysis that quantifies the economic benefits of switching to this safer, more efficient supply source. We encourage you to contact our technical procurement team today to request specific COA data and route feasibility assessments tailored to your volume requirements. By partnering with NINGBO INNO PHARMCHEM, you secure a reliable supply of high-quality intermediates while optimizing your manufacturing costs and enhancing your supply chain resilience.
