Scalable Aqueous Synthesis of 2-Mercaptopyridine and 2,2'-Pyridyl Sulfide for Global Markets
The chemical industry is constantly seeking more efficient pathways to produce high-value heterocyclic compounds, and Patent CN113387880A presents a significant breakthrough in the integrated preparation of 2-mercaptopyridine and 2,2'-pyridine thioether. This innovative technology addresses critical pain points in the synthesis of sulfur-containing pyridine derivatives, which are essential building blocks for a wide range of pharmaceutical and agrochemical applications. By utilizing a unique composite catalytic system involving cesium acetate and specific cocatalysts in an aqueous medium, the process achieves the simultaneous production of two distinct valuable products from a single reaction pot. This dual-output capability not only maximizes atom economy but also drastically simplifies the downstream processing requirements typically associated with multi-step syntheses. For global procurement teams and R&D directors, this represents a paradigm shift towards greener, more cost-effective manufacturing strategies that align with modern sustainability goals.
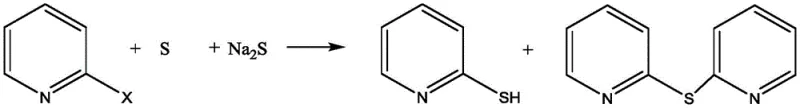
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditionally, the synthesis of 2-mercaptopyridine and its dimeric thioether counterparts has relied on methodologies that are fraught with significant operational and safety challenges. Conventional routes often employ 2-hydroxypyridine as a starting material, reacting it with hazardous reagents such as phosphorus pentasulfide or sodium hydrosulfide under harsh conditions. These methods frequently necessitate the use of expensive and toxic transition metal catalysts, such as copper complexes, which introduce the risk of heavy metal contamination in the final product—a critical failure point for pharmaceutical intermediates. Furthermore, many legacy processes depend on volatile organic solvents that pose substantial safety risks regarding flammability and toxicity, while also generating complex waste streams that are difficult and costly to treat. The inability to efficiently co-produce related thioethers often results in lower overall yields and increased raw material consumption, driving up the total cost of ownership for manufacturers.
The Novel Approach
In stark contrast, the novel integrated preparation method disclosed in the patent utilizes 2-halogenated pyridine, specifically 2-chloropyridine or 2-bromopyridine, as a robust and inexpensive starting material. The core innovation lies in the deployment of a composite catalytic system where cesium acetate acts as the primary catalyst, synergistically enhanced by cocatalysts such as EDTA, tetrabutylammonium bromide (TBAB), or L-proline. This system operates effectively in water, a benign and non-flammable solvent, thereby eliminating the safety hazards associated with organic media. The reaction proceeds under reflux conditions to simultaneously yield both 2-mercaptopyridine and 2,2'-pyridine thioether, effectively doubling the utility of the input materials. This approach not only reduces the generation of unwanted byproducts but also streamlines the purification process through simple extraction and crystallization steps, offering a clear pathway for cost reduction in pharmaceutical intermediate manufacturing.
Mechanistic Insights into Aqueous Phase Catalytic Substitution
The mechanistic foundation of this process relies on a nucleophilic aromatic substitution facilitated by the unique properties of the cesium-based catalytic system in an aqueous environment. The sulfur source, derived from the combination of elemental sulfur and sodium sulfide, generates reactive polysulfide species in situ. The cesium cation, due to its large ionic radius and low charge density, acts as an effective phase transfer agent, enhancing the nucleophilicity of the sulfur species towards the electron-deficient carbon at the 2-position of the pyridine ring. The addition of cocatalysts like EDTA or TBAB further stabilizes the reaction intermediates and improves the solubility of the organic substrates in the aqueous phase, ensuring a homogeneous reaction environment that drives high conversion rates. This careful balance of catalytic components prevents the formation of complex polymeric sulfur byproducts that often plague traditional sulfuration reactions.
From an impurity control perspective, the absence of transition metals is a decisive advantage for producing high-purity organic chemicals. Traditional copper-catalyzed routes often leave trace metal residues that require rigorous and expensive purification steps, such as chelation or specialized filtration, to meet regulatory standards for API intermediates. By utilizing an alkali metal salt (cesium acetate) and organic cocatalysts, the new method ensures that the final product profile is free from heavy metal contaminants. The separation strategy leverages the differential solubility of the two products: the neutral 2,2'-pyridine thioether is extracted into an organic solvent like ethyl acetate, while the acidic 2-mercaptopyridine remains in the aqueous phase until acidification induces crystallization. This orthogonal separation mechanism ensures high purity for both outputs without the need for complex chromatographic techniques.
How to Synthesize 2-Mercaptopyridine Efficiently
The synthesis protocol outlined in the patent provides a robust framework for laboratory and pilot-scale production, emphasizing simplicity and reproducibility. The process begins with the preparation of the catalytic solution, followed by the controlled addition of the halogenated substrate and a prolonged reflux period to ensure complete conversion. Post-reaction workup involves a straightforward liquid-liquid extraction to separate the neutral thioether, followed by pH adjustment of the aqueous layer to isolate the mercaptan. This streamlined workflow minimizes unit operations and reduces the potential for product loss during transfer and handling. For detailed operational parameters and safety guidelines, please refer to the standardized synthesis steps provided below.
- Dissolve sulfur, sodium sulfide, and a composite catalyst (cesium acetate with EDTA, TBAB, or L-Proline) in water under heating at 90°C.
- Add 2-halogenated pyridine (chloro or bromo) to the mixture and reflux for 20-30 hours to facilitate nucleophilic substitution.
- Cool the reaction, extract with ethyl acetate or ether to separate the thioether, and acidify the aqueous layer to crystallize 2-mercaptopyridine.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain leaders, the adoption of this integrated synthesis method offers tangible strategic benefits that extend beyond mere chemical efficiency. The shift to water as a primary solvent significantly lowers the logistical burden and storage costs associated with managing large volumes of flammable organic solvents. Additionally, the use of commodity chemicals like 2-chloropyridine and sodium sulfide ensures a stable and resilient supply chain, mitigating the risks associated with sourcing specialized or scarce reagents. The ability to produce two marketable products from a single batch effectively diversifies the revenue stream from one production run, enhancing overall asset utilization and return on investment for manufacturing facilities.
- Cost Reduction in Manufacturing: The elimination of expensive transition metal catalysts and the replacement of hazardous organic solvents with water lead to a substantial decrease in raw material and waste disposal costs. The simplified workup procedure, which relies on basic extraction and crystallization rather than complex purification technologies, further reduces energy consumption and labor hours per kilogram of product. This holistic reduction in operational expenditure allows for more competitive pricing structures in the global market for fine chemical intermediates.
- Enhanced Supply Chain Reliability: By utilizing widely available starting materials such as 2-halogenated pyridines and common inorganic salts, the process minimizes dependency on single-source suppliers for exotic reagents. The robustness of the aqueous reaction system also implies greater tolerance to minor variations in feedstock quality, ensuring consistent production output even under fluctuating supply conditions. This reliability is crucial for maintaining continuous production schedules and meeting the just-in-time delivery requirements of downstream pharmaceutical clients.
- Scalability and Environmental Compliance: The inherent safety of the water-based process facilitates easier scale-up from pilot plants to commercial production without the need for extensive explosion-proof infrastructure. Furthermore, the reduction in hazardous waste generation aligns with increasingly stringent environmental regulations, reducing the compliance burden and potential liability for manufacturing sites. This green chemistry approach not only future-proofs the production asset but also enhances the corporate sustainability profile, which is becoming a key differentiator in B2B procurement decisions.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this integrated preparation technology. These insights are derived directly from the experimental data and comparative examples provided in the patent documentation, offering a transparent view of the process capabilities. Understanding these details is essential for technical teams evaluating the feasibility of adopting this route for their specific product portfolios.
Q: What are the primary advantages of using water as a solvent in this synthesis?
A: Using water eliminates the safety hazards and high costs associated with volatile organic solvents, while simplifying waste treatment and reducing environmental impact.
Q: How does this method improve product purity compared to traditional transition metal catalysis?
A: By avoiding transition metal catalysts like copper, the process prevents heavy metal residue contamination, which is critical for pharmaceutical grade intermediates.
Q: Can this process be scaled for industrial production of agrochemical intermediates?
A: Yes, the use of cheap raw materials like 2-chloropyridine and simple extraction workups makes the process highly scalable and cost-effective for bulk manufacturing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 2-Mercaptopyridine Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of this aqueous catalytic technology for the production of high-value sulfur heterocycles. As a leading CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory discovery to industrial reality is seamless and efficient. Our state-of-the-art facilities are equipped with rigorous QC labs capable of verifying stringent purity specifications, guaranteeing that every batch of 2-mercaptopyridine or 2,2'-pyridine thioether meets the exacting standards required for pharmaceutical and agrochemical applications.
We invite you to collaborate with us to leverage this advanced synthesis route for your supply chain needs. Our technical team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements, demonstrating how this method can optimize your total cost of ownership. Please contact our technical procurement team today to request specific COA data and route feasibility assessments, and let us help you secure a sustainable and cost-effective supply of these critical intermediates.
